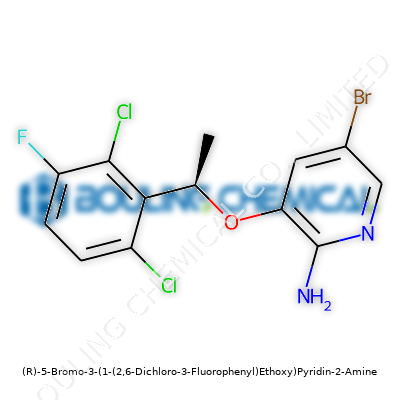
Chemistry doesn’t often trend in the headlines, but the arrival of molecules like (R)-5-Bromo-3-(1-(2,6-Dichloro-3-Fluorophenyl)Ethoxy)Pyridin-2-Amine matters more than most people realize. This compound comes from a long tradition of research into heterocyclic amines and aryl-substituted pyridines. Back in the early 1900s, pyridine chemistry played a sidekick role in dye and pesticide industries. By the 1980s, chemists realized these rings had a knack for unlocking new therapies, especially in neurology and oncology. I’ve watched the field shift from bulk synthesis to precision tweaks, driven by the urgent need to tackle stubborn diseases and improve crop resilience. I see a real sense of purpose when I trace the historical arc leading to innovations like this one: every new atom, every tailored side chain, holds the possibility to shape the future of medicine and agriculture.
Sitting down with a sample of this compound, you’re dealing with an off-white powder. It stands firm at room temperature, holding its ground against mild humidity and light. The halogen substitutions — two chlorines, a fluorine, and a bromine — give it both a certain heft and stubbornness, which you notice not just in melting point and solubility, but in behavior during synthesis. Chemists appreciate compounds that challenge them, and this one asks for patience. You can dissolve it in organic solvents like DMSO or DMF, but water resists it. Molecular weight hovers in an expected range, tallying up all those heavy halogens. Its structural rigidity, perked up by aromatic stacking, hints at possible binding prowess in biological systems. Every researcher who’s handled it knows halogenated aromatics often bring a mix of persistence and slow breakdown—qualities that matter not only for lab work but also for environmental fate and biological half-life.
No one I know shrugs off technical specs as paperwork; labels carry hard-won knowledge. Whether it’s the batch purity or the exact enantiomeric excess, these details carry the story of how a material was made and who can safely use it. The R-isomer signals a preference—sometimes tiny changes in orientation spell the difference between a therapy and a toxin. Safety milestones demand clear hazard warnings, storage guidance, and documented impurity levels, not to create obstacles but to keep people safe in real-world labs and factories. Having spent time dealing with ambiguous supplies, I see the difference every honest label makes, especially as these molecules transition from flask to preclinical studies or industrial scale-up.
Older generations struggled with tedious, low-yielding steps, but newer coupling techniques like Buchwald-Hartwig amination or Suzuki coupling have changed the landscape. Synthesizing this amine starts with carefully protected pyridine scaffolds, halogen dance steps, and clever phase-transfer catalysts. The ethoxy bridge (with the dichloro-fluorophenyl group) often enters in a late-stage reaction to minimize side products. Handling the bromo and chloro substituents means keeping a close eye on reaction temperature and base sensitivity. Years spent elbow-deep in the fume hood teach respect for the unpredictability in every batch, but also foster an appreciation for the skill that goes into each gram of pure product. Chemists don’t just follow recipes—they adapt and problem-solve. Each route gets refined over time, informed by old lab notebooks and noisy experiments.
Every compound is a jumping-off point. (R)-5-Bromo-3-(1-(2,6-Dichloro-3-Fluorophenyl)Ethoxy)Pyridin-2-Amine lends itself to further tweaks: you might see bromine exchanged via cross-coupling, or the amine swapped or protected to build bigger, more complex molecules. These modifications help tailor the base compound for specific biological or materials goals. A small change in the side group often changes potency, selectivity, or even simple properties like color and stability. In drug development, for example, chemists will test a family of analogs, sometimes watching side effects or potency swing wildly with a single atom shift. Over years, this process has separated blockbusters from dead ends. One thing I keep seeing: success comes from flexibility, a willingness to keep what works and fix what doesn’t.
Most compounds accumulate nicknames as they move through the research pipeline. Synonyms might reflect the order of discovery, the lab where it first showed promise, or even just a code name for prepublication secrecy. Although the IUPAC name sets the baseline, many researchers remember shorter monikers that speed up daily lab talk. Consistency matters—lost in translation between labs means wasted time or buy-in to the wrong compound. In my time, the lack of a standardized vocabulary led to real mistakes, reinforcing why we need rigorous naming at every step, especially for newer entries into the chemical world.
People outside the lab sometimes miss how real the risks are, especially with halogenated aromatics. Masks, gloves, and fume hoods aren’t decorations—they’re the armor. Toxic fumes, persistent residues, and the threat to both operator and environment compel tight controls and regular audits. I’ve stood in the shoes of those cleaning up forgotten spills and seen firsthand how chemistry corners cut today cause future headaches. It isn’t bureaucracy; it’s basic care for everyone’s health. Forward-thinking labs update standard operating procedures with every new batch, always learning from incidents and improving transparency. The push toward greener solvents and safer reaction conditions isn’t just jargon; it’s the future of responsible science.
So why put this effort into one molecule? This scaffold opens doors in medicinal chemistry, especially as researchers screen novel amines for kinase inhibition or anti-inflammatory roles. There’s a push to brave new ground with tough diseases that have shrugged off earlier drugs. In agrochemicals, similar compounds deliver targeted pest control, growing with precise impact and lower mass applied per acre. I remember projects targeting resistant weeds—a process made harder and more important by increasingly strict environmental rules. And then there’s materials science, where halogen-rich molecules play a part in specialized coatings, even electronic applications. Few discoveries are silver bullets, but the steady drip of new chemistry expands the toolkit to meet tomorrow’s challenges.
The R&D journey rarely looks like a straight line. Screening for activity starts in the test tube, with just enough of the compound to see if it binds or inhibits a protein target. Promising candidates move into animal models, dosage tweaks, metabolism studies. Every promising blip on a chart leads to more questions: does it cross the blood-brain barrier, cause off-target effects, show signs of bioaccumulation? Teams of chemists, biologists, and safety experts circle back, reworking the structure, improving yield, and searching for ways to manufacture it at scale. Years can slip by, and setbacks are common, but advances come from this relentless trial and error. I’ve learned to respect the grind—true discovery means sticking with it long after the novelty wears off.
People care about whether new chemicals are safe, and with good reason. Halogenated drugs can pack a punch but sometimes build up where you don’t want them. Early studies seek out mutagenicity, organ toxicity, and breakdown products, especially since some similar molecules stick around in tissues or in the environment far longer than intended. Regulatory agencies push for careful recordkeeping and tough tests—nobody wants another thalidomide story. I’ve noticed a healthy skepticism from toxicologists, guiding projects away from blind optimism. Risk assessment here isn’t just paperwork; it’s the line that stands between promise and regret.
No one can map out the entire future of new molecules, but I see momentum building behind compounds like this. As machine learning points out new targets and automates synthesis routes, the pace of discovery accelerates. Yet, the core value comes from patient, curious researchers and their willingness to learn from failure. Sustainability pressures nudge research toward greener production; the hope is to deliver powerful tools that pay dividends in health and nutrition without leaving toxic legacies. I’ve watched these trends merge—a new synthesis here, a safer process there—leading to innovation that respects both people and planet. It’s this blend of progress and responsibility where excitement meets accountability, and it’s what keeps science rolling forward, one compound at a time.
Walk into any pharmaceutical research lab today and odds are high you’ll spot new molecules designed to push medicine forward. (R)-5-Bromo-3-(1-(2,6-Dichloro-3-Fluorophenyl)Ethoxy)Pyridin-2-Amine, though quite a mouthful, turns up a lot lately among medicinal chemists. Its main claim to fame comes from its place in the development of targeted therapies—particularly kinase inhibitors. Those have grown crucial for cancer research and certain inflammatory diseases.
Cancer cells rely on rogue signaling pathways to outmaneuver the body’s own defenses. Kinase enzymes act like switches, sparking uncontrolled cell growth or survival. Many modern drugs try to shut down those switches. The chemical structure of compounds like (R)-5-Bromo-3-(1-(2,6-Dichloro-3-Fluorophenyl)Ethoxy)Pyridin-2-Amine lets researchers design molecules that fit exactly into the pocket of specific kinases, blocking their action.
The drive to improve selectivity and reduce side effects lights a fire under this kind of research. Many standard chemotherapies don’t discriminate between healthy and cancerous cells. Precision kinase inhibitors, on the other hand, promise a smarter hit by locking onto only particular job sites in cancer pathways. Molecules like this pyridine-based compound give scientists the starting point for creating these smart drugs.
I’ve spoken with chemists who’ve spent months reworking a molecule's shape—or even just rearranging a single atom—to dial in more powerful drug candidates. The chemical groupings in this compound offer both flexibility for modification and a backbone recognized for stability in biological systems. That means it serves as a lead structure scientists can tweak, creating analogs with improved activity or fewer side effects.
A lot of promising treatments begin their journey in small flasks, with massive attention paid to each atom. Small changes may create big leaps in stopping mutant kinases that drive lung, breast, or blood cancers. Sometimes, these molecules also unlock treatments for stubborn inflammatory diseases, where overactive kinases lead to tissue damage.
Recent literature points to compounds like this one in preclinical research pipelines. Scientific databases trace hundreds of kinase inhibitors from lab benches into advanced trials, highlighting the vital role of lead structures with pyridine or halogenated groups. By focusing on molecules able to block disease-driving proteins at ultra-low doses, researchers offer hope for patients facing resistance to older drugs.
Of course, cutting-edge chemistry raises the bar for careful stewardship. Potent new compounds deserve respect in both handling and discussion. Regulatory bodies expect rigorous proof before even the possibility of clinical use. Journals often flag recent advances long before the general public hears about them, but each step forward means safer, smarter therapies for tomorrow’s patients.
Looking past the string of chemical names, real progress relies on dedicated teams marrying imagination with discipline. From lab bench to bedside, the evolution of promising molecules like this one carries huge responsibility—and even bigger hope. Smart research and smarter oversight together build a pipeline for therapies that aim to strike disease at its roots, renewing the focus on better patient outcomes every step of the way.
Out in the real world, chemical purity isn’t just some technical term thrown around by scientists. It can define the difference between life and death, profit or loss, and clarity or confusion. Growing up with an uncle who ran a small soap-making business out of his garage, I saw firsthand how a misstep in the sourcing of sodium hydroxide affected an entire batch. One bag, labelled at 95% purity instead of the 99% he usually got, turned his soap from smooth and safe to flaky and harsh. That difference forced him to toss hundreds of bars into the trash.
The numbers next to “purity level” hold real weight. Laboratories set strict standards because even a tiny trace of contamination can mess with results. In pharmaceuticals, a single percent of impurity can make a drug unsafe or less effective. Food processing outfits will pay a premium for the cleanest lime juice concentrate, for example, because lesser grades can introduce off-flavors, or worse, unlisted allergens. After spending summers working quality control at a food plant, I remember how a lower-purity preservative triggered a recall because of unexpected by-products. It didn’t just waste money—trust, once broken, took years to repair.
The purity issue starts at the roots: how raw materials get handled, shipped, and processed. Dusty storage bins, leaky pipelines, or tired filtration systems will slip something unwanted into the mix. Elevation in purity doesn’t come accidentally, either. It follows skilled labor, investment in advanced equipment, and rigorous testing. X-ray diffraction, chromatography, and spectroscopic methods now dig even deeper, uncovering minute traces undetectable just a few decades ago. Regulation has stepped up too, as governments have learned the cost that comes with ignoring the problem. Take the 2008 incident where substandard heparin, laced with contaminants, made its way through global supply chains—lives were lost, confidence eroded, and scrutiny tightened overnight.
People tend to assume that purity is only for the white-coated folks huddled around Bunsen burners. That’s not the case. Take environmental clean-up. Removing toxins from contaminated soil or water calls for using agents with a high level of certainty—unexpected contaminants can react unpredictably and worsen an already tough situation. Even artists care: pigment purity decides how a painting will age and whether colors fade or stay vibrant. Anyone assembling electronics needs metals and plastics that won’t introduce faults or short circuits in the delicate components of a phone or satellite.
Transparency helps. Manufacturers do best to make purity test results available for each batch, not just rely on averages or historical numbers. Customers deserve to know, and oversight only strengthens trust in the market. Better supply chain practices, real-time tracking, and regular independent audits together form a solid defense. Routine staff training can keep workers up to speed on current risks and the best ways to handle materials.
There’s always room for pushing standards higher. Chemical purity is much more than a statistic. It’s the foundation for safety, reliability, and trust—on the lab bench, in the factory, and out in the real world.
I’ve seen a good number of labs treat chemical storage as a box-checking exercise. People line up fancy-looking vials, squint at labels, then slide them into a generic cabinet, and walk away. But (R)-5-Bromo-3-(1-(2,6-Dichloro-3-Fluorophenyl)Ethoxy)Pyridin-2-Amine, like many specialized research chemicals, deserves better. Its structure isn’t just a bunch of letters and hyphens—it translates to real consequences if storage goes sideways. Let’s dig into why.
Fluorinated, chlorinated, and brominated aromatics have a track record in the literature: they often display sensitivity to light, moisture, variable temperatures, and air. Let moisture creep in, and you’ll risk hydrolysis. Let sunlight hit the solid directly, certain bonds might break down, spoiling the purity and reliability that research hinges on. Imagine days’ worth of synthesis going up in smoke because sunlight snuck under a shelf—it’s not a minor risk.
Labs and companies pay a premium for these reagents based on expected reliability. Bring down purity by storing it at the wrong temperature, or in loose containers, and reproducibility goes out the window. These chemicals tend to come with significant price tags because of tricky synthesis steps. Trying to salvage degraded material often costs more in failed experiments than buying fresh stock. I learned early on that controlling your storage environment protects your research budget just as much as your samples.
No need for industrial-scale cold rooms if the workflow is small and contained. Most researchers get away with a dedicated fridge or a climate-stable cabinet. I always group light-sensitive aromatic amines into amber glass bottles—you won’t catch direct sunlight making a mess of next week’s scheduled tests. Screw caps with solid liners beat plastic snap-tops every time, keeping out atmospheric moisture and airborne junk.
If you’ve got a desiccator handy, toss in a silica gel pack with it. For those dealing with high-value samples in low volumes, vac-sealing them in foil bags before opening makes a real difference in shelf life. I’ve seen some labs liven up the space with tape flags blaring ‘No Light’ or ‘Dry Only’ reminders right at eye level. It sounds basic, but one distracted intern can turn a good month of work into a loss if warning signs get ignored.
Batch records, inventory logs, and digital tracking aren't just bureaucratic noise. They prove essential when someone six months later needs to revisit a result and faces a degraded bottle. It’s tough to trust science built on a shaky foundation. If you’re working under GLP, FDA, or similar oversight, proper documentation isn’t optional. Seeing a clear storage trail lets investigators and auditors follow the continuity of sample integrity—they often look for patterns in errors, and bad storage pops up more often than folks admit.
Too many labs rely on that ‘everyone knows’ sense to keep best practices alive. But if someone skips a step, or if a rule never made it to the new guy’s training list, that’s when trouble walks in. Open discussions, monthly training check-ins, and honest conversations around near-misses build a culture that doesn’t take chemical storage for granted. At the end of the day, safeguarding (R)-5-Bromo-3-(1-(2,6-Dichloro-3-Fluorophenyl)Ethoxy)Pyridin-2-Amine isn’t just about saving money—it’s about sticking to real science and making the most of every experiment.
Many people remember chemical formulas from high school, written in chalk on a blackboard—H2O, CO2, NaCl—shorthand for water, carbon dioxide, and salt. Chemicals may seem like the business of lab coats and glassware, but the truth is, formulas reach into every corner of daily life. Each formula tells us which elements come together, and how many of each, to form a particular substance. That information guides what we cook, clean, and even breathe.
Understanding chemical formulas brings clarity to what’s actually in products on the shelf or food on the table. For instance, folks allergic to gluten might want to know that the gluten protein’s makeup includes long chains of carbon, hydrogen, oxygen, and nitrogen. For someone with diabetes, every gram of glucose holds the formula C6H12O6—every carbon and oxygen atom matters. The right formula, clearly written, helps people avoid risk.
Many household cleaners list sodium hypochlorite as an active ingredient; its formula, NaOCl, lets chemists and consumers know exactly what’s inside. If you care about the environment, understanding chemical formulas becomes practical: it’s easier to spot ingredients from fossil fuels versus those more easily broken down by nature. Beyond fear, sometimes curiosity drives the need to look up a formula. Maybe you want to know why vinegar (acetic acid, CH3COOH) fizzes with baking soda (sodium bicarbonate, NaHCO3). That reaction stays memorable because formulas show what happens at the atomic level—the reason behind every bubble and fizz.
Molecular weight tells us how “heavy” a single molecule is, measured in units called daltons or atomic mass units. In the real world, whether cooking or creating medicines, knowing a molecule’s weight spells the difference between the right dose and a disaster. For medicine, molecular weight helps pharmacists determine how much active ingredient reaches the bloodstream. In food science, it affects how molecules blend and interact, allowing manufacturers to achieve the right texture, consistency, or flavor. Even athletes take interest: glucose’s molecular weight indicates how quickly the body absorbs sugar for quick energy in a marathon.
The chemical code isn’t always easy to crack for those outside science. Many product labels stay confusing because they use industry terms or list obscure additives. Schools could help by making chemistry more engaging—focusing on experiments relevant to daily habits, so students see formulas as tools, not chores. Public health campaigns should explain in plain language what certain substances do, and why the formula behind them matters. This invites better choices and sparks trust in science, not just from experts but from everyone who picks up a package at the store.
By demanding clear labeling and open information, communities become empowered. Apps and digital tools that scan product barcodes and translate formulas into everyday language already exist—heading in the right direction. Scientists must step out of technical silos, speaking in ways people understand. This builds bridges between research and the choices people make every day.
I remember the frustration of standing in a store aisle, balancing a single-use packet in one hand and a jumbo economy-size bag in the other. Some folks need only a little, others need a lot, but there’s never a perfect fit for everyone. This isn’t just about consumer preference. It touches on cost, convenience, and even sustainability.
Take food, for example. Families often run through a bulk supply of rice or detergent in a month, but if you live alone or in a dorm room, that oversized bag will probably go stale or harden before you get through half. So why force people to buy more than they want, or make them pay extra for tiny portions padded with unnecessary packaging?
Oversized packaging doesn’t just crowd your cupboard. It fills trash cans and landfills, and there’s evidence to show single-use packaging makes up a major share of household waste. The Environmental Protection Agency reported that containers and packaging made up about 28% of municipal solid waste in the United States in recent years. Small packages multiply the wrappers, bags, and boxes tossed away every day, and by the end of the week, every household has a sack full of what feels like just packaging.
There’s a flip side, though. In the health care industry, for example, smaller sterile packages can cut contamination risks. Precision matters where every dose counts. In this case, single-use certainly makes sense. In households and retail, though, most people push for options that match their pace.
The problems show up at checkout and in the kitchen or garage. Nobody wants to pay more per ounce simply because companies only sell something in small packages. At the same time, nobody wants to throw away expired products or watch money evaporate into stuff not being used. Buying in bulk saves money—if you use it in time.
People on tighter budgets get hit the hardest. Big packages usually have lower unit costs, but not everyone can afford a giant container upfront even if it saves money over time. Some folks can’t make room for big supplies, either, especially apartment dwellers or folks with mobility issues.
Choice solves a lot of these problems. Food co-ops and refill stores have popped up to let people buy as little or as much as they need. Even big brands have started offering alternatives, with recycled or compostable packaging options and multiple sizes that meet changing needs. I’ve seen grocery stores try “bring your own container” days—a step in the right direction.
Transparency helps. Letting customers know exactly how much product comes in each package, and the cost per unit, gives them the power to decide what’s right for their wallet and their conscience. Clarity should extend to recycled content, disposal instructions, and sustainability claims. Without clear info, shoppers can’t make confident choices.
It’s not enough to toss a couple of options on the shelf. Companies have a responsibility to meet today’s realities—smaller households, rising costs, and a planet struggling under piles of throwaway wrappers. Greater packaging options empower people, cut waste, and give a nod to individual circumstances. That’s something worth pushing for in every store aisle.