Trifluoroacetic anhydride didn’t arrive in the chemical world by accident. It emerged from the curiosity of chemists exploring the potential of fluorinated compounds in synthesis and industry, driven by the remarkable ways fluorine can alter the properties of organic molecules. In the decades following the mid-1900s—when organic fluorine chemistry started catching fire—this molecule quickly became more than a laboratory curiosity. Its power to alter reactivity, tune selectivity, and toughen molecular structures made it an essential building block for labs across the globe. I’ve seen it move from niche availability to widespread adoption, especially as demand for fluorinated pharmaceuticals climbed.
Trifluoroacetic anhydride stands out with its sharp, pungent odor and almost eerie clarity. Usually it’s clear and colorless, with a low boiling point and a tendency to bite you with fumes if you open the bottle careless. On a molecular level, two trifluoroacetyl groups are linked by an oxygen bridge, set up to transfer that acyl group in a flash. What’s always impressed me is how reactive it remains, even stored on a shelf, as long as water doesn’t sneak in. Chemists appreciate how its structure, thanks to those trifluoromethyl groups, provides a unique blend of electronegativity and steric protection—you see it handle acylation or dehydration reactions that other reagents just can’t touch with the same finesse.
Making trifluoroacetic anhydride doesn’t call for magic, just careful design. Industrially, it often comes from the reaction between trifluoroacetic acid and a strong dehydrating agent, such as phosphorus pentachloride or thionyl chloride. I recall the first synthesis I watched: the solid acid got doused in a measured stream of chlorinating agent, producing a hiss, a cloud, and soon, shimmering product distilled from the mix. Once produced, clear labeling becomes a matter of safety. Bottles in every lab carry hazard diamonds and warnings—this isn’t a friendly solvent you spill and shrug off. Quality control backs up purity claims, including GC-MS or NMR scans hiding in a drawer in the lab manager’s office if anyone doubts what’s in the bottle.
Few reagents in the modern organic lab do so much, so quickly. Trifluoroacetic anhydride brings power to acylation reactions, speeding up ester and amide formation, and helping activate carboxylic acids. Medicinal chemists reach for this stuff to shield or modify reactive sites, always chasing the next best drug candidate by tweaking molecular fingerprints. Polymer chemists lean on it, too, especially when building materials that must survive heat, corrosion, or everyday chemical abuse. This isn’t just for esoteric academic papers—it lands in agriculture, electronics, and elsewhere. With this much flexibility, it’s no surprise that new applications keep cropping up every year.
In the wilds of research, language gets slippery. Trifluoroacetic anhydride, TFAA, or trifluoroacetyl anhydride—each name calls up the same substance. Browsing papers from Japan to Germany, you see a half-dozen variations, but the chemistry always points back to that telltale smell and bite. Markets sometimes turn up branded synonyms, but the chemistry community usually sticks to the core names.
Safety protocols for trifluoroacetic anhydride read as serious business—and they aren’t written for show. The liquid tears up mucous membranes and skin on contact, stings nostrils, and reacts fiercely with water to form trifluoroacetic acid—all hazards I’ve seen firsthand in teaching labs. Gloves, face shields, and working in a fume hood aren’t suggestions; they mark the difference between an accident and a normal workday. Before anyone opens a bottle, they check chemical compatibility and spill kits. Regulations like OSHA push for strict handling, and most labs lean hard on written procedures, sometimes even audit logs, to track each transfer or dilution.
Looking ahead, the story of trifluoroacetic anhydride stands far from finished. The hunger for new drugs, materials, and crop protectants keeps pushing its chemistry into untested territory. Fluorinated building blocks play a central role in everything from replacing outdated pesticides to radical drug design strategies. At the same time, concerns over sustainability and toxicity push researchers into tough questions about safer disposal, greener synthesis, and alternative reagents. Some labs work on catalyst-based methods to trim the waste and energy consumption; others consider recycling spent material or designing alternatives with shorter environmental tails. Things get lively in regulatory discussions—governing bodies double-check allowable uses and push for transparency, especially as analytical methods become sharper at catching trace remnants in pharmaceuticals or consumer goods.
Toxicological studies on trifluoroacetic anhydride grow with each decade, led by concerns in drug manufacturing and environmental protection. The parent acid, trifluoroacetic acid, lingers in the environment; this isn’t a trivial matter in watersheds or near pharmaceutical plants. Long-term health data on the anhydride itself show it doesn’t belong anywhere near unprotected skin or lungs. Regulatory agencies watch for chronic exposure data and keep raising the bar for permissible workplace concentrations. Responsible labs have skin-in-the-game and tend to update internal documents regularly, responding to new research quickly—nobody wants a repeat of the days when safety data lagged behind actual practice by a generation.
Chemists know a workhorse when they meet one. Trifluoroacetic anhydride continues to support new syntheses, drug design, and the evolution of materials. In my experience, grant applications mention it as part of ambitious projects, and review committees expect to see its safe, justified use wherever new scaffolds or protective groups are central. The push for “greener” chemistry, though, stays persistent. Researchers who remember their mentors’ stories of early mishaps now drive improvements in training, documentation, and waste handling. Looking at the next decade, it’s clear this reagent will keep adapting—spurred by both the creative work in synthesis and the sobering responsibilities of modern chemical stewardship.
Every chemical has a story, and some get more attention than others. Trifluoroacetic anhydride, or TFAA as most chemists know it, punches above its weight class in research labs and industrial shops. You probably won’t hear about it outside specialist circles, but its usefulness stretches into fields like medicine, agriculture, and electronics — usually far from the headlines.
TFAA shows up as a catalyst or reagent in chemical synthesis. Back in grad school, we used TFAA to tweak molecules in ways simpler acids can't manage. It packs a punch, making molecules more reactive or protecting chemical groups during complex processes. This chemical's unique properties let it swap out functional groups quickly, so building blocks for new medicines or advanced materials often trace their heritage to reactions using TFAA.
The pharmaceutical industry counts on precision. One slip, and you’re stuck with a compound that won’t pass muster. TFAA helps chemists add fluoro groups or protect sensitive parts of a molecule, clearing the way for more selective reactions. Take antiviral drugs or some painkillers as examples. Their synthesis often depends on well-timed additions and removals of parts of the molecule — TFAA makes those steps efficient and reliable.
Few outside agrochemical development realize how many crop-protection agents come from finely tuned chemical reactions. TFAA works behind the scenes to give rise to new herbicides and pesticides, helping wheat fields and rice paddies deliver the yields we count on. Precision in synthesis means these compounds break down when intended rather than sticking around in water or soil. By working with TFAA, scientists can shape molecules to create effective solutions with less environmental drag.
Electronics keep getting smaller and more complex. Manufacturers use organic molecules to create specialty plastics and coatings that protect your phone or help solar panels run longer. TFAA forms part of the route for fluorinated polymers—these materials resist heat, chemical spills, and wear. Without chemicals like TFAA moving reactions further, much of our miniaturized tech would be vulnerable or unreliable.
Not every story about a clever chemical ends well. TFAA has a reputation for being tough to handle. It reacts quickly, and contact with water or moisture can produce corrosive acids like trifluoroacetic acid. Laboratories require strict ventilation and careful waste disposal routines. This isn’t an ordinary risk; just a splash or whiff can mean trouble for people in the room. Some researchers have asked for greener alternatives, but many syntheses have no substitute for what TFAA brings to the table.
I think more thought can go into how labs use and dispose of reagents like TFAA. Chemists are quick to adopt new practices when they’re shown a safer or more sustainable route. Solvent recycling and novel containment devices already make workplaces safer and limit environmental release. Regulatory agencies keep a close watch, and academic groups keep chasing new ways to cut down on hazardous waste or use less aggressive chemicals — but practical options aren’t always ready. Giving more people a window into the purpose and risks behind chemicals like TFAA could keep innovation thriving with responsibility front and center.
Few substances in a chemist’s cabinet command quite as much respect as trifluoroacetic anhydride. This isn’t just another solvent or simple reagent. Its strong reactivity, combined with a reputation for causing harm if mishandled, means every bottle serves as a reminder that safety isn’t box-ticking but a commitment.
Anyone who’s had a splash of even dilute acid on their skin knows that discomfort can spiral fast. Trifluoroacetic anhydride brings that to another level. The liquid releases corrosive fumes and attacks skin and eyes almost instantly. This isn’t just a minor irritation; exposure demands immediate rinsing and medical help. Splash goggles and face shields don’t just look serious—they are. Nitrile gloves and chemical-resistant aprons matter because lab coats are no match for something this aggressive.
I’ve stood in rooms where a whiff of the wrong reagent left the air tasting sharp. Ventilation, real ventilation—not just a cracked window—is essential with chemicals as volatile as this. A certified chemical fume hood changes the game. Without it, fumes fill a room, risking respiratory burns. Respirators rated for acidic vapors have become, for some jobs, as basic as wearing closed shoes.
There’s an old lab joke about ‘adding acid to water, never water to acid,’ but with trifluoroacetic anhydride, stakes run higher. Even small spills meeting moisture can trigger violent reactions, shooting out corrosive spray. So, it’s wise to keep sources of water far away when working with the neat liquid. Spills on benches demand spill pillows and specialty adsorbents, not paper towels or rags.
Cabinets for this kind of material look different. Forget the main stockroom shelf—dry, cool, and well-labeled secondary containment is non-negotiable. Metal shelving can corrode, and wooden ones don’t hold out against spills. Dedicated secondary containers—hard plastic bins—give an extra layer of protection. Forgetting to check for leaks or old containers spells disaster, so inspecting these storage spots becomes second nature.
Mistakes develop faster when working tired, distracted, or rushed. Pre-planning before opening a fresh bottle saves trouble later. Emergency eyewash stations and showers must work and sit within reach, not down the hall or in another room. Having solid neutralizers and plenty of spill kits within arm’s length isn’t an option you skip because ‘it’ll be fine.’
Reading a safety data sheet means little if the information doesn’t translate to real action. Stories pass through labs—near-misses and actual injuries—reminding everyone that checking for cracked gloves or old face shields always pays off. Pairing up when working with dangerous reagents, even just for moral support and an extra pair of eyes, cuts down on risk. Training doesn’t stop after a first day; continual walkthroughs and practice make responses second nature.
It pays to lean into less hazardous alternatives when possible. If a synthesis step can use a milder reagent, that’s usually worth the trade-off in time or yield. Investing in advanced ventilation and protective gear easily outweighs the cost of a single accident. Cultivating a culture where concerns get voiced freely keeps everyone safer.
Working with trifluoroacetic anhydride means respecting its power and putting practical safety first, every time.
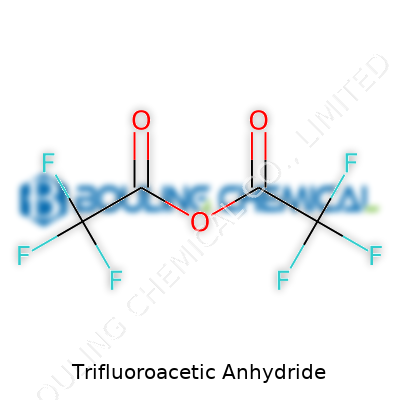
Many chemists flash a grin when they hear about Trifluoroacetic Anhydride. You see the name splashed across synthetic protocols, especially in organic synthesis work. Trifluoroacetic Anhydride, or TFAA as most folks call it, comes with a chemical formula of C4F6O3. Boiling down the structure, the molecule sports two trifluoroacetyl groups bound by an oxygen atom. Formally, chemists will draw it as (CF3CO)2O, which paints a direct picture of those two arms joined by an oxygen bridge. Each arm carries a strong trifluoromethyl punch, thanks to the CF3 pieces that grab attention with both their reactivity and their resistance to breakdown.
Anyone who’s spent hours in a lab appreciates shortcuts that don’t sacrifice quality. Trifluoroacetic Anhydride quickly became a darling in acetylation and dehydration reactions. The strength of the carbon-fluorine bonds keeps the molecule stable, yet the anhydride bridge makes it plenty eager to react with water and alcohols. That blend delivers reliability you don’t find by accident. Researchers often use it to introduce trifluoroacetyl groups, a trick that helps make molecules less prone to unwanted side reactions. Its strong electron-withdrawing effects also make molecular tweaking more predictable, especially when handling sensitive intermediates.
TFAA found its way onto my bench during a late-night project. The job called for selective dehydration in a step where traditional acetic anhydride would not cut it. The result was so clean I almost didn’t trust my own yield. Handling it showed me a side of chemistry where efficiency meets raw power—the stuff that runs on both theory and real-time gut checks. Seeing how fast TFAA pushes a reaction forward, I started to respect why many protocols describe it as “unforgiving” without enough ventilation or precise additions. The fumes carry that harsh kick, making you double check your safety glasses with each pour.
TFAA comes with clear benefits, but that intensity can spill over. The aggressive nature means even small mishandlings produce plenty of regret. Spills on a bench, or worse, on skin, demand immediate attention. The fumes impact not just the user, but the air quality in shared workspaces. This raises another talking point about tightening up lab practices. Investing in sealed reagent bottles, gloves that actually block acids, and efficient fume hoods isn’t just busywork—it’s a concrete way to prevent exposures and protect the team. Training matters, too. Watching a seasoned chemist handle TFAA teaches more about respect for chemicals than a dry safety handout ever could.
Making science safer isn’t just an abstract policy—it’s about dialing in the habits that keep discoveries flowing and people healthy. Trifluoroacetic Anhydride highlights a bigger lesson: chemicals offer incredible power, but that power needs channeling. No one wants to lose productivity or health over careless mistakes. Attention to detail and prioritizing safety gear means even a molecule with a reputation as sharp as TFAA fits into the lab with less drama and more results. Good practice carries more weight than any formula on paper.
Trifluoroacetic anhydride shows up in a lot of labs, especially for folks working in organic chemistry and pharmaceuticals. I remember cracking open a new bottle back in grad school, only to realize pretty quickly that this isn’t something you want lying around on a stockroom shelf with everyday chemicals. Getting storage right reduces the risk of accidents and protects the people who work with these materials every day.
This stuff is volatile and reacts with water. Even a little bit of humidity gets it to break down fast, releasing strong acids and fumes. You don’t want that contamination getting into other reagents, and you definitely don’t want those vapors anywhere near your breathing space. The level of irritation and corrosive damage this can cause to skin, eyes, and lungs isn't up for debate—a quick look at published lab accident reports makes that clear. There’s a reason regulatory guidelines call Trifluoroacetic anhydride a high hazard.
Working next to someone who ended up with a chemical burn after a careless spill left a mark on how I look at storage. The right setup isn’t just extra work or red tape—it’s the difference between a safe day in the lab and a ride to the hospital.
A cool, dry, well-ventilated spot in a chemical storage cabinet offers the best bet. Polypropylene or polyethylene containers, tightly sealed, do better than glass with regular stoppers because they keep out moisture and air. The closer you can keep it to room temperature or lower, the less risk of unexpected breakdown. Never put it near acids or bases, and keep oxidizers far away. Mixing fumes or a drop of the wrong liquid starts a chain reaction fast.
My old supervisor drilled this into us: always label the container clearly with the full name and hazard information—skip the abbreviations everyone forgets. Store it behind a lock if you can. This isn’t overkill; it’s prevention. Nobody wants a mix-up caused by a busy day or a distracted coworker.
Lab culture matters as much as the storage setup. Newer chemists watch what senior folks do, not just what’s written on a policy sheet. I’ve seen attitudes change for the better after a safety seminar included firsthand stories about what goes wrong when shortcuts get taken. Open discussion makes people more willing to push for new cabinets or better signage. Regular inspections should not feel like an interruption, but a sign someone has your back.
Waste management adds another layer. Trifluoroacetic anhydride doesn’t just vanish after use. Storing leftover material or contaminated disposables in robust, labeled containers until they’re properly neutralized keeps everyone safer, myself included. Rushing disposal or leaving residues behind risks accidents long after the experiment ends.
Institutions that invest in dedicated storage units and clear protocols see fewer incidents. I’ve seen simple improvements—like silica gel packs inside the container, mandatory double gloves during handling, and routine checks for leaks or loose caps—cut down risk. Training sessions that focus on the “why” behind each rule build respect that no printed instruction alone can cover.
Trifluoroacetic anhydride doesn’t forgive mistakes. Good storage isn’t just about following rules; it’s about respecting what the chemical can do. That mindset protects people, data, and careers. If you work around chemicals like this, demand the right storage and everyone walks away safer.
A lot of folks outside chemical labs probably haven’t heard of trifluoroacetic anhydride, but for researchers, it pops up in synthetic chemistry all the time. People familiar with it will tell you this stuff is not just some ordinary solvent you spill and wipe up. It’s corrosive, it fumes, and it can mess you up fast if you’re careless about your protective gear. Breathing in even trace amounts feels like someone’s dragging sandpaper through your nose and lungs. This isn’t just anecdotal: published medical case reports describe significant respiratory irritation and, over time, real risk to lung tissue.
The moment trifluoroacetic anhydride hits skin, it burns. It can blister with barely a drop. The eyes are even worse; direct splashes don’t just sting—they can cause lasting damage. A lot of chemists work with eye wash stations nearby for good reason. Even with a fume hood and nitrile gloves, accidents happen. Vapors are invisible and can escape unnoticed from an open vessel, spreading across the bench in seconds. I once saw a colleague drop a flask and lose a full layer of skin in less than a minute, despite rinsing immediately—a lesson that stays with you.
Stories like these sound dramatic, but safety data backs them up. OSHA materials, scientific journals, and chemical databases all point to acute toxicity, chemical burns, and problems ranging from headaches to pulmonary edema. A chemical that can chew through lab gloves is not something you want near your hands or eyes. The need for ventilation, splash-proof goggles, and chemical-resistant gloves is not negotiable.
Immediate action saves the day. No one should waste time scanning safety manuals mid-crisis. If it hits the skin, strip off contaminated clothing and rinse—fast, and keep rinsing for at least 15 minutes. If it gets in the eyes, hit the eyewash station and force them open under gently running water, no matter how much it hurts. Inhalation means fresh air and medical attention, period. Ingesting it? Head straight to emergency care. People get embarrassed or try to downplay lab accidents, but it’s better to overreact here. Hospitals need to know what they’re dealing with, so don’t skip details about the exposure.
Books and regulations give us the rules, but culture makes them stick. Too many young scientists learn by watching rather than reading. If senior staff ignore protocols or cut corners, others follow. Consistent safety briefings before working with aggressive agents like trifluoroacetic anhydride keep teams sharp. Posting emergency procedures near work benches isn’t a formality—people forget protocols in panic mode. Leadership should share near-misses and lessons learned openly, and there’s no shame in reviewing the basics.
Hard barriers, like splash guards and working under hoods, reduce risk, but they can’t replace a workforce that’s switched on and alert. Chemists should double-check labels and never pour from unmarked bottles. Disposable nitrile gloves can’t always stand up to powerful reagents—thicker, specialty gloves work better. People who’ve watched reactions run wild will tell you that good habits and a healthy respect for chemicals save careers, and sometimes lives.
Nobody wakes up hoping for a lab accident. Learning respect for hazardous chemicals often comes the hard way, but it doesn’t have to. It only takes one bad exposure to trifluoroacetic anhydride to prefer a few extra minutes of prep, checks, and cleanup over a trip to urgent care.