Sulfuryl chloride has roots tracing back to an era when chemists chased new compounds with a kind of wild curiosity, long before today’s organized industry. It sprang into wider use after World War I, with growing demand for chlorinating agents and intermediates that could keep pace with the rapidly shifting world of dyes, pesticides, and pharmaceuticals. Its aggressive chlorine content meant labs and factories could push the boundaries of organic chemistry, producing molecules that advanced everything from basic research to large-scale applications. My first encounter with sulfuryl chloride happened in a university setting, where our lab’s fume hood became the stage for generations of young scientists marking their first steps in synthesis. It’s not flashy, but its historical shadow looms large where chemical innovation finds a home.
This compound boils at temperatures not too far above the warmth of a kitchen oven and pours with a sharp bite that stings the nose. Chemists know it by its formula SO2Cl2, staring out from bottles labeled with hazard warnings in block red letters. Anyone who’s handled it recognizes a clear, fuming liquid that shouldn’t linger in sunlight or be left uncapped for long. Its direct and pungent qualities speak to its reactive power, helping drive chlorination and sulfonation projects in both academic and industrial settings. Those who’ve worked with it remember how quickly it can react or how its vapors demand respect from even seasoned researchers.
As a liquid with a density slightly greater than water, sulfuryl chloride finds itself tricky to clean up if spilled. It evaporates quickly, filling enclosed spaces with fumes that irritate eyes and lungs in seconds. The bond between sulfur and chlorine, strengthened by accompanying oxygen atoms, brings an energy that wants to transfer those chlorines somewhere else given the right trigger: often light, heat, or the sheer presence of an organic molecule. Most can agree that the substance corners the market on being both useful and hazardous. Its decomposition liberates not just chlorine gas, but also sulfur dioxide—each a problem for lungs, equipment, and the environment.
Bottles arrive bearing warning symbols and UN numbers recognized across industries. Chemists prioritize clear labeling: “Corrosive,” “Oxidizer,” “Respiratory Hazard.” Each shipment comes with data sheets describing limits for exposure, corrosion risk, and storage temperature. This is more than bureaucracy; real tragedies unfolded in old labs when those precautions didn’t exist. Anyone trained to work with sulfuryl chloride knows to check the labels twice and never ignore an unfamiliar smell where this chemical gets opened.
Traditional synthesis walks a fine line between efficiency and danger. Passing dry chlorine through sulfur dioxide at controlled temperatures, with the help of a catalyst, produces a gas mixture that condenses into the clear liquid found in supply rooms. Over time, engineers buckled down on greater containment, aiming to squeeze every bit of product from each run without bleeding out to the surrounding air. Workers in these plants understand the risks, and many old-timers can recall anecdotes of procedures updated after lessons learned the hard way.
Sulfuryl chloride kicks off reactions with alkenes and aromatic compounds, making it a reliable chlorinating agent where other chemicals fall short. It cleaves C-H bonds in a flash, transforming otherwise stubborn structures into building blocks for dyes, drugs, and agricultural chemicals. Organic chemists count on it for its willingness to hand over both chlorine and sulfonyl groups, broadening what gets tested in fields from polymer science to pharmaceuticals. At the bench, it rarely disappoints in efficiency—if the user gives it the careful attention it demands.
This substance reaches the market under names like “sulphuryl chloride,” “SO2Cl2,” and “sulfuric dichloride.” Translation can sometimes muddle the waters, but the molecular formula stays the same. In meeting rooms, safety seminars, and conference posters, fellow chemists instantly recognize its character regardless of language or brand. For those focused on research, commercial catalogs group it among reactive intermediates, distinguished more by handling protocol than marketing flourish.
Discussions about sulfuryl chloride in the lab almost always start with safety. Protective gloves, eye shields, and proper ventilation aren’t luxuries—they’re non-negotiable parts of the workday. Accidents involving sulfuryl chloride make headlines, prompting investigations and changes in standard operating procedures. It pays to remember that a fume hood or proper mask can make a vital difference. Teams working with this chemical rely on rigorous training and regular drills, especially in larger plants where a small leak can churn out clouds of corrosive gas. Over the years, countless improvements led to a culture where preparation and response get equal billing to technical skill.
Sulfuryl chloride earns its keep in places as varied as pharmaceutical plants, rubber processing facilities, and specialty chemical synthesis labs. Industrial-scale production of pest control agents, dyes, and polymers can hinge on access to a reliable supply. Polyester fibers, flame retardants, and reagents all trace ancestry back to routes running through this compound. In academic research, it helps probe reaction mechanisms and develop proof-of-concept molecules. My own experience tells me that for all its danger, life in a chemical lab would look very different without it. Its reach spans far past its small volume on the storeroom shelf.
Innovation has a way of bending even old warhorses like sulfuryl chloride toward new uses. Green chemistry initiatives encourage researchers to look for alternatives, reduce waste, and engineer closed processes that capture toxic byproducts. Some research tackles milder and more selective chlorination, hoping to bring the same results with less collateral damage to people or the planet. There’s a parallel track focused on better sensors and containment—technologists never quite finish the job of making its use safer. In conferences, veterans trade stories about close calls and breakthroughs, showing how collective memory guides future work just as much as fresh data.
The health hazards are real and thoroughly documented. Sulfuryl chloride prompts both immediate and delayed responses: respiratory distress, burns, and lasting lung injuries from brief contact. Animal studies chart its toxicity in fine-grained detail, supporting regulatory restrictions and first aid protocols across the globe. Workers’ experiences and epidemiological surveys drive the call for more effective safeguards, especially where chronic exposure can chip away at quality of life over time. Nobody who’s lived through a spill or near-miss shrugs off these risks as abstract; they inform a cautious respect that future generations will hopefully inherit.
People expect tomorrow’s chemical industry to balance efficiency, profit, and responsibility better than ever before. Sulfuryl chloride brings plenty to the table if properly managed, but its reputation as a hazardous yet essential tool pushes the industry to rethink old standards. Innovations in automation, safer analogs, and rigorous training aim to minimize harm without giving up the results this chemical delivers. The demand for sustainable methods and greener alternatives grows, yet for now, sulfuryl chloride keeps its foothold in key reactions. The story moving forward looks like a dialogue between experience and progress, as researchers blend respect for tradition with a hunger to solve problems more cleanly and safely.
Sulfuryl chloride doesn’t greet you on a shelf at your local hardware store, yet it works hard in places few people look. I’ve watched this chemical turn up in the stories of pesticide production, the world of pharmaceuticals, and the effort to keep water clean. The name might not sound familiar, but its influence spreads far and wide.
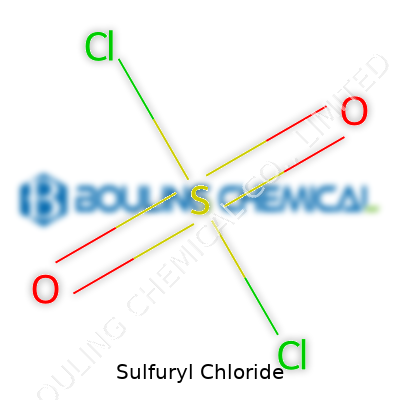
Anyone walking through a chemical manufacturing plant would spot sulfuryl chloride playing several roles. This compound, a colorless liquid at room temperature, shows up where there’s a need for a strong chlorinating and sulfonating agent. In plain language, it’s a heavy hitter for adding chlorine or a sulfonyl group to other chemicals, a step that opens doors for endless other ingredients.
Pesticides don’t just drop from the sky. They’re built piece by piece, and sulfuryl chloride usually gives them the chlorine atoms that make them work as intended. The same goes for some pharmaceuticals. Drugs combating infection and inflammation can owe part of their punch to chlorine atoms placed by reactions involving sulfuryl chloride. In my time learning from industrial chemists, I saw this chemical regarded the way a solid wrench is in an auto shop: reach for it when you really want a job done.
Water treatment plants sometimes use sulfuryl chloride for disinfection or to aid in the production of chemicals that purify water. Given the growing problem of contaminants in drinking water, a reliable cleaning tool counts for a lot. People trust the water coming from their taps. If the right tools stop bacteria and viruses in their tracks, every community benefits.
Sulfuryl chloride doesn’t exist without problems. Its reaction with water leads to the production of hydrochloric acid and sulfuric acid, both of which cause harm if handled carelessly. I’ve seen chemical workers suit up head-to-toe before dealing with it. Health agencies keep a close watch, and with good reason. Leaks or spills cause respiratory issues, skin burns, and environmental harm.
Regulators in the United States, Europe, and Asia have laid out strict guidelines for transport and use. Keeping this chemical under control isn’t just a matter of legal compliance. It’s about making sure people in the surrounding areas keep their health, and workers on the inside get home in the same shape they started the day.
Some companies have started shifting toward less hazardous chemicals for certain steps, but change isn’t easy or cheap. Science schools and industry partnerships continue to teach safer work practices while looking for new methods that might sidestep the risks sulfuryl chloride brings to the table.
Chemistry keeps evolving. Finding new paths to the same end products—without as much risk—remains a shared goal for companies, regulators, scientists, and the communities living near these plants. As consumers pay more attention to how everyday goods are made, the push for safer, more sustainable methods gains momentum.
Anyone who’s spent time in a chemical lab learns this fast: sulfuryl chloride demands respect. A direct taste of its fury comes at the moment a bottle gets uncapped and sharp fumes waft out. Inhaling that will never be just a “bad smell”—it feels like acid digging into the throat. Forget working with this chemical in T-shirts or that dog-eared lab coat with the fraying sleeves. Safety starts with covering up. A heavy-duty lab coat, chemical-resistant gloves that actually fit, and full-coverage goggles keep risk in check. I’ll never forget seeing a peer suffer a splash burn that ate through cotton gloves as if they weren’t there. Neoprene or butyl rubber gloves are the real shield. Splash-proof goggles alone are not enough—a face shield helps, especially when transferring larger quantities.
A safe lab isn’t about having one fancy fume hood tucked in the corner; it’s about using that hood every single time something volatile comes out, no matter how busy things feel. Sulfuryl chloride fumes will corrode metal in no time and rough up lungs even faster. The investment in local ventilation pays back every time a flask comes out undamaged and nobody coughs in the room. Proper airflow makes worries about uncontrolled vapor release disappear. Strong noses can sense leaks, but good airflow means you never have to rely on human error or luck.
One careless spill on a bench has a way of turning a calm day upside down. Water reacts with sulfuryl chloride faster than you’d expect, kicking out choking hydrogen chloride gas. I’ve seen people underestimate what a single drop can ruin. Dry tools, well-sealed containers, and tight lids are non-negotiable. Even pipettes and syringes must be checked—water inside means you’re playing with disaster. Storage makes a difference, too. No one should store sulfuryl chloride near anything damp or acidic.
Every university orientation on chemical safety gets ignored by some. Experience rewrites those habits. You might think accidents only happen to people who rush, but even slow, practiced hands can make mistakes. Spill kits, neutralizing agents, and eyewash stations need to be more than decoration. Check them often, show new people how to use them, run drills. Watching someone freeze after an accident because they never practiced with the eyewash or spill kit makes clear where priorities belong. Emergency routines stick with you only if you walk through them under real pressure, so drills matter.
Lab teamwork can be a lifeline or a liability. Leaving sulfuryl chloride out for the next shift or labeling containers half-heartedly leaves others open to trouble. Many a chemist, myself included, has returned to a fridge only to find a bottle sweating after condensation built up inside—another accident in waiting. Clear, bold labels and reliable communication between coworkers are habits that prevent those mistakes.
In the rush to mix, synthesize, or run an assay, the drive to skip a few steps can feel tempting. But sulfuryl chloride, with its pungent fumes and unpredictable reactions, enforces a strict order. Better gear, reliable airflow, and solid routines don’t just protect property—they keep the people in the lab safe enough to do their jobs again tomorrow. If only every lesson didn’t have to stem from a close call, the world would see fewer scars, fewer shutdowns, and a lot less fear.
Sulfuryl chloride isn’t something you keep next to window cleaner or motor oil. I’ve seen its power firsthand in lab settings—a bottle with a bite. Even among experienced professionals, mishandling this liquid sets off more than just alarms. It’s not about following some bureaucratic line in an obscure manual. This is about real danger: violent reactions with water, sharp fumes that don’t care about job titles, and the simple, persistent risk of heat causing pressure to build until things go badly. Mistakes hurt people, damage property, and set off headaches you never want to explain to a fire marshal.
This chemical doesn’t forgive shortcuts. It reacts fast with moisture, and those reactions come with toxic hydrochloric acid vapors. Humidity isn’t some distant threat, it’s right there in the air, and even a microscopic leak can let water in to start something nasty. Experience in storage environments taught me the quickest lesson—one missed detail leads to an expensive clean-up and a panic you won’t forget.
Sulfuryl chloride prefers temperatures closer to basement than boiler room. Heat can make the liquid decompose, cranking up both pressure and risk. That’s why storage in a separate, climate-controlled cabinet or dedicated shed, far from sunlight or hot machinery, pays for itself long before an accident does. I’ve lost count of the times I’ve seen casual setup in a shared chemical store eventually lead to problematic leaks or corrosion on valves. A sealed steel drum with tight, checked closures blocks out water and keeps sunlight off the label.
Some folks think big drums mean getting a better deal. Experience says differently: more volume in storage just means more liability. A spill of a liter is already a huge headache, but a drum can put an entire facility at risk. Storing only what’s needed, in designated areas with clear labels, limits the fallout from any mistake. Invest in proper containment trays and ventilation, and never store sulfuryl chloride near anything combustible or reactive. Mixing this with ammonia, for instance, can create a real crisis.
It’s never enough to give folks a page of safety rules. On-the-ground training changes things. I’ve watched new technicians freeze up when an alarm goes off, even if they passed the quiz. Practice with real equipment—gloves, eye protection, chemical-rated respirators—makes the difference between control and chaos. Regular drilling with spill kits and emergency shutdown drills builds skills nobody wants to use but everyone should have.
Sulfuryl chloride tests every lapse in discipline. Every shortcut or lazy day increases risk. Even tiny spills need quick, experienced attention and plenty of ventilation. It’s not an everyday chemical, and storing it as if it were is the easiest way to discover just how dangerous negligence can get. The battle is ongoing: monitor stocks, check containers for rust or pressure bulges, and keep up with the latest regulatory advice. Never treat sulfuryl chloride as a minor footnote in the chemicals cabinet.
Sulfuryl chloride, with the formula SO2Cl2, rarely comes up in everyday conversation, yet its impact reaches much further than most people realize. In my days sorting through glassware and odd-smelling bottles in college chemistry lab, sulfuryl chloride stood out both for its piercing odor and the stern safety warnings written across its label. This clear liquid, heavier than water and quick to fume in the air, commands respect not just for its reactivity but also because it stands as a key player in shaping industrial processes that most people take for granted.
Sulfuryl chloride grabs your attention as an oily, colorless liquid. It boils at about 69°C and gives off fumes almost as soon as you open the bottle. Spilling even a drop skews the air with a choking, sharp smell that leaves no doubt about the need for a fume hood. The liquid packs a punch—chlorine atoms tightly bound to a sulfur dioxide core. You feel the heaviness of the vapors, enough to see them hugging the floor and seeking out every low spot. This density means any accidental release lingers far longer around our feet and equipment, not drifting away lightly as with some vapors.
Ask anyone with experience in the lab, and they’ll tell you: sulfuryl chloride shows up every time you need to add chlorine to something without using pure chlorine gas. The classic use? Making pesticides, pharmaceuticals, and specialty chemicals. Companies reach for it when other methods pose higher risks or raise costs. In one memorable case at my university, we used sulfuryl chloride to make a disinfectant precursor—quick, reliable, and less fussy than dragging out cylinders of chlorine gas. Its efficiency earned a place on the industrial shelf.
There’s no tiptoeing around the hazards. Even a brief whiff of sulfuryl chloride sets off coughing; touch means burns and blisters. Combine moisture and this liquid, and you get a burst of hydrochloric and sulfuric acids—not something you want near bare skin. As chemists and plant workers see it, sulfuryl chloride demands tight control: thick gloves, steady ventilation, and eyes always on the job. Over the years, cases of lung damage hit the news when facilities let safety practices slide. Accidents rarely stay small with a chemical so volatile.
The best answer to the dangers of sulfuryl chloride involves preparation, vigilance, and culture. Training workers matters more than a padded safety manual sitting unread in a drawer. Labs and factories do better keeping the substance in sealed systems, drawing on it only when every piece of protective equipment is in place. Industry could invest in better monitoring for leaks, sensor alarms, and emergency air extractors. Exploring greener alternatives and reducing sulfuryl chloride use when safer reagents can take over may help in the long run, especially as stricter regulations take shape worldwide.
Most folks don’t think about sulfuryl chloride unless they work in a lab or chemical plant. I remember my first chemistry lab internship—the sight of those bright red drums and the odor that stung my nose left a vivid imprint. There’s a reason for those memorable storage methods. Sulfuryl chloride isn’t the sort of stuff you’d want spilling or leaking. Once exposed to water or humid air, it starts throwing off hydrogen chloride, which turns into hydrochloric acid. Nobody wants a surprise acid cloud in a loading dock.
Picture a shipment of this chlorinating agent—big steel drums, tightly sealed, sometimes pressurized steel cylinders for larger loads. These are not just for show. After years working in chemical manufacturing, it becomes clear: minimizing leaks isn’t all about regulations; it’s about protecting workers, the public, and the environment. A drum with a robust seal keeps the compound where it belongs and away from people and groundwater.
Getting sulfuryl chloride from a factory to a customer isn’t as simple as loading it on a truck. Transportation laws are strict. Rail cars, tank trucks, and even shipping containers need to bear warning labels because unexpected exposure can be deadly. Drivers and logistics workers must wear proper gear and stick to routes meant to minimize risks. I’ve seen more than one shipment delayed until everything checked out—no one cuts corners where dangerous chemicals travel public roads.
History shows what can go wrong when transport steps are skipped. Chemical spills in industrial accidents often trace back to poor packaging or carelessness during handling. Flames, heat, or even a careless bump can turn a leaky drum into a serious problem. This risk fuels a constant push for better containers—double closures, extra-thick steel, tamper-evident seals. It takes a team approach: manufacturers, shippers, and emergency response routines all shape how safely a load arrives.
Even with solid systems, nothing’s perfect. New innovations aim for safer, lighter containers, better alerts for leaks, and smarter logistics that monitor shipments in real-time. There’s talk in the industry about more robust tracking tech and better coordination between companies, local authorities, and hazmat teams. In practice, keeping training up to date often does the most good. Every time workers rehearse emergency drills or update their handling procedures, the community stays safer.
Broadly speaking, careful handling of sulfuryl chloride stands as a model for transporting hazardous stuff safely. It’s not just about following government rules; it’s about respect—respect for the people doing the work, the neighborhoods along the route, and the wider environment we all share. Having seen the fallout from mistakes firsthand, I know: every dollar and minute spent getting this right pays off in prevention, not headlines.