Sodium iron pyrophosphate hasn’t enjoyed the media spotlight, but its story runs deep in the pages of chemistry journals. Back in the early and mid-1900s, interest in iron compounds grew as scientists searched for better ways to address iron deficiency and manage chemical synthesis. The compound’s unique makeup—sodium, iron, and pyrophosphate—seemed promising both in food enrichment and as a material for batteries. Early studies started with simple iron salts, yet the interest shifted as researchers noticed that pyrophosphate ligands could stabilize iron in unusual oxidation states. Scientists, especially those focused on nutrition and metal chemistry, saw a potential bridge between the stability of iron phosphate and the solubility features of sodium salts. By the 1970s, reports popped up on using sodium iron pyrophosphate in food fortification, mostly to tackle iron deficiency without the metallic taste or instability brought by simpler iron salts. Its journey continues today in labs trying to tweak its structure for better uses in medicine, nutrition, and even battery cathodes.
Sodium iron pyrophosphate isn’t a staple in every lab or warehouse; it finds itself pulled into service wherever iron supplementation meets technical requirements. People know it mostly for its role in food science and, more recently, for its electrochemical properties. The compound has a hard-to-replace balance: it resists easy breakdown in moist environments, doesn’t leave a harsh metallic sensation, and fits regulatory requirements for iron addition to foods in many regions. Nutritionists and chemists appreciate that it meets the needs for controlled iron release, whether as a fortifying agent in wheat flour or as a component in capsules. Beyond food, it’s pushed into pilot projects as a cathode material for sodium-ion batteries, where iron’s redox flexibility gets put to use in new energy storage prototypes. Each application calls for subtle changes, but the compound’s resilience and tailorability never slip out of focus.
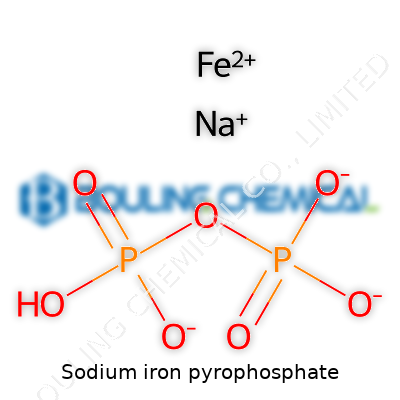
Sodium iron pyrophosphate lands on the bench as an off-white to yellowish powder, depending on its hydration state and method of synthesis. The material holds together well and resists clumping, as long as it stays dry. On a molecular level, the formula usually reads as Na2FeP2O7; the pyrophosphate group bridges iron ions, lending the compound more structural integrity than simple iron phosphates. Water solubility comes slow and partial, which actually helps in food applications since it shields flavor while still letting iron release after digestion starts. The iron remains mostly trivalent, but redox chemistry lets it shift in specific uses. Melting only comes at punishingly high temperatures, a point making it useful for high-temperature applications if folks someday venture there. Its pH profile, insolubility at neutral pH, and predictable reactivity makes scale-up less nerve-racking for manufacturers.
Technical grades of sodium iron pyrophosphate need to hit certain marks: high purity, a well-defined particle size, and low contamination by heavy metals or unreacted phosphates. Food-grade regulations expect iron content to stick within narrow bounds and for the sodium to match up with declared specifications, given the nutritional focus. Labeling has to keep up with government standards, naming the compound in ingredient lists and sometimes detailing the bioavailable iron content rather than just gross iron. Regulatory agencies—like the FDA or EFSA—keep tabs on quality standards, making sure any product under this label meets claims both for identity and nutritional impact. Precision matters, since both under- and over-fortification have consequences that show up in public health reports.
Making sodium iron pyrophosphate usually starts with fairly simple chemicals: sodium salts (like sodium carbonate or sodium pyrophosphate), iron salts (commonly ferric chloride or sulfate), and controlled pH conditions. The trick comes in managing the sequence and speed of mixing, often while heating, to ensure the right form crystallizes. By fine-tuning temperature and reaction time, producers nail down the particle characteristics critical for food fortification or battery applications. Filtration, washing, and drying take up a decent chunk of processing time—removing excess reactants and stabilizing the final powder. The process tends to avoid harsh solvents or extreme pH, a rare advantage in industrial settings where worker safety and cost drive every detail.
Pyrophosphate chemistry gets a lot of mileage in laboratories, thanks to its ability to coordinate metals. Chemically, sodium iron pyrophosphate shows a decent stability profile under mild conditions. Strong acids or bases crack it open, releasing free iron or breaking the pyrophosphate bridges. Under reducing conditions, a creative chemist can tug the iron into a lower oxidation state, potentially shifting how it behaves in biological or electrochemical setups. In materials development, doping the compound or plugging in different alkali metals changes everything from conductivity to bioavailability. As demand shifts, research teams look for ways to nudge the balance towards greater ionic mobility without losing the core structural benefits. The interplay between iron and phosphate brings out a flexibility that simpler salts just can’t match, opening new paths both for fortification chemistry and materials science.
Sodium iron pyrophosphate often shows up labeled as sodium ferric pyrophosphate, ferric sodium pyrophosphate, or sometimes just iron sodium pyrophosphate. In scientific literature, you’ll see abbreviations like NaFePP or similar variants, especially in technical tables. On food packaging, names must match established codes to keep things clear for regulators and consumers. This means you won’t find quirky brand names; the compound keeps its identity straight, a necessary step when nutrition and safety both ride on proper identification.
Handling sodium iron pyrophosphate rarely poses outsized risks, especially compared to plenty of other metal compounds. Standard safety gear—gloves, dust masks, and good ventilation—handles most concerns, since the powder can irritate the respiratory tract with heavy exposure. No one wants iron dust in their lungs, no matter how healthy it might seem in cereal. Food-grade batches keep close tabs on contaminants. Manufacturing rules stress tight quality checks, since trace levels of unreacted iron salts or heavy metals threaten the otherwise strong safety record. Workers in large facilities follow strict guidelines for weighing, transferring, and storing the material. Waste disposal follows the normal flows for iron-containing phosphates, which usually means observing local waterway discharge rules to protect aquifers and streams. The compound stands out for offering low toxicity and few environmental hazards compared to many additives, but that shouldn’t breed carelessness on the floor.
Most sodium iron pyrophosphate that leaves the factory ends up in food and nutrition, fortifying products like flour or infant cereal. The slow release and mild flavor make it a go-to for companies aiming to provide non-heme iron without the taste or digestion problems of other fortifiers. Some teams have pushed it into dietary supplements, where the gentle release pattern helps avoid iron overload. Newer research tracks its utility in sodium-ion batteries, where it serves as a cathode material. Here, the properties that help it in the gut—stability and predictable redox activity—translate to longer battery lifetimes and safer performance. A handful of investigations go even further, exploring therapeutic uses and potential roles in slow-release medicine delivery. Compared with many iron compounds, sodium iron pyrophosphate finds a friendlier reception in both food labs and battery prototypes alike.
Research into sodium iron pyrophosphate blends old-school nutrition science with new pushes in materials chemistry. Food scientists spin out studies on its uptake and bioavailability, comparing it to other iron fortifiers under different dietary patterns. Interest stays high because the need for safe, palatable iron sources never really fades—especially as bread and flour fortification remains a frontline defense against anemia worldwide. On the battery side, lab groups test tweaks to the molecular framework. Changing the phosphate network or balancing different alkali elements gives rise to better-performing cathodes, responding to demand for lower-cost, environmentally safer alternatives to lithium in storage devices. Grants and biotech collaborations push the work into wider, interdisciplinary fields, from pharmaceutical delivery to environmental remediation. Most of these projects blend nutrition, chemistry, and physics, with results carrying practical implications that could reshape commercial usage.
Every chemical in food fortification faces a gauntlet of toxicity checks, and sodium iron pyrophosphate is no exception. Animal studies, cell culture trials, and long-term dietary surveys all point to a solid safety track record. Iron itself raises alarms mainly in cases of overdose, and here the compound’s slow dissolution in the gut works as an advantage—protecting against quick spikes in blood iron that can damage organs. Researchers watch closely for any chronic effects tied to the pyrophosphate group, but human dietary studies show little risk at the doses used for fortification. Even so, agencies demand constant vigilance, since shifting dietary habits or genetic polymorphisms might one day reveal vulnerabilities not seen in earlier research. Ongoing studies keep an eye out for metabolic disturbances that could surface if the compound enters wider use outside the nutrition sector, such as in pharmaceuticals or advanced materials.
The horizon for sodium iron pyrophosphate keeps expanding. With worldwide anemia rates stubbornly high, staple food fortification remains one of the most effective ways to fight iron deficiency—and here, the compound fills a vital niche for populations sensitive to taste and gastrointestinal side effects. At the same time, advances in sodium-ion battery tech shine a spotlight on cheap, abundant cathode materials, fueling another wave of research. For this compound, structural tuning and creative chemistry might deliver new battery lifetimes and storage densities. Environmentalists watch with interest too, as more sustainable, benign additives move center stage in industrial strategy. Pharmaceutical teams consider it a candidate for specialty iron therapies and controlled drug release, pointing to untapped opportunities as chemical modification techniques improve. As these applications mature, the need for ongoing safety evaluations, consumer education, and supply-chain monitoring will only grow. This chemical’s blend of utility, safety, and versatility signals it won’t slip out of the conversation anytime soon.
Sodium iron pyrophosphate shows up quietly in the list of ingredients on breakfast cereals and nutrition bars. Most people skip past it without a thought, yet it plays a bigger role than you’d expect. See, many places in the world still battle iron deficiency. Tiredness, trouble focusing, and stunted childhood growth often trace back to a simple lack of iron. That’s not some distant concern either—I’ve known plenty of folks who couldn’t shake off fatigue until they got their iron sorted.
Adding iron to food isn’t a new idea. The challenge comes from iron’s personality. Regular iron salts, dumped straight into wheat flour or rice, often change the flavor, color, or shelf life. Not everyone enjoys bread that tastes faintly metallic or cereal that looks slightly rusty. That’s where sodium iron pyrophosphate steps in. Unlike other iron additives, it blends in almost unnoticed. The food’s color stays the same. The taste doesn’t give away any hint of metal. Nobody wants to eat an off-tasting staple food, and this little compound keeps that from happening.
Focusing on young children brings this closer to home. Schools in lower-income neighborhoods put a lot of work into making sure students have nutritious meals. For a kid who might only get one nourishing meal a day, hidden helpers like sodium iron pyrophosphate can make all the difference. Studies—some led by the World Health Organization—show that adding this type of iron to basic foods fits right into feeding programs, especially in regions where anemia could derail a child’s ability to learn, grow, or even fight off common bugs.
Fortification in general sometimes gets a bad rap. Some critics worry about possible overdose, changes in food traditions, or the safety of long-term consumption. Most of the evidence doesn’t support big risks at the levels used in public health programs. The flip side looks more serious: iron deficiency lingers as one of the world’s leading causes of lost productivity. Countries with iron fortification in place have seen anemia rates drop, especially among women and kids.
Here’s something I’ve heard from relatives, too: Certain iron pills can upset your stomach. Sodium iron pyrophosphate offers a gentler way. Since the iron is bound in a more stable complex, people report fewer tummy troubles. Supplements shouldn’t have tradeoffs that drive folks away.
What can we do? For a start, spread some truth about what food fortification means and how it works. Support for independent testing and transparency helps ensure that what's in your cereal matches the box. Giving food makers practical, science-backed guidelines keeps everyone’s health on the right track. A world where iron deficiency belongs only in the history books comes from choices that might look small, like including something as low-profile as sodium iron pyrophosphate in the foods we eat every day.
Sodium iron pyrophosphate isn’t a name people often spot in household kitchens. You’ll mostly see it in ingredients labels on boxed cereals or fortified foods. The substance exists because food producers want to sneak more iron into diets without changing taste, color, or shelf life. Iron is a must for our bodies—just ask anyone who’s felt the drag of low energy from anemia. Still, a chemical-sounding ingredient tends to make parents or health-minded folks stop and wonder if that “fortification” does more harm than good.
While sodium iron pyrophosphate sounds industrial, the science behind it points to a mineral salt that combines iron with sodium and phosphorus. Doctors and nutrition researchers often point to iron deficiency as one of the top nutritional problems worldwide, especially among children and pregnant women. More food companies are seeking ways to patch this nutritional gap without causing unwanted taste or digestive issues.
Groups like the European Food Safety Authority and the FDA have run safety checks on sodium iron pyrophosphate. Studies have checked its toxicity and absorbability, and regulators looked at whether it causes allergic reactions. The basic verdict? At approved levels in food, it looks safe for most healthy folks. Researchers do warn that mineral absorption rates run lower for this iron compound than for some others, but the upside is far fewer complaints of metallic aftertaste or darkened foods.
Iron fortification using sodium iron pyrophosphate doesn’t usually tip people over into iron overload. Still, that doesn’t give a free pass for people with conditions such as hemochromatosis or chronic kidney issues. For these people, any added iron can spell trouble. That’s why most public health guidelines recommend talking with a healthcare provider if someone’s managing a condition that affects mineral metabolism.
People tend to pay little attention to what’s in their fortified breakfast cereal beyond sugar and fiber. Having had a long run in the food industry, I’ve seen the push and pull between food science and customer worries play out in real life. When companies want to make a “good source of iron” claim, scientists often suggest sodium iron pyrophosphate since it doesn’t turn foods an off-putting shade or taste like rusty nails. Most customer complaints don’t mention side effects—just occasional curiosity about long chemical names.
Most nutrition experts point out that getting enough iron is far tougher for some groups—like vegetarians, menstruating women, or toddlers on picky diets—so added iron in foods serves an important public role. Of course, no supplement or fortified ingredient replaces healthy eating habits. You want iron from spinach, beans, or lean meats, but the reality of busy life means some families need the boost from fortified foods.
Any time a long, cost-efficient name appears on a food label, skepticism creeps in. It’s smart for consumers to ask questions. Check up on official guidance from groups like the FDA or CDC, as well as what scientific reviews say. Don’t rely only on what shows up in viral posts or clean-eating blogs.
Public health groups need to keep monitoring these additives and back their approvals with regular studies, not just one-time reviews. Industry transparency isn’t just a trend—it’s how trust gets built. When in doubt, mixing fortified foods with whole foods remains a common-sense way to snag the benefits of iron without tipping the balance.
Growing up, I watched relatives deal with fatigue, headaches, and a pale look that hardly ever seemed to fade. Iron deficiency hit their communities hard. Doctors at local clinics usually brought up red meat and iron supplements, but many people couldn’t afford the former or struggled with side effects from the pills. Over time, food technologists began pushing for better food fortification. Sodium iron pyrophosphate entered the scene as a less-talked-about yet quietly effective way to bring iron back into diets.
Not every iron supplement sits well with the stomach. Some folks can’t shake off the nausea or constipation that tags along with the standard iron salts. Here’s the kicker: sodium iron pyrophosphate is less likely to cause these gut problems. It slides into dry foods — flour, rice, cereals — without throwing off flavor or color. That matters, especially for picky kids and anyone who turns down anything that “tastes off.” The simple act of eating breakfast can mean one less trip to the pharmacy.
Anyone who’s followed debates around food fortification knows there’s no silver bullet. But sodium iron pyrophosphate has been used in dozens of countries trying to stamp out anemia in children and pregnant women. UNICEF and the World Health Organization have pointed toward generous evidence: when food carries this compound, iron levels creep back up. It might move slower than iron sulfate, but side effects don’t derail entire policies, and that steadiness counts.
Years of research reveal sodium iron pyrophosphate holds its own. Scientists found it holds up well in hot, humid conditions without breaking down. Housewives in South Asia, where humidity runs high, trust that their rice and flour won’t spoil or look odd after fortification. People get their needed iron and skip the metallic aftertaste—a common killer of iron programs. Not every solution gains ground like this one because too many ideas ignore local traditions and food habits. The strength of sodium iron pyrophosphate sits in its low-key adaptability.
It’s unfair to pin all hopes on one chemical. Experts still debate: should we aim for faster iron absorption or focus on minimizing side effects? For low-income families, reliable access trumps everything. Some NGOs push for even wider use, arguing that every fortified meal chips away at national health burdens. Policymakers face tough choices, but one fact stays clear: children and expectant mothers who eat fortified foods see real improvements in energy and focus.
What cuts through in all these debates is personal experience. A community that able to function, learn, and work without dragging exhaustion around gains a lot more than numbers on a blood test. My own family’s journey drove home how many lives could turn around if more governments and food producers looked past short-term hurdles. Sodium iron pyrophosphate might not catch headlines but quietly, it proves health progress often comes in simple, everyday servings.
Walk down any grocery aisle, and you’re face-to-face with a quiet revolution. Bread, breakfast cereal, infant formula—all getting pumped up with nutrients you won't spot by taste or sight. Sodium iron pyrophosphate, a mouthful of a name for a salt that helps fight iron deficiency, keeps sneaking into foods that tend to show up on just about every breakfast table. The story of how it lands in your morning toast isn’t just about chemistry or factory lines—it’s about public health, everyday habits, and food science coming together.
Iron keeps people going, but plenty folks don’t get enough in their diets, especially where meat or leafy greens cost more or aren’t always available. Food makers started thinking about sneaking a little extra iron where it matters. The classic route used ferrous sulfate or ferrous fumarate. They do the job but can make bread look funny, turn cereal gummy, or leave a metallic tang that makes kids push plates away.
This is where sodium iron pyrophosphate steps in. It brings iron, but leaves color and flavor mostly intact. Food companies pick it for foods sensitive to taste or color changes—think rice, noodles, or milk powder. It also skips stomach upset, something you notice if you’ve ever taken a straight iron pill with breakfast and felt queasy.
Adding sodium iron pyrophosphate to food starts at the mill or processing plant. For flour, it can join the flour stream before packaging, getting sifted and tumbled for even distribution. Cereal makers add it during mixing, before extrusion and toasting. Ready-to-eat baby foods or powdered milks see it blended with vitamin mixes. Not all iron salts behave the same way, but sodium iron pyrophosphate’s main trick is playing nice with everything else—no funky aftertaste, no weird colors, pretty stable shelf life.
From what I’ve heard talking with nutritionists and food tech folks, keeping the iron bioavailable—meaning your gut can use it—is the real balancing act. This salt isn’t the speediest to absorb, but it works reliably and doesn’t stir up side effects. Some countries set regulations around how much lands in food, so manufacturers run checks to keep iron content right in the tight window set by health agencies.
There’s no one-size-fits-all cure for global iron deficiency, but fortifying common foods gives a real boost, especially in places where eating habits don’t bring much natural iron to the table. Schools and hospitals usually line up behind these efforts, because fixing iron levels can sharpen thinking and boost energy for both kids and adults. Still, there’s always a need for good science: tracking whether absorption matches up for different diets or age groups, checking if taste or texture changes quietly sneak by and turn folks off.
If more people understood how fortification really works—the behind-the-scenes way nutrients like sodium iron pyrophosphate slip into everyday foods—they might worry less about “additives” and care more about beating hidden hunger. I’ve spent time volunteering at food banks, where folks rely on staple goods. Big improvements sometimes come from these small chemical tweaks, especially when it means one less cause for fatigue or anemia. So while sodium iron pyrophosphate might not make a splashy headline, it’s making a quiet difference meal by meal.
Walking through a grocery store these days, food labels shout about added vitamins and minerals. A few decades ago, iron deficiency was a big problem. Nowadays, you’ll find iron in breakfast cereals, bread, meal powders, and even snack bars. Food makers have tried all sorts of additives. One of these is sodium iron pyrophosphate, a chemical form of iron that sneaks into fortified food without leaving a nasty taste or smell. The thing is, most people have no idea what this ingredient does once you eat it.
Iron matters, plain and simple. It keeps blood healthy and staves off tiredness. Certain groups—pregnant women, small kids, and some vegetarians—sometimes get less than they need. The World Health Organization pushed for iron fortification in foods in the hope of cutting down anemia cases. Sodium iron pyrophosphate seems handy because it delivers iron without making bread or cereal look gray or taste metallic. The promise is a less-offensive iron, easy to sneak into food.
Additives always worry some folks. Research on sodium iron pyrophosphate has shown it’s tolerated well in small doses, especially in studies looking at children and young women battling iron deficiency. Common iron supplements like ferrous sulfate sometimes upset stomachs. By comparison, sodium iron pyrophosphate appears a bit gentler on digestion. This helps people stick with supplementation, so the iron can actually do its job.
Still, too much iron—from any source—will cause trouble. Iron overload isn’t common just from food unless you already have a rare genetic issue with iron metabolism. For the general population, the bigger problem comes if supplement use doubles up with fortified foods and multivitamins, leading to excess iron. Symptoms usually show up as stomach pain, constipation, or nausea. Some folks might notice darker stools. These reactions mostly crop up with higher doses than you see in ordinary foods.
I’ve met parents worried about chemicals in snacks. That concern about sodium iron pyrophosphate often boils down to the word “pyrophosphate”—it sounds like science class, not something from grandma’s kitchen. But every compound used to enrich food needs approval by groups like the Food and Drug Administration, and they look closely at long-term exposure. So far, animal studies and fortification trials haven’t turned up signs of major health risks at normal levels.
One real debate: people with chronic kidney disease must watch their intake of certain minerals, including sodium and phosphate. Extra phosphate, over the years, can build up in bodies that can’t filter it well. People in this group already get advice from doctors to avoid added phosphate in processed foods, whether it comes attached to iron or not.
Most folks will see no side effects from eating foods with sodium iron pyrophosphate, especially at the levels allowed in most countries. Still, everyone benefits from reading nutrition labels and not piling on iron pills without a reason. Kids and adults with health conditions, or folks using iron pills, should talk to a doctor before making big diet changes.
Iron fortification played a big part in reducing anemia worldwide, but nothing beats a varied diet. Those with lingering worries about food additives should trust their gut and choose unprocessed options when possible. It’s not about panicking over every unfamiliar word on a label, but about staying informed and making choices that fit your own health and lifestyle.