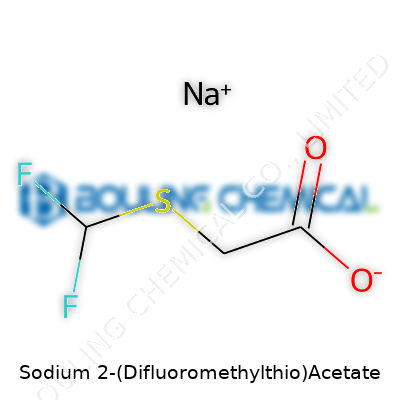
Sodium 2-(Difluoromethylthio)acetate sits at the intersection of old-school organic chemistry and fresh innovation. For decades, chemical research has hovered around the idea of tweaking small molecules in search of new reactivity or better biological profiles. The introduction of difluoromethylthio groups brings a unique twist to that story. Early research in the '80s spotlighted difluoromethyl modifications for their stability and electronegativity, especially in pharmaceuticals and agrochemicals. Over time, as synthesis routes for these motifs improved, commercial and academic labs turned their eyes to compounds pairing this group with versatile acetate chemistry. Sodium salts like this one moved into the spotlight as intermediates, surfacing in patents and research papers exploring new routes for active pharmaceutical ingredients and crop protection agents.
Recognizing the rise of fluorinated compounds in everything from drug discovery to advanced materials, Sodium 2-(Difluoromethylthio)acetate carved a spot as a niche but valuable building block. Chemists praise its manageable handling and relatively straightforward synthesis compared to some more exotic fluorinated chemicals. It bridges the gap between the durable, often stubbornly inert difluoromethylthio group and the very reactive carboxylate moiety. The result is an intermediate that allows synthetic chemists to pivot between sulfur, fluorine, and acetic chemistries without sweating over harsh conditions or unstable reagents.
This compound shows up as a crystalline, probably white or off-white solid, with the unmistakable sodium salt character: typically water-soluble, often forming free-flowing powders. Its difluoromethylthio group injects a notable lipophilicity that shifts its solubility compared to simpler acetate salts, but the balance usually lets it dissolve or disperse in a range of lab solvents. The presence of two fluorine atoms changes everything—the electron-withdrawing kick from fluorine atoms throws off proton acidity, slows common decomposition pathways, and pushes the chemical’s stability beyond what you’d expect from a sulfur acetate. The physical stability helps in storage and handling, sidestepping those headaches that unstable reagents can cause for both researchers and the folks in shipping and receiving.
Specifications always matter, especially for chemicals making their way into regulated research or manufacturing. Labs require tight control over impurities, especially since trace contaminants in building blocks like this compound often snowball in downstream processes. Good batches of sodium 2-(difluoromethylthio)acetate arrive with purity above 97 percent, often supported by NMR and chromatography data. Hydrate levels, particle size, and sodium content get extra attention, as even slight shifts throw off synthesis work or analytical calibration. Labeling needs to clearly flag fluorine content—which matters for both waste handling and chemical compatibility—as well as GHS hazard classification, reflecting the compound’s potential irritant or corrosive properties.
Preparing sodium 2-(difluoromethylthio)acetate often starts from difluoromethylthiol or its derivatives, passing through alkylation or thiol-ene reactions with a suitable haloacetate or chlorinated acetic acid, followed by base treatment to yield the sodium salt. Techniques here evolved as fluoro-organic chemistry grew up—historical approaches used harsh reagents, but greener or more benign methods now get the job done with better yields and less toxic waste. Key process steps—especially quenching and neutralization—have to keep the decomposition risk low and minimize exposure to fluorinated side products that can prove tough to clean up.
This compound thrives as a modular piece in synthesis plans. Its acetate fragment undergoes classic nucleophilic substitution, condensation, and decarboxylation. The difluoromethylthio group tells a different story. That sulfur, sandwiched between a methyl and two robust fluorines, offers a surprising resilience to oxidation, enabling chemists to explore selective cross-coupling or oxidative transformations. Both pharma and agrochemical scientists probe its ability to stabilize active drug fragments and help shuttle fluorine into metabolic hot spots—fending off unwanted degradation in vivo or in field conditions. Sometimes, simple hydrolysis, alkylation, or amidation unlocks even more complex target molecules.
Every chemical ends up with a roster of alternate names, and this one is no exception. While "Sodium 2-(difluoromethylthio)acetate" remains the IUPAC anchor, researchers and suppliers also refer to it as sodium difluoromethylthioacetate, S-(difluoromethyl)-acetic acid sodium salt, or even less systematic abbreviations in-house. Supply catalogs add further variants, based on salt hydrate form or loose translation from other languages, so double-checking chemical structures remains essential for avoiding mix-ups in ordering or process planning.
Working with sodium 2-(difluoromethylthio)acetate asks for the usual lab discipline that comes with handling reactive organic salts, made a touch stricter by the presence of fluorine and sulfur. Inhalation of dust or direct skin contact could lead to irritation or sensitization—a point I’ve seen come up in even well-run labs that got hurried. Protective gloves and splash goggles become routine in my own work, and well-ventilated fume hoods keep potential exotherms or off-gassing within safe limits. Waste shows up as a specific concern, since liberation of low-level fluorinated volatiles demands collection and treatment, not casual drain disposal. Institutional safety teams have clear input here, often calling for detailed risk assessments before first receipt and use of new batches.
The reach of sodium 2-(difluoromethylthio)acetate threads through pharmaceuticals, crop protection, and synthetic organic chemistry. Drug developers press these fluorinated units into design motifs for new candidates with tweaked bioavailability and metabolic resistance. Farmers benefit through newer, more robust plant treatment agents derived from such structures, extending residue life and weather resistance. I’ve watched as start-ups in my network tap this chemical for high-performance coatings or specialty adhesives where standard sulfur or fluorine compounds just don’t cut it—proof that niche chemistry often leads to mainstream breakthroughs over time. The fine chemical sector, always hungry for new building blocks, continues to integrate compounds like this into multipurpose libraries for rapid process screening.
Labs keep exploring what this compound can achieve. Academic chemists, especially those involved in fluorine methodology, explore ever-milder and increasingly economical ways to append difluoromethylthio fragments onto active molecules. Within pharma and agro, the R&D conversations revolve around how these tweaks translate into performance—what off-target effects can be reduced, which metabolic processes might be slowed? Each patent wave brings new reaction types, often highlighting the swapped-in difluoromethylthio as the critical difference behind improved properties. This is an area that feels alive with possibility: better yields, safer work flows, smarter waste management, and discovery of still more applications.
Nobody wants an innovation that backfires due to environmental or human health hazards. Toxicity screens tell a complex story with fluorinated organics, and this compound fits squarely in those concerns. While acute toxicity ranks below more notorious fluorinated chemicals, chronic effects, bioaccumulation, and the ultimate fate of either the intact compound or its breakdown products remain active research topics. Scientists look for both short-term cytotoxicity and longer-term ecological impacts. Proper animal and cellular models stay critical for assessing risks, and as regulatory frameworks evolve, new data drives process improvements and updates to safety practices.
Momentum keeps building. Better, cleaner methods for fluorinated thioacetates mean broader access, lower cost, and more robust safety protocols. Green chemistry advocates push for recyclable catalysts and minimized waste, which helps quiet the environmental concerns that fluorine chemistry sometimes raises. On the application end, ongoing advances in drug design, crop yield improvement, and high-performance materials keep pulling compounds like sodium 2-(difluoromethylthio)acetate into the limelight. Partners across academia, industry, and regulation collaborate more closely than ever, driving a feedback loop of safer, more efficient chemical innovation. I’ve seen firsthand that the bridge between synthetic ingenuity and practical utility often rests on unsung but carefully designed intermediates—and sodium 2-(difluoromethylthio)acetate has a clear seat at that table for years to come.
Every so often, a chemical name lands on a desk or in an email, and it brings more questions than answers. Sodium 2-(Difluoromethylthio)acetate, with its mouthful of a name, falls into that category. Its formula, C3H3F2NaO2S, sums up a story of curiosity in chemistry. But to many outside the lab, those letters and numbers raise only eyebrows and not much else.
Start with the “difluoromethylthio” group. Chemists prize these kinds of side chains, often because they provide special properties for pharmaceuticals and agrochemical research. Attaching fluorine atoms to molecules can improve how drugs work in the body and how stable they stay on the shelf. Sulfur swaps introduce a different kind of stability and often change how a compound interacts with biological systems. Put sodium on the end, and now it dissolves in water with a little gusto, broadening where it can go next.
C3H3F2NaO2S tells chemists this molecule carries two fluorines, one sulfur, and links to a sodium salt. Structurally speaking, that’s not trivial. Each substitution or branch on the molecular backbone changes both how it behaves and how it can be used. The minute you sprinkle fluorine into things, everything — from metabolism to environmental persistence — shifts. I remember looking into the role of fluorine in various drugs while working on a chemistry project. It struck me just how often these tiny changes can lead to bigger breakthroughs or tougher regulations.
Outside the lab, someone might shrug at the idea of fine-tuning molecules. But it plays out in real life, for better and for worse. These compounds can lay the groundwork for new medicines. They can also raise health and environmental questions, since fluorinated and sulfur-containing compounds don’t break down easily in nature.
The challenge sits in balance. Many fluorinated molecules led to some of the world’s most important drugs and crop protectants. Still, that persistence in the environment signals a need for caution. History taught us about problems with persistent organic pollutants. Stories of PFAS — the so-called “forever chemicals” — highlight how things designed to last can end up everywhere, from water supplies to our blood.
As more researchers start using complex compounds like sodium 2-(difluoromethylthio)acetate, it puts pressure on the field to weigh benefits against risks before scaling up. Even as new molecules open doors in medicine and agriculture, difficult questions about lifecycle and waste should not get swept under the rug. Good science means thinking about the full picture, from the first beaker in the lab through the journey to wastewater and landfill.
Improvement starts with careful design—building chemicals that do their jobs but do not linger beyond their welcome. Investing in greener synthesis, transparency about environmental fate, and tighter industry oversight matter. No single formula solves everything, but strong, shared accountability between innovators, regulators, and consumers helps keep progress on track without piling up new chemical concerns.
Plenty of chemicals fly under the radar, but Sodium 2-(Difluoromethylthio)acetate shapes more than a few harvests each year. Over the past decade, I’ve watched the pressure for sustainable and efficient crops change how farmers and scientists think about every input, especially chemicals involved in protecting plant health. This compound stepped into the spotlight for its work against weeds and pests, offering a new angle in fields where resistance to old-school formulas kept building up.
On most farms, weed control runs up huge bills and soaks up hours that could go to other work. Traditional herbicides start losing ground as weeds adapt and push back. Sodium 2-(Difluoromethylthio)acetate stands out because its unique chemical structure means it attacks plant growth in ways that differ from older chemicals. The product targets specific plant enzymes, so you get results without heavy-handed doses. You don’t see the same crop damage or toxicity problems that make many farmers anxious when trying something new. Real feedback from growers shows how switching to modern options like this one helped them dodge resistant weeds that used to overrun whole fields.
Big industrial farms usually make headlines for adopting new weed killers, but home gardeners have picked up on this compound as well. I’ve met hobbyists frustrated with weed sprays from local stores—spending more every year for less and less control. By turning to this compound, they find help with persistent weeds that laugh off whatever’s in the average bottle. It works as a selective herbicide, going after problem plants without wiping out everything green in sight. This changes how people approach yard management and chemical use around the house, especially near pets and kids where safety matters.
Everyone whose pay depends on the crop yield sweats over pesticide laws and consumer worries. Even if you don’t farm, the cost of groceries and food supply reliability boil down to what happens out in the fields. Sodium 2-(Difluoromethylthio)acetate caught attention from food safety agencies because of its short breakdown cycle and relatively low residue levels in food. Compared to stubborn chemicals that stick around on produce or run off into water, this is a step toward safer consumption. Studies show that food treated with it meets the tough standards set by many markets, including the US, Europe, and Australia.
It’s not just herbicides in the spotlight—this compound plays into fungicide research too. Plant diseases shape entire growing seasons, sometimes wiping out tons of food before anyone gets a taste. Scientists keep looking for options with new chemistry to throw off fungi and blights that learn how to beat traditional methods. Early field trials blend Sodium 2-(Difluoromethylthio)acetate with other active ingredients, giving disease management teams one more tool to fight evolving bugs and molds.
Thinking ahead, the solution requires more than just swapping bottles. Fighting resistance means careful rotation of chemicals and honest data sharing from fields, labs, and processing lines. Tools like Sodium 2-(Difluoromethylthio)acetate give choices, but keeping them effective means using them responsibly, not turning to them as a crutch at every hint of trouble. Farmers, regulators, and researchers carry the load here, working out best practices that stretch real results without causing trouble downstream.
As someone who’s walked the rows, talked with growers, and followed the science, I see Sodium 2-(Difluoromethylthio)acetate as proof of what good chemistry can achieve. Progress keeps coming, but it stays meaningful only when tied to open data, real-world feedback, and a focus on food safety for everyone.
Anyone who’s ever watched a loaf of bread sprout mold on the counter knows storage can make or break a product. These guidelines aren’t just fine print—how we treat products day to day can change their safety and value. In my years working alongside pharmacists, food handlers, and even warehouse crews, one lesson keeps coming back: most spoilage and accidents trace back to bad storage, not bad luck.
Look at medications with instructions like “keep below 25°C, protect from moisture”. A bottle sitting near a bathroom window or in the glove compartment loses punch long before the expiration date. Too much heat wrecks chemical stability, so drugs might stop working or turn risky. For food, even small lapses bring pests or breed germs that don’t just ruin dinner—they send people to the hospital.
Humidity ruins everything from crackers to electronics. I‘ve seen electronics in tropical climates corrode fast when left unsealed. On the other hand, dry air can shrivel produce or dry out ointments. That’s why you’ll find silica packets or “store in a cool, dry place” warnings—they aren’t marketing extras.
Some items really don’t like light. Certain vitamins and photosensitive medicines break down in daylight, and so do many natural oils. Orange bottles in a pharmacy aren’t just for looks—they block UV rays that gradually change the product inside. As for air, oxygen exposure speeds up oxidation. Anyone who’s tasted stale chips or watched fruit go brown has seen oxidation at work.
Storage also matters for safety. Kids and pets don’t read warning labels, and that’s how accidental poisonings happen. While working in a retail pharmacy, locked cabinets kept dangerous medicines out of easy reach. In food prep, sealed bins and clear labeling stop cross-contamination and help staff grab what they need on a busy shift.
Good storage helps avoid waste, too. Shelf-life stretches longer when products get steady temperatures and protection from moisture. Households that organize pantries and maintain fridge hygiene throw away less; warehouses tracking date codes cut out panic recalls and product loss.
There’s always a clever gadget—the smart fridges with reminders, climate-controlled warehouse shelves, even phone apps that alert when medication storage is off. Those are luxuries. The basics work: keep medicines away from sinks and heat. Seal snacks in airtight containers. Don’t shove raw meat above ready-to-eat food. For most products, this isn’t high science. It’s common sense backed by experience and a look at what goes wrong.
Consumers and businesses both get hit in the wallet when storage fails. Clear guidance, reasonable monitoring, and an understanding of why these rules matter keep products safer and families healthier. My advice comes from real mishaps I’ve seen. If a label suggests a particular spot or temperature, trust it. The cost of ignoring storage advice usually shows up sooner than anyone thinks.
Long, complicated chemical names have a way of turning people off right away. Most folks see “Sodium 2-(Difluoromethylthio)acetate” and just want to back away. Still, ignoring the conversation wouldn’t be a smart move. My experience kicking around in research labs taught me that complicated chemistry can sometimes hide very real risks. Sodium 2-(Difluoromethylthio)acetate has been catching some attention in specialty chemical and agricultural circles. People want to know if it’s dangerous to work with, or hazardous over time.
Sodium 2-(Difluoromethylthio)acetate isn’t exactly a classic household name—for most of us, it’s a specialty tool, sometimes used by chemists, possibly as an intermediate in making crop protection agents or pharmaceuticals. The big question is: Does this stuff do harm to people or the environment?
Data from similar difluorinated compounds suggest we should take their risks seriously. For example, organofluorine chemicals don’t break down easily in water, soil, or air. Some can slip into water supplies and hang around for years. In a world already struggling to handle PFAS contamination—think “forever chemicals”—that sort of environmental persistence raises red flags.
Toxicity can be tricky to nail down with less common compounds. Early studies with other difluoromethyl derivatives sometimes show liver and kidney impacts after high exposure in animals. My time reviewing chemical safety reports reminded me just how much difference small changes in a molecule can make. Some seemingly minor tweaks flip a safe compound into something that upends a worker’s health, or leads to a costly environmental cleanup. So, it pays to assume careful handling is necessary, even if no major spills or mass exposure events have hit headlines yet.
Even when a chemical is relatively obscure, regulators look out for anything persistent, bioaccumulative, or toxic. If Sodium 2-(Difluoromethylthio)acetate displays these qualities, restrictions or reporting rules may follow. Most regulatory agencies stress the value of “precautionary principle” here: take steps to reduce risks even if the full toxicological picture isn’t painted.
Engineers and safety managers at chemical plants have learned to build safeguards like local ventilation, personal protective equipment, and sealed systems into every process involving unfamiliar organofluorines. That’s not overkill—it’s just learning lessons from earlier chemical regrets. Nearly every older chemical that wound up on the “hazardous” list started with uncertainty, small leaks, and workers trusting that “no news is good news.”
Trustworthy suppliers run repeated toxicity tests and publish real data, but that info sometimes stays behind the scenes. Demand for more transparency could push companies to release safety and ecological impact data for Sodium 2-(Difluoromethylthio)acetate. Real-world exposure scenarios should also receive careful risk assessments. Just because the stuff sits in a drum somewhere out of sight, doesn’t mean it’s someone else’s problem.
If I’ve learned anything in years of following chemical safety stories, it’s that transparency, clear labeling, and diligent training keep surprises at bay. Chemical names are intimidating. Scrutiny, safety data, and solid rules are not.
Anyone with a background in chemistry or pharmaceutical work has run into the question of molecular weight like it’s an old friend. Even if someone doesn’t care about chemicals day-to-day, this little number can shape so much about how things behave – from laboratory work to industrial synthesis or even drug design. I remember the first month in an undergraduate organic chemistry class, sitting with a cheap calculator and scratch paper, plugging in atomic weights just to get something as fundamental as this. Accuracy mattered. Get a digit wrong, and downstream work gets messy—yields crumble, reactions drift, and everything needs to be recalculated.
Let’s break this down: sodium 2-(difluoromethylthio)acetate. It doesn’t roll off the tongue, but it says plenty about the compound’s structure. You have a sodium atom, a difluoromethylthio group, and acetate. Here's the important part—finding the molecular weight isn't arcane knowledge, but it’s easy to lose track of a hydrogen here or a fluorine there. Precision counts.
Drawing from experience, the calculation goes by counting each atom:
Add these up and you’ll land at about 163.11 g/mol for sodium 2-(difluoromethylthio)acetate. Double-checking, the numbers add up. This isn’t just an academic exercise. In real labs or production settings, someone somewhere needs to weigh out a clean batch, calculate solubility, or figure out dosages for a study. Scrambling to find an accurate molecular weight slows everyone down.
Molecular weight sets the tone for so much. In drug discovery, a compound with a molecular weight under 500 g/mol tends to get more attention, since large molecules struggle to cross cell membranes. Synthetic chemists track it to ensure they don’t waste precious reagents. In manufacturing, those digits tie straight into quality control and batch documentation. Getting it wrong leads to regulatory headaches and wasted money.
Plenty of companies build robust reference tables, but mistakes still happen. It’s why I always favored checking trusted sources like the CRC Handbook or PubChem for exact numbers, instead of trusting a secondhand spreadsheet. As someone who once rushed through a weigh-out, only to catch the error an hour and three steps later, I know the pain a simple math mistake causes. It’s humbling and expensive.
It isn’t glamorous, but hammering down these basics delivers real savings down the line. Training new chemists to check, double-check, and respect these values keeps error rates low. Digital tools and calculation software help, too, when they’re reliable and up-to-date.
As new compounds enter the market and research gets more complex, this sort of attention to detail builds trust. Workers learn to question their sources, confirm calculations, and build a habit of precision. Small choices, like nailing the molecular weight for this compound, lay the groundwork for everything else.