Sodium bifluoride has carved out a quiet niche for itself in chemistry and industry, but the journey started long before its modern usage. Chemists in the early 1800s, eager to experiment with the new world of fluorine-based compounds, stumbled across sodium bifluoride in their attempts to isolate and purify hydrofluoric acid. Its unique properties didn’t get lost in the shuffle—a mix of curiosity and trial pushed the compound into laboratory routines. Later, as industrial-scale chemistry took off, sodium bifluoride left the lab bench and stepped squarely into the worlds of glass etching and metal treatment. It’s a pretty classic story of how a compound moves from obscure curiosity to industrial workhorse—there’s never one sudden moment, just slow progress fueled by curiosity, new needs, and repeated use.
At first glance, sodium bifluoride seems straightforward: a white, crystalline salt that dissolves easily in water. It doesn’t jump out as a star player unless you know what you’re looking for. Yet, in jobs like surface cleaning, water fluoridation, or aluminum processing, it proves its value time and time again. Folks working with it often appreciate the balance it offers—reactive but manageable. Compared with wilder fluorine compounds, sodium bifluoride feels like a steady hand.
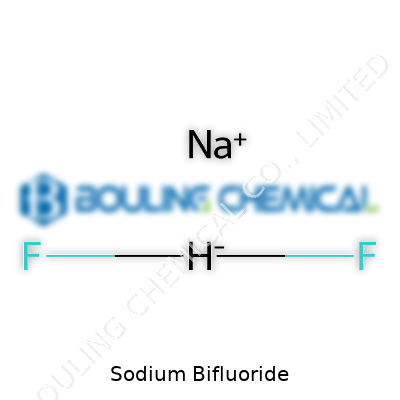
Sodium bifluoride isn’t flashy. You get a solid that handles humidity without fussing too much, a decent shelf life, and predictable reactions with water. The chemical formula, NaHF2, hints at its dual character. Pour it in water, you get sodium ions and bifluoride ions, and these set off a dance of acid-base action. You can pick up a faint vinegar-like smell when the solid interacts with moisture—hydrofluoric acid escaping. Anyone who’s spent time in a lab knows to take that as a warning. It’s dense—over two grams per cubic centimeter—which might not matter unless you’re weighing out drumfuls for an industrial process. Its melting point sits well above the boiling point of water, meaning it handles heat without issue unless you start pushing things hard.
Specifications don’t just collect dust on paperwork. They tell you everything about how a batch will act when the chips are down. Purity levels matter, especially for uses that touch drinking water or electronics. Companies keep a close eye on moisture content, free acid, and sodium fluoride traces. Most global regulations call for clear hazard labels and instructions—nobody likes surprises when handling fluorides. You see warnings about skin corrosion and toxicity, along with advice on storing in dry, sealed containers. Fail to respect those labels and the compound gets dangerous, fast.
The classic route comes straight out of the textbook: run hydrofluoric acid over sodium carbonate or sodium fluoride, tap off the water, and sodium bifluoride drops out. The method works on a small scale or in a fifty-gallon drum, which keeps production costs lower than other complex reagents. Some operations fine-tune temperatures and concentrations for better yields, but the basics rarely change—a quick rinse with cold water might finish the job, sparing no one from the sting of raw hydrofluoric acid. Once the process settles, you get a white crystalline product ready for packaging and use.
Sodium bifluoride isn’t content to sit quietly on a shelf. Expose it to moisture, it gives up hydrofluoric acid vapors. Toss it in a mix with acids or bases, and you watch bifluoride ions swing in with some punch—vapor-phase etching, cleaning stubborn stains, or tweaking aluminum surfaces to an almost surgical level of detail. At higher temperatures or with the right partners, it can react further to produce sodium fluoride or act as a scavenger for trace metals. For folks who like to tinker, sodium bifluoride’s chemistry offers a flexible platform for further modification. Researchers toy with doping it or coupling it with other salts to create custom reagents for tough jobs. But the wild card remains its ability to release hydrofluoric acid—there’s risk and reward in each reaction.
It goes by a handful of names: sodium hydrogen difluoride, sodium acid fluoride, or sometimes just SBF among veterans of the trade. Each name hints at its chemical structure, but watch for confusion on shipping documents or safety data sheets. Even retail suppliers use variants, reflecting the position in a lineup of fluorides. I’ve seen “difluorosodate” on older chemistry manuals or “sodacid” as a brand name, mostly outside English markets. Do the research, line up synonyms, and cross-check to make sure everyone’s talking about the same powder.
Handling sodium bifluoride safely demands respect and attention. Shortcuts bring serious consequences—I’ve met folks with chemical burns who didn’t realize quite how quickly bifluoride penetrates gloves or even fabrics. Hydrofluoric acid lurking in dust or vapors eats through bone, not just skin. Regulations require heavy-duty gloves, face shields, good ventilation, and—above all—a clear protocol for accident response. Most shops keep calcium gluconate gel nearby as a first-aid post-exposure treatment. On waste disposal, nothing goes down an ordinary drain; facilities collect and treat used solutions as hazardous waste, following strict environmental rules. These standards are not just bureaucracy—they reflect real lessons paid for with pain and lost work time.
Sodium bifluoride shows up everywhere: etching glass to a soft matte finish, prepping metals, cleaning stubborn mineral deposits, and helping formulate cleaning agents for tough industrial grime. Water treatment facilities use it as a source of fluoride ions, while aluminum fabricators lean on its surface-modifying abilities. Some sectors eye it for electronics and ceramics work, since its reactivity can fine-tune surfaces beyond what most acids allow. Hobbyists and small foundries dabble with it for pickling metals, though safety practices are crucial at any scale. As fluoride policies and industrial cleaning needs evolve, applications expand and change, but the chemistry remains.
Research teams dig into sodium bifluoride’s structure, reaction patterns, and environmental impact. Academic groups probe ways to limit hazardous byproducts during use and disposal. Some focus on tweaking crystal growth or adjusting its conversion into sodium fluoride, hoping to tailor properties for niche applications in batteries or advanced ceramics. Ongoing studies look for ways to capture or neutralize hydrofluoric acid, reducing overall risk. The push for “greener” processes in the chemical industry also forces a close look at bifluoride’s whole lifecycle, from mining feedstock to final disposal. New research could shape regulations and spark fresh uses or safer handling protocols.
Toxicity doesn’t take a holiday just because a chemical is useful. Sodium bifluoride deserves every warning it’s given—skin contact leads to deep burns, and inhalation can cause lasting respiratory damage. Researchers measure how it moves in soil and water, tracking the spread of fluoride ions and possible impacts on groundwater. Long-term exposure studies in animals show risks to bones, teeth, and kidneys. Medical teams handling accidental exposures face tough odds unless proper treatments are started early. Ongoing lab studies compare various protective barriers, seeking the best fabric or material mix for gloves and clothing. The more we learn, the tighter regulations get, but the balance between utility and safety is tough to strike. User education makes the biggest difference—folks who know the risks can take steps to avoid them, while ignorance stacks the odds in favor of accidents.
Where does sodium bifluoride go from here? As industries search for sharper tools to process metals, treat water, and clean up old sites, sodium bifluoride holds a seat at many tables. Environmental rules may steer demand toward lower-toxicity alternatives, but the compound’s unique reactivity keeps it in the conversation. Technologies like battery production, ceramics manufacturing, and advanced etching all offer possible growth, provided safety and disposal challenges find answers. R&D teams could find ways to trap or recycle hydrofluoric acid on-site, making bifluoride safer to handle. As regulatory agencies tighten rules on workplace exposure and emissions, producers will face tough choices about process changes or extra safeguards. Every improvement costs money, but the payoff comes in worker safety, environmental protection, and peace of mind.
Sodium bifluoride isn’t something most folks will find in their kitchen cupboard, but it helps make a lot of things in modern life tick. This chemical, known in labs as NaHF2, stands out mostly for its strength as a cleaner and an industrial processing agent. It’s not some household staple, yet it plays a surprising part in the world around us, from etching glass to keeping swimming pools in good shape.
One of the better-known jobs for sodium bifluoride lies in glass etching. Factories use it to get that frosted look on showers and windows—the kind of smooth texture that feels both fancy and practical. It works by reacting with the silica in glass, letting designers carve logos or create patterns that never rub off. Artists prefer this method too; it delivers precision and makes glass stand out without needing lasers or expensive machinery.
Another place sodium bifluoride turns up is in the world of metal. Factories use it to clean up surfaces before aluminum or steel parts move to the next step—maybe painting, maybe coating. Without these prep steps, paints can flake and protective layers peel. It’s also involved in pickling stainless steel, stripping off rust and scale to leave metal shining and ready for further processing. It’s a reliable way to make sure that shiny railing or that gear inside a machine stays strong and looks sharp.
I remember as a kid, swimming at the community pool and enjoying the clear water—not knowing back then about the chemicals working behind the scenes. Pool technicians sometimes use sodium bifluoride as a source of fluoride ions, which help tackle bacteria and algae. That means less green slime and fewer chemical headaches for anyone maintaining community swimming areas. Operators depend on it as part of their public health toolkit, and it does its part quietly.
Foundries—factories that pour melted metal—also depend on sodium bifluoride. They use it to help flux metal, pulling out unwanted oxides and improving the flow, which results in castings that last and don’t break down soon after leaving the shop floor. It’s tough work, often hot and dirty, and the right chemicals can make a real difference in both product quality and worker safety.
None of this is to say sodium bifluoride should be handled lightly. It can cause burns if spilled or misused, and the dust isn’t friendly. Safe handling means gloves, proper ventilation, and training. Companies that use this chemical invest heavily in worker safety—something that benefits the community as well. I’ve spoken with folks in the field who stress that a focus on strong safety measures pays off, preventing injuries and fines and, more importantly, building trust in local workplaces.
Engineers and researchers keep searching for less hazardous options that still get the job done. Some industries now use alternative etchants or safer pre-treatment chemicals for metals, hoping to reduce reliance on sodium bifluoride. These efforts matter, as stricter environmental rules push companies to find products that won’t harm workers or leave a toxic trace.
Sodium bifluoride will probably stick around for a while, at least where its unique properties are called for. Its story is one of hidden contributions—a powerful cleaner and etching tool that’s kept at arm’s length, managed with care, and quietly shaping the infrastructure of daily life.
I remember working at a pool supply store fresh out of high school. Bags marked with bold hazard symbols always made me nervous, and sodium bifluoride topped the list. Back then, I didn’t understand the big deal—now I do. Sodium bifluoride isn’t something to scoff at. It’s a chemical with important uses, but there’s no denying it brings genuine hazards, especially for health.
Sodium bifluoride shows up in a few places: water treatment, glass etching, even in some cleaning products. It might sound unremarkable, but its chemistry packs a punch. This salt breaks down to produce fluoride ions and hydrofluoric acid. Hydrofluoric acid may sound familiar from chemistry class nightmares. The acid can burrow through skin, attacking deep tissue and even bones. Exposure isn’t just painful—it can be downright dangerous.
Skin contact is a huge risk. I’ve seen what can happen from accidental splashes. Burning, redness, and blisters usually show up fast. Worse, the pain sometimes feels mild at first even as the acid keeps working. That false sense of security means some people delay washing it off, which only increases the threat. Eyes are even more sensitive; permanent damage or even blindness can happen if sodium bifluoride dust or liquid hits the eyes. Breathing in dust hits the respiratory system. Coughing, sore throat, wheezing—those symptoms can escalate quickly for anyone with asthma or weakened lungs.
Sodium bifluoride doesn’t just irritate. It’s toxic. Swallowing some can trigger nausea, vomiting, diarrhea, and severe abdominal pain. In larger doses, it can mess with the heart’s rhythm or even halt it altogether. Victims sometimes need hospital-level treatment for these exposures, including specialized gels or calcium injections to stop the chemical from binding to calcium in the body and causing deadly low calcium.
This chemical is no rookie when it comes to workplace incidents. The CDC and OSHA both warn about exposures, and strict handling rules exist because of its reputation. Inhaling or swallowing dust doesn’t always happen by accident. Sometimes inadequate training or casual attitudes about protective gear lead to mishaps. Over the years, those mishaps have resulted in lost wages, burned skin, and in rare cases, fatalities.
Solutions demand commitment, not half-measures. Training workers goes far beyond reading a label—it means hands-on demonstrations and honest discussions about risks. Personal protective equipment does its job only if it’s used correctly. Gloves made from nitrile or neoprene, goggles that create a solid seal, proper ventilation—these basic steps carry real power. It’s not about bureaucracy; it’s about people going home with all their fingers, their eyesight, and no emergency room stories to tell.
Consumers should double-check product labels. If you handle cleaning products or other substances with this chemical, the same respect that’s demanded in an industrial setting applies at home. Keeping products locked away and using them in well-ventilated areas goes a long way toward staying safe. The same goes for quick access to running water—washing right away can make all the difference after a spill or splash.
Sodium bifluoride’s dangers often hide in plain sight. It works well for tough jobs but doesn’t belong anywhere it can hurt people. This substance reminds us every day: just because something is legal or widely used doesn’t mean it’s harmless. Awareness, solid education, and practical steps keep folks from learning its risks the hard way.
Sodium bifluoride pops up in industry more than most people realize. It plays a role in cleaning, water treatment, even in some etching processes. This stuff keeps warehouses running, but not without a catch. Many overlook just how reactive and hazardous it can be. After seeing warehouse mishaps up close, I understand that safe storage is no minor detail—it's a community responsibility.
Stacking drums in a corner or letting bags rest near a leaky tap sounds trivial until you deal with a spill or a chemical burn. Sodium bifluoride grabs moisture out of the air, and those tiny white granules turn dangerous fast. It reacts with acids, releases toxic gas, and eats through less resilient metals. Equipment corrodes, containers get weak spots, and people end up sick or worse.
I’ve watched the results of cutting corners—corroded shelving, emergency shut-downs, and coworkers in the hospital. Moisture is the enemy here. Humid spaces, condensation on ceilings, or careless routine all open the door to trouble. Only storage in cool, dry areas slows down the risks. Airtight containers, made from plastic or lined with non-corrosive material, block out humidity. Anyone who has cleaned up a powdery spill knows that even a little airborne sodium bifluoride stings eyes and throats. Good lids and solid bins aren’t just handy, they protect lives.
OSHA and NIOSH don’t tiptoe around it: they call for designated storage zones, closed containers, and clear hazard signage. I’ve seen inspectors raise eyebrows at warehouse setups that looked neat, but sat next to lunch rooms or had poor ventilation. Sodium bifluoride gives off hydrofluoric acid if it even thinks about water, so anything less than proper separation from water sources just doesn’t cut it. No joke—hydrofluoric acid can burn down to the bone and won't always show damage right away.
To do storage better, crews need practical training, not just a safety video on repeat. Workers should see what "secure storage" means in real spaces, not just on paper. The system ought to support routine checks—someone looking for rust, leaks, and missing labels helps more than any stack of paperwork ever will. Clear spill protocols save panic and confusion. Sensitive materials like sodium bifluoride don’t forgive sloppiness.
Labels matter, but knowledge and teamwork matter more. Experienced staff should show new hands why these steps save trouble. I’ve witnessed close calls avoided because someone paid extra attention on a long shift. Keeping sodium bifluoride safe is about thinking ahead, looking out for the next guy, and building habits you don't have to think twice about.
The real risk comes from shortcuts, rushed procedures, or just plain forgetting how nasty sodium bifluoride can get. Every workplace that deals with it faces tough choices, but cutting corners helps no one. By showing what good storage looks like, and sharing stories about what happens when practices slip, people take the threat seriously. Only by working smarter and sticking together do we keep this quiet danger from becoming tomorrow’s headline.
I still remember the sting of caution I felt the first time I unboxed a drum of sodium bifluoride in the backroom of an industrial plant. The label had warnings splashed across it, a thick chemical scent in the air, and my supervisor leaning over my shoulder with advice as sharp as the crystals in the drum. Working with this substance isn't just another ninth-grade chemistry experiment; it’s something that demands a heavy dose of respect. Sodium bifluoride goes quickly from helpful to hazardous if you ignore the risks attached to it.
Sodium bifluoride slots easily into industrial cleaning, glass etching, and even some water treatments, riding on its aggressive reaction with calcium and silicates. But its punch isn't just for the grime in pipes or glass surfaces; it can dig into skin and lungs too. The substance releases hydrogen fluoride, which doesn’t play around. A little slip, a forgotten glove, or lifting your mask for “just a minute,” can mean burns or much worse. Medically documented incidents have shown that even brief, low-level exposure causes skin irritation, eye damage, or respiratory distress. It doesn’t care if you’re a pro or a rookie.
Gloves and goggles aren’t extras—they’re essential. In my years on the plant floor, I watched newcomers who figured splash goggles would fog up and get tossed aside. Within weeks, someone always got a warning lesson: eye redness, coughing fits, a rash where sleeves ended. Nitrile gloves and a full-face shield are the bare minimum—rubberized aprons and boots add a level of security you’ll never regret. Face masks with proper acid vapor filters beat a simple dust mask any day. The point is straightforward: if the gear feels over the top, you probably need it.
Never trust an open door as your air filter. Sodium bifluoride dust and fumes hang low in stagnant rooms. I saw ventilation make the difference on more than one occasion, with local exhausts catching particulates before anyone noticed. Negative pressure systems work well, and even portable extraction fans make chemical “clouds” less likely. Find ways to keep air moving—those lungs aren’t factory parts you can just replace.
Too many accidents start with casual storage. Sodium bifluoride belongs in a moisture-free zone, sealed tight and away from other acids or oxidizers. Water kicks off dangerous reactions, even in small amounts—imagine a shelving leak or a spill from a nearby pipe. In my old crew, close calls happened because people stacked incompatible drums or taped over splits in bags. Spent containers need to be washed and stored for disposal, not reused for “just water”.
The best defense against chemical injury has always been hands-on training. OSHA doesn’t hand down rules for fun—those classes replay emergency drills that save lives. Pair every training with a healthy dose of skepticism about shortcuts. If you see spills, report them; if you’re not sure about safe handling, ask. Overconfidence bites hard, and chemicals like sodium bifluoride never care who’s holding the scoop.
Sodium bifluoride lurks in a surprising number of everyday places—from industrial cleaners and textile factories to metal surface treatments. It is a compound that packs a punch, breaking apart grime, etching glass, and processing metals. The trouble with this chemical is that it rarely stays put. Rinsed down drains, it sometimes flows beyond factory fences, carrying potential problems into soils and rivers. I grew up near an industrial park that used similar chemicals, and we never swam in the creek behind the property. Most families shared quiet worries about what seeped into our groundwater.
Fluoride, the backbone element in sodium bifluoride, can throw off the delicate balance of aquatic life. Fish and insects depend on a specific range of mineral concentrations to breathe and move. When sodium bifluoride enters a river, it splits into fluoride ions. Concentrations above safe levels cause fish to swim erratically, even stop eating, and sometimes don’t survive. According to the US Geological Survey, long-term fluoride buildup poses a risk of bone damage for animals that drink from affected sources. They do not have the luxury of choosing bottled water.
Plants absorb minerals through their roots, pulling up whatever lingers in nearby dirt. Farmers across the world have noticed how fluoride exposure stunts the growth of crops and leaves plants vulnerable to disease. Even relatively low levels lead to browning leaves, twisted roots, and weaker yields. While most commercial operations claim they’re responsibly managing waste, there is no easy fix once contaminated runoff leaks into farmland.
Factories that use sodium bifluoride create dust and fumes, especially during cleaning or maintenance. These particles don’t magically disappear. Research from environmental health groups highlights that chronic fluoride exposure causes respiratory irritation, sore throats, and longer-term health issues. Community members living near heavy-using plants face the most exposure, so regular checks and transparent monitoring make a real difference. I’ve watched local leaders organize community air testing, sometimes holding up jars in town meetings filled with “smog catch” from backyards—a vivid reminder that what leaves a smokestack doesn’t just vanish.
Environmental oversight rarely matches the speed of industrial growth. For sodium bifluoride, stronger regulations for discharge and waste handling mark a starting point. This involves more than fines; it takes robust inspections, public reporting, and real consequences for shortcuts. Switching to closed-loop systems, where rinse water is treated and reused on site, offers a proven way to keep harmful byproducts from spreading.
Communities benefit from clear labeling and education about chemicals like sodium bifluoride, especially around schools, parks, and farms. Simple steps—like regular soil and water testing—cast a spotlight on problem areas before they spiral out of control. My own community saw cleaner water and healthier fish populations after pressuring local companies to upgrade their waste treatment and share quarterly data. Taking action does not demand heroic effort, just steady attention and honest communication.
Sodium bifluoride will keep finding its way into industry for the foreseeable future. Its real-world impact depends on whether oversight and public vigilance remain strong. If we want healthy soil and clean rivers, everyone—workers, companies, regulators, neighbors—has a stake in being alert. Facts show that cutting corners with this compound causes more costs down the road, both for people and for nature.