Looking at many of the chemicals sitting on shelves in research labs, few have built up as much reputation as trifluoroacetic acid, or TFA. Its story stretches back nearly a century, when chemists first realized that swapping hydrogen atoms for fluorine in acetic acid packs a surprising punch. Chemists in France and Germany worked through cumbersome, dangerous early syntheses that relied on highly reactive fluoro-compounds. With time, TFA production grew safer and cleaner, catching attention in both academic and industrial settings. The reasons for this popularity go well beyond its intimidating acidity. TFA keeps showing up in organic synthesis, peptide chemistry, analytical tools, and even pharmaceuticals. For a compound discovered in the shadow of wartime chemical experiments and early fluorine chemistry, it carved out quite an unlikely footprint.
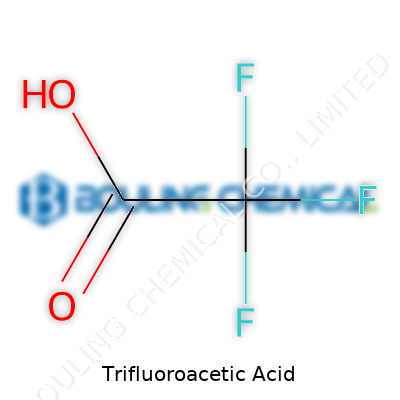
Grab a bottle of TFA, and you’d notice a pungent smell and a volatility that fills the air quickly if uncapped. Here we’re looking at a simple formula: CF3COOH. The three fluorine atoms at the tip of the molecule ramp up the electronegativity, making it far more withdrawing than regular acetic acid. This molecular tweak makes TFA stronger than many common acids. It comes as a clear, colorless liquid at room temperature. Its boiling point sits lower than one might expect for an acid—just under 73°C—a detail that poses both advantages and headaches when working at the bench. Water grabs onto TFA easily, and it dissolves a wide range of organics and inorganics, making it a universal solvent for researchers hunting purity or efficient reactions. In the hands of an experienced chemist, TFA offers an extra gear, and that low pKa turns simple reactions fast and clean or even unlocks impossible synthetic routes.
In research and manufacturing, the technical grade of TFA really matters. Impurities, water content, and residual acids can make or break outcomes, especially with sensitive catalysis or pharmaceutical work. The strongest grades guarantee purity above 99 percent, with water levels kept razor-thin. Working with TFA means watching the container, as its vapor creeps along benchtops and finds the smallest cracks. The labeling must warn about corrosiveness and volatility. Standard amber glass or fluoropolymer bottles keep TFA safe from breaking down. Even experienced workers feel the sting when TFA spills: gloves, goggles, and fume hoods become daily necessities, not just best practices. Anyone who has had TFA catch their skin remembers the pain. Its fumes burn the nose and throat. This isn’t a chemical you ignore in shared spaces.
Producing TFA at scale calls for careful chemistry. The original, dangerous routes used antimony-based fluorination, but now industrial synthesis favors oxidation of 1,1,1-trifluoro-2,2,2-trichloroethane (TCTE, more often called chlorofluorocarbons) with a catalyst. Another common approach starts from fluoroform, a byproduct of Teflon manufacturing, oxidized using relatively benign agents. These industrial routes reflect a broader shift toward safer, greener chemical practices. Even so, every pathway has environmental risk: separating TFA from unwanted byproducts demands serious handling. Reliable, high-purity supply lines directly affect success in research labs and drug factories. If a shipment runs late or doesn’t pass quality control checks, entire experiments stall. Recent years have seen supply squeeze due to regulatory changes and environmental restrictions, making chemists acutely aware that TFA is not just another lab stock.
TFA’s reputation in chemistry comes in part from its ability to cut through complexity. It can cleave stubborn protecting groups in peptide and organic chemistry, enabling rapid deprotection without attacking other sensitive parts of a molecule. That selective power saves time and money in long synthesis chains. For analytical chemists, TFA enhances the signal in liquid chromatography, pushing peptides and proteins through columns with better separation. Its volatility means that after a reaction, TFA can be removed easily, leaving behind pure product. Still, TFA’s strength can be a double-edged sword. Some reactions run too fast, or leave behind unwanted side-products if not carefully controlled. TFA is known to react violently with strong bases and fine metal powders—reactions I’ve seen surprise even experienced professionals.
Out in the chemical world, TFA answers to many names including trifluoroacetic acid, trifluorethanoic acid, and by the shorthand CF3COOH. Some older texts refer to fluoroacetic acid or even “perfluoroacetic acid,” but those can cause confusion. Standardizing nomenclature helps researchers find relevant information without missteps. Accurate labeling, both for storage and regulatory reasons, isn’t some trivial box to check. Fields such as pharmaceuticals and environmental science can hinge on naming: one mislabel led a colleague to nearly dose a sample with tenfold the acid prescribed. Consistency keeps dangerous slip-ups at bay in international, multidisciplinary projects.
Everyday lab work with TFA shapes an appreciation for preparedness. Despite its clear appearance, it eats through skin and metal like many strong mineral acids. Spills pit stainless steel and dissolve rubber, corroding equipment over time. Only carefully chosen materials—Teflon, borosilicate glass, and some high-grade plastics—hold up. Fume hoods stand as mandatory, not optional gear. The acid’s vapor creeps, and even a small spill needs immediate neutralization with sodium bicarbonate, ideally followed by professional disposal. Mistakes create not only risk of burns, but insidious long-term exposure. Regulatory guidance now limits use and storage quantities, keeping both professionals and the environment safer. In my experience, no one forgets safety drills with this acid—its sharp, lingering odor leaves a powerful reminder.
Few chemicals roam as far across industrial and academic territory as TFA. Peptide synthesis remains the signature application, as the acid strips away protecting groups and opens short, efficient synthetic pathways. Look further and TFA shows up as a modifier in HPLC (high-performance liquid chromatography), and as a strong solvent for specialty reactions. Solubility with both organic and inorganic compounds lets pharmaceutical chemists optimize drug molecules for better yield and purity. Manufacturers of agricultural products and fine chemicals also depend on TFA as a catalyst or building block. Even in electronics, TFA assists in producing specialty materials that power everything from smartphones to solar panels. This broad spectrum pushes researchers to continually optimize use—balancing cost, purity, regulatory restrictions, and safety considerations.
R&D labs worldwide find themselves both dependent on, and challenged by, TFA’s properties. This chemical’s strength and volatility spark ongoing research into alternatives with milder side effects but similar capability. Academic chemists dig into greener approaches, aiming to recycle TFA after use, or synthesize it with less toxic waste. Analytical scientists developing new mass spectrometry methods rely on TFA, but also try to find less corrosive substitutes to prolong the life of sensitive instruments. Peptide drug developers experiment with modified deprotection strategies, seeking ways to use lower concentrations—or to switch acids entirely—without leaking performance. In daily practice, I’ve watched teams rework classic progressions just because of fluctuations in TFA availability or cost. These stories highlight a broader challenge: progress in chemistry rarely comes from breakthroughs alone, but from thousands of tweaks, optimizations, and adaptations in response to the sometimes stubborn nature of chemicals like TFA.
Despite its utility, TFA’s toxicological profile draws concern. Studies trace pathways showing how TFA, persistent in the environment, can contaminate soil and groundwater through improper disposal or accidental release. Certain plants and aquatic life respond poorly even to low concentrations, with research underlining bioaccumulation worries. In the human body, TFA’s robust acidity causes tissue damage upon contact, with inhalation leading to airway and lung irritation. Chronic exposure remains a challenge, as even trace amounts in pharmaceutical or agricultural products could spark regulatory scrutiny. Companies and research institutions alike increasingly look for closed-loop systems or enhanced reclamation protocols to cut down on emissions. My own experience lines up with the research: strict adherence to disposal and storage guidelines is part of building credibility, not just ticking a box for auditors.
Across nearly every sector touching advanced synthesis, TFA remains both a tool and a challenge. As green chemistry comes to the forefront, new rules and corporate commitments push for better management, more effective reclamation, and stronger controls on handling and waste. Researchers explore biodegradable alternatives that keep desirable chemical traits but ease off environmental persistence. Technology developers seek in-line processes that recover and reuse TFA on-site, reducing demand and stemming toxic releases. I’ve seen grassroots collaboration with environmental scientists and waste engineers begin shaping new best practices. TFA’s legacy may shift, not as the go-to strong acid, but as an early symbol of chemistry’s ability to adapt, innovate, and answer global challenges with both rigor and responsibility.
Trifluoroacetic acid, known in labs as TFA, isn’t something most people encounter in daily life. Yet, for anybody who has spent time in a chemistry laboratory, it’s about as familiar as a steaming cup of coffee. TFA steps up as a strong acid in organic chemistry. Its power comes from those three fluorine atoms, which pull electrons away from the rest of the molecule and make it hungry for reactions.
I walked into my first organic chemistry lab and met TFA as a reagent for peptide synthesis. Anybody working with proteins knows assembling a chain of amino acids gets tricky. TFA breaks bonds between a growing peptide and its protecting groups, letting scientists isolate the molecule they want. This might sound specialized, but it supports entire fields—biotech and pharmaceuticals count on reliable reagents to produce life-saving medicines. TFA, though far from the spotlight, helps labs craft medicines that real people need.
Academics get excited explaining chromatography, and for good reason. Instruments like high-performance liquid chromatography (HPLC) need substances that help separate complex chemical mixtures with precision. TFA acts as an ideal additive to the mobile phase, sharpening those blurry peaks scientists see on computer screens. I remember the first time I swapped in trifluoroacetic acid during a complicated protein analysis and watched the results jump in quality. Clean separation isn’t just a lab metric—it means better data, fewer wasted experiments, and faster progress toward answers that can impact disease research.
Take a look at the clothing label that mentions stain resistance. Many garment treatments rely on fluorine chemistry, and TFA often plays a role in both the process and testing. Electronics manufacturers and agricultural researchers use TFA as a building block or testing material too. Every time you pick up a device with specialized coatings or crop treatments that promise efficiency, there’s a real chance the development traced back to reactions involving TFA in some form. I believe sometimes the things we overlook—like these reagents—actually drive progress on big issues, from better food security to safer electrical parts.
None of this undercuts questions about risk. TFA is toxic and persistent in the environment. Researchers have found traces of it in water sources and air, raising red flags for health experts. Fluorochemicals in general take ages to break down, so simple carelessness anywhere can mean long-term problems everywhere.
What can be done? For one, scientists and industry leaders can keep looking for greener alternatives. Efforts are already underway to cut back use of persistent fluorochemicals where possible. Companies storing and transporting TFA need proper training and strict protocols, with government oversight that responds to new science. As for working in a lab, it matters to keep students keenly aware of proper handling, storage, and disposal. I’ve always felt that real progress comes when safety moves from a checklist to a habit.
TFA reminds me that progress in science rides on the shoulders of sometimes-dangerous tools. Its presence in life-saving research, industrial chemistry, and high-stakes diagnosis means it isn’t just a bottle with an intimidating label tucked away on a shelf. As curiosity and need push us forward, staying careful, creative, and transparent about toxic substances lets society benefit from chemistry without inheriting its regrets.
I remember my first stint handling trifluoroacetic acid in a college chemistry lab, long before I took workplace safety seriously. Its vinegary bite cut through the usual scents in the fume hood. Sometimes, substances with the strongest smells warn us best. Trifluoroacetic acid packs a triple punch with its reactivity, its volatility, and the speed it packs a stinging burn.
Anyone working in synthetic chemistry or pharmaceuticals recognizes this acid. Chemists count on it for peptide cleavage, as a solvent, and whenever an ultra-strong acid is a must. But convenience can blind us. Trifluoroacetic acid can scald skin on contact, cause immediate eye damage, and even scar the lungs if the fumes escape the hood. Its corrosive bite isn't just skin-deep; it soaks through nitrile gloves if you don’t check for rips. Few things get my heart pounding faster than seeing careless pipetting or someone cradling an acid bottle with no goggles on.
Let’s get specific. The United States Occupational Safety and Health Administration warns about its risks. Inhalation can lead to chest pain, coughing, and even pulmonary edema, which could quickly turn into a life-threatening emergency. Splashing the acid in your eyes doesn’t get fixed with a bit of water—it means a trip to the ER, often followed by lasting vision problems. Even small spills seem trivial until you spot the ice-like burns that betray a few careless drops.
Another problem with this acid is its volatility. Once a bottle opens, it starts to fume. Without a proper fume hood, nobody escapes the choking sensation for long. I once saw a junior researcher unscrew a bottle on an open bench, and in seconds, the sharp odor spread through the room, leaving a cluster of sneezing, watery-eyed coworkers. A quick scan of accident reports in academic labs shows this isn’t a rare event.
Many believe a sturdy pair of gloves and goggles solve the problem. Personal protective equipment should always be checkmarked, but culture matters more. In my experience, the safest labs enforce strict training, not only rules taped to cabinets. Every time new students walk in, they review the safety sheets, but better labs go beyond—walking through best practices and asking “what if?” Critics might call this overkill, but seeing someone’s panic after a chemical splash brings home the lesson that procedures must come alive every working day.
Up-to-date hoods, dermal barrier gloves, face shields, emergency eyewash stations—these tools matter, but only if people respect the risk. A locked storage cabinet keeps bottles away from untrained hands. Alerts next to the acid remind you to double-check everything. Disposal presents its own headache, since trifluoroacetic acid doesn’t break down easily and poses an environmental hazard. Professional hazmat disposal is the only real route, which means small labs sometimes face costs that tempt shortcuts.
Experience teaches that trifluoroacetic acid gets dangerous the moment it’s underestimated. Real safety is built on routine, on asking for help before tackling new protocols, on calling out unsafe behaviors without hesitation. Trust among coworkers keeps accidents at bay more than any brand of glove ever could. The real hazard lies in letting familiarity breed carelessness. In the rush of lab work, it’s easy to forget that this acid is always waiting for a mistake. Give it the respect it demands, and most dangers stay in check.
Trifluoroacetic acid doesn’t ask for much—just a little respect and diligence. I spent plenty of days in the lab, and I remember the unmistakable sharpness that stings your nose as soon as you crack open a bottle. It isn’t a substance to take lightly. One drop where it shouldn’t be, and you realize just how corrosive this acid can be. Not just to skin and eyes, but to metals and other basic materials in your workspace. It etches reminders into surfaces and equipment that outlast your memory of the day.
Glass and high-grade PTFE bottles put up the best fight against this acid. Anything less melts, cracks, or slowly crumbles over time. Someone once tried storing it in a cheap plastic bottle, thinking it’d be “just overnight.” The bottle softened and leaked onto the benchtop by morning. Corrosion marks stayed for years. So, lab-quality glass or specific fluoropolymer containers do the job right. Resealable lids with secure threads stop fumes from sneaking out—fumes that corrode metal shelving or cause headaches when ventilation slips.
Leaving trifluoroacetic acid near heat sources or in direct sunlight asks for trouble. The stuff boils at a surprisingly low temperature, and vapors can fill a small room quicker than you expect. I learned early: always pick a cool, dry chemical cabinet, well below eye level. Acids don’t belong on the top shelf. It’s easy to underestimate how one accidental bump could bring a bottle down. Every drop released can spell disaster for floors, gear, and anyone standing nearby.
It’s tempting to store similar-looking bottles together for convenience, but that’s a rookie mistake. Acids next to bases set up a potential chemical war. One faulty seal or splash, and you’ll witness violent reactions, sometimes with choking fumes. Only acids—especially strong non-oxidizing ones—sit near trifluoroacetic acid, and each container gets an acid-only label. Pay attention to chemical compatibility charts; it’s not overkill. I’ve seen benches evacuated for mixing up cap colors and contents.
Gloves, goggles, and a face shield aren’t just for pouring. Even uncapping or moving the bottle sends vapors wafting. I remember once thinking I could handle a split-second transfer barehanded. You never see it coming until a tingle turns into a burn. By then, you’re washing under cold water and writing incident reports the rest of the afternoon.
Spills happen, even to the careful folks. I keep calcium carbonate or sodium bicarbonate powder nearby for quick neutralization. Spill kits with absorbents rated for high-strength acids live in every corner of the lab. Ventilation isn’t just for comfort, it pulls dangerous vapors away from your face and out of breathing range. Emergency showers and eyewash stations still get checked every month. No one ever thinks they’ll need those, until they do.
Trifluoroacetic acid works for powerful chemical transformations, and it rewards serious attention to safe handling. A little upfront caution sets the tone for smoother, safer research—and fewer costly mistakes. This isn’t about paranoia, just the reality that safe storage and respect for chemicals protect both people and progress.
Science gets its fair share of intimidating names. Trifluoroacetic acid isn’t exactly what you’d call user-friendly at first glance. Still, its chemical formula, CF3COOH, represents something serious and practical in the world of research, development, and industrial progress. Some may brush past details like this, yet the right combination of atoms shapes how safe, efficient, and transformative discoveries become.
During my years working with molecular biology, chemicals like trifluoroacetic acid are far more than dry textbook entries. Every bottle labeled CF3COOH brings a level of expectation, caution, and trust. Professionals count on it in protein purification, high-performance liquid chromatography, and synthetic chemistry. For projects hinging on tiny margins, something as minute as a wrong formula turns into lost time, wasted samples, and sometimes equipment damage.
Accuracy in names and formulas seems pretty basic, but so does looking both ways at a crosswalk. Miss either step, and consequences stack up fast. The trifluoromethyl group (CF3) sitting next to a carboxylic acid group (COOH) creates a molecule more acidic than plain acetic acid. This unique punch allows it to drive certain chemical reactions forward when less potent acids just don’t cut it.
Mistakes don’t tend to stay small when it comes to strong acids. CF3COOH holds enough corrosive power that goggles and gloves are part of the daily routine. This isn’t about ticking boxes; it’s about protecting vision, skin, and sometimes entire labs. I’ve seen what happens when newcomers brush aside proper identification or skip double-checks before adding something to a reaction—cloudy beakers, ruined experiments, and sometimes hurried calls for help. Keeping clear on the formula is step one.
Mislabeling, especially in shared facilities, infects the trust between scientific teams. One misplaced bottle marked with a slightly wrong formula compels everyone to pause and play detective. This opens space for stress, unnecessary cleaning, and time spent decoding what should have been straightforward. Everyone’s job gets easier with a mutual commitment to accuracy.
No lab is infallible. But that’s why rigor in recording and cross-checking details like CF3COOH anchors good science. Academic journals highlight cases every year where researchers face setbacks due to oversight or complacency—missed decimal points, misspelled formulas, and confusing abbreviations. These little errors snowball, sometimes tarnishing reputations or derailing grant applications.
With the world increasing scrutiny on research reproducibility and scientific credibility, accuracy is non-negotiable. Every team relies on a foundation of verified knowledge, whether they’re in pharmaceuticals, environmental monitoring, or education. Mistakes don’t care about intentions—outcomes count far more.
A little old-fashioned diligence pays off. Double-checking chemical formulas like CF3COOH may not inspire cinematic drama, but those small, conscientious habits ripple outwards. They transform the culture of science toward respect, shared responsibility, and ultimately, better discoveries. A lab that gets the basics right feels safer, communicates better, and adds value for everyone touched by its work.
Trifluoroacetic acid, often just called TFA, has a reputation among chemists. Anyone who has handled it remembers the biting, sharp smell and the way TFA eats through gloves if you are not careful. More than a few folks have stories about pinhole burns or a ruined pair of pants after a spill. TFA isn’t just fussy; it’s highly corrosive and volatile, and that means disposal isn’t just tossing it down the sink. It can end up in groundwater, damage pipework, or injure someone further down the line if not treated with respect.
In plenty of labs, good chemical hygiene is a matter of habit. With TFA, the stakes get higher since it has toxic fumes and can linger in the environment. When poured down drains, TFA turns into trifluoroacetate, a persistent pollutant. While not as headline-grabbing as some, it raises issues for both people and ecosystems. Research connects it to aquatic toxicity and warns that it accumulates over time. The global science community has an obligation to address such substances carefully, based on evidence and experience.
The usual approach to lab waste often starts with neutralization—think of diluting acids and bases until they’re safe enough to enter the standard waste stream. This method doesn’t cut it with TFA. Adding a base, such as sodium bicarbonate, doesn’t magically make it non-toxic. The resulting salts hang around, and most wastewater plants aren’t built to handle them effectively.
True safe disposal starts with collecting TFA wastes in dedicated, airtight containers—clearly labeled, with hazard stickers. Storing those bottles on designated shelves is more than just bureaucracy; it cuts the risk of accidental mixing or hidden leaks. Even a seasoned lab worker relies on double-checking labels and keeping incompatible substances apart. Peer reminders and double-signoffs help, too.
Rules aren’t just red tape: the EPA and similar agencies in other countries classify waste like TFA under hazardous waste regulations for good reason. They expect professionals to hand it over to licensed chemical waste companies. These companies use incineration at high temperatures or advanced treatments instead of ad hoc dumping. Trying to bend the rules risks environmental fines, legal trouble, and more importantly, harm to communities.
Disposing of trifluoroacetic acid the right way starts before the bottle is opened. My own mentors instilled the habit of planning for waste not just as an afterthought, but during every experimental design. Purchasing only what gets used, substituting less hazardous chemicals whenever possible, and sharing surplus between labs all help cut down volumes for disposal. Those small steps, taken seriously, add up for cleaner science.
TFA demands this respect not because it is novel, but precisely because so much modern chemistry relies on it. Keeping people safe and waters clean requires stubborn diligence, honest record-keeping, and viewing every bottle of waste as a shared responsibility. That attitude matters more than any single set of instructions or guidelines.