Chemists began taking a closer look at the family of salicylate compounds in the late 1800s in the search for less harsh alternatives to natural plant extracts and as part of a broad hunt for new medical and analytical aids. Potassium 4-methoxysalicylate traces its roots to this wave of organic chemistry exploration. Adding the methoxy group to salicylic acid’s aromatic ring produced fresh possibilities for synthesis, yielding new physical and chemical profiles compared to its parent compound. Potassium salts offer their own advantages for researchers, often improving solubility and safety when compared to other counterions.
Potassium 4-methoxysalicylate stands out among salicylates by blending a modest chemical backbone, inherited from salicylic acid, with a methoxy twist at the para position. Laboratories and industrial users favor this molecule not because it's the flashiest, but because it brings repeatable results. Ease of handling grows with the potassium salt; solubility matches up nicely for aqueous work and chemical reactivity stays resilient, feeding research spanning dyes, pharmaceuticals, and analytical reagents. Curiosity about this compound never really fades, especially in contexts where predictability and moderate reactivity go further than sheer raw performance.
Up close, potassium 4-methoxysalicylate forms as a crystalline solid. Its molecular nature reflects both the strength of ionic pairing and the recognizable aromatic ring that marks so many pharmaceutical intermediates. The white or off-white appearance, neutral smell, and broad compatibility with water all spring from the methoxy group's electron donation and the salicylate’s chelating tendencies. Experience in the lab has taught me to appreciate how potassium’s presence gives this compound stability and a safer profile than some sodium or ammonium salts. Stability under ambient conditions means that it survives typical shipping, storage, and handling. Standard melting points come in higher than many organics of similar size, something that matters for both practical use and technical design.
What matters to chemists staring at a bottle is what rides along with the labeled weight. High purity, usually above 98 percent by HPLC or titration, sends a clear message that the sample delivers what it promises. Regulatory and research settings both monitor for trace metal contamination, moisture, and pH, all crucial to avoid chemical surprises. The chemical label carries synonym names and precise mass, with batch-level traceability needed for serious research or regulated product manufacture. Correct CAS number assignment and clear hazard pictograms signal compliance and a real intent to put safety ahead of careless shortcuts.
Laboratory synthesis of potassium 4-methoxysalicylate starts with methylation of 4-hydroxysalicylic acid, converting the phenolic group ortho to carboxyl into a methoxy ether. Translating that into a potassium salt comes simply through neutralization with potassium hydroxide. In an industrial context, the process calls for consistent quality control and careful removal of byproducts. Choice of solvents and reaction temperatures shape the efficiency of each step, with excess base avoided to prevent degradation or discoloration. Attention to purification pays real-world dividends—crystallization from water or ethanol works well and helps prevent trace impurities from sticking around.
Chemically, the methoxy group offers both an invitation and a barrier. It shields the aromatic ring from electrophilic attack at the para position, altering potential downstream modifications compared to unsubstituted salicylates. Still, the free carboxyl and phenolic oxygens invite esterification, amidation, and coupling for custom synthesis needs. Chemists tap this core when pursuing derivatives for fluorescent dyes or pharmaceutical research. On paper, nucleophilic substitutions and metal chelation round out the list, but in practice, the gentle reactivity often makes this molecule a preferred stepping stone rather than an unstable end product.
Within international literature, potassium 4-methoxysalicylate appears under several names. Some call it potassium p-methoxysalicylate or 4-methoxy-2-hydroxybenzoic acid, potassium salt. This variation reflects differences in naming traditions—pharmaceutical versus industrial or academic versus commercial. Chemical suppliers often stick with IUPAC nomenclature for clarity, but as a user, it pays to check structures in addition to names to make sure there’s no mix-up. Overlooking synonyms can cause supply chain confusion or worse, a mismatch in regulatory filings.
Nobody in real-world chemistry shrugs off safety or leaves it to regulatory paperwork. Potassium 4-methoxysalicylate generally ranks as low hazard, but even supposedly gentle organic salts can irritate eyes, skin, or the respiratory system. I learned to wear gloves, goggles, and dust masks in settings where powdered forms get moved from bag to beaker. Safe handling means more than chemical labeling—it anchors to established exposure limits, spill cleanup protocols, and storage practices that keep oxidizers and incompatibles away. No reputable supplier today skips safety documentation or skips hazard training. Operational standards lean on protocols drawn from agency guidelines and real-world lessons, aiming for not just rule-following, but true risk reduction.
Demand for this compound comes mainly from analytical chemistry and niche areas of pharmaceutical research. In water analysis kits, potassium 4-methoxysalicylate has carved out a spot as a reliable reagent for some colorimetric reactions. Its chelation and moderate reactivity support both titrations and the preparation of stable indicator solutions. Pharmaceutical labs lean on it during early-stage route scouting for new active compounds—by offering a versatile aromatic scaffold, it helps speed up synthesis screening. Some specialty dyes in textile or diagnostic use lean on its decent solubility and gentle electron-donating effect, though these markets tend to fluctuate. I have never come across a mass-market consumer product listing this compound, but in the world of specialty chemicals, that's more a badge of precision than of obscurity.
R&D circles view potassium 4-methoxysalicylate less as a breakthrough and more as a building block. Research teams exploring new organic transformations appreciate the stability and solubility that this salt brings. It performs well in organic synthesis development, providing a starting point for fluorophore precursors or custom ligands. Years of scientific literature back up hundreds of derivative studies where minor tweaks to this base structure yield new biological probes or metal-organic frameworks. The adaptability of its functional groups, especially the methoxy and carboxyl arms, means that chemists rarely set it aside as too risky or too limited. Exploring new catalytic cycles and dye chemistries often brings this salt’s underlying chemistry to the surface, highlighting its value even after decades on the scene.
Common sense and regulatory checks both demand a clear view of toxicity before greenlighting regular use of any new or standard reagent. For potassium 4-methoxysalicylate, available animal toxicity data suggest low acute oral and dermal toxicity, similar in scope to potassium benzoate or related salicylates. Repeated exposure testing shows little evidence of chronic harm at typical laboratory concentrations, but sustained high doses or aerosols can kick up respiratory or mucous membrane irritation. No one should treat it as food-grade or suitable for human consumption, and the requirement to avoid groundwater contamination remains in force as with all potassium or organic salts. Researchers continue to monitor both acute and chronic outcomes, especially before scaling up to pilot plant levels or exposing populations beyond laboratory walls.
Looking forward, potassium 4-methoxysalicylate’s story isn’t about sudden disruptive innovation, but stable incremental progress. Synthetic chemists come back to it as a trusted intermediate where performance and stability still matter. Improvements in green synthesis—especially cleaner methods for both methylation and salt formation—could cut down on byproduct waste, a win for both economics and environmental impact. Analytical kit makers eye more sensitive test designs, relying on this salt for consistent standard performance. Pharmaceutical and materials science researchers will likely discover subtle advantages in custom-tailored derivatives off this base, especially as interest grows in sustainable chemistry and better diagnostics. Safe handling, rigorous testing, and regulatory transparency will keep its profile solid and dependable. Because sometimes in chemistry—as in life—what endures is what matters most.
Looking at the name Potassium 4-Methoxysalicylate, most folks without a chemistry degree might just keep walking. Tucked behind that tongue-twister, though, you’ll find a compound making quiet but real contributions, mostly in the fields of medicine and chemical analysis. Over years spent working with health-related reporting and the regular habit of reading research journals, I’ve seen how compounds like this one play their part out of the spotlight.
Potassium 4-Methoxysalicylate stands out in laboratories, particularly for its role in chemical analysis. Scientists value it as a reagent, a substance used to trigger reactions so something invisible becomes measurable. In my experience covering research labs, this compound helps researchers identify trace metals or elements in samples, whether for water quality or pharmaceutical production. Like a good referee, it helps keep things fair and clear by allowing only the needed chemical reactions to show up in a test.
Its chemistry gives it the right shape and properties to bind with metal ions. In doing so, it changes color under certain circumstances. Color changes aren’t just for show—scientists lean on them to measure the concentration of metals without the need for expensive machines every time. For laboratories running heavy metal screenings or those keeping tabs on purity levels, having reliable reagents like this one slashes both costs and turnaround time.
Compounds similar to Potassium 4-Methoxysalicylate show up in the story of aspirin’s development and the ongoing hunt for drugs that fight pain and inflammation. While this specific potassium derivative rarely tops the starring roles, salicylate compounds form the backbone for many over-the-counter drugs lining pharmacy shelves. I remember years ago, diving into the origins of painkillers and being surprised just how much modern medicine borrows from simple chemical tweaks like the methoxy group added to salicylic acid.
Within pharmaceuticals, sometimes doctors and researchers eye less familiar derivatives to see if they promise better results or fewer side effects. It’s a reminder of how medical chemistry always keeps evolving—trying, testing, discarding, or elevating new molecules based on what research and patient outcomes show. Every new twist on an old compound starts with a handful of fundamentals, and Potassium 4-Methoxysalicylate fits into this ongoing experiment.
Turning attention to environmental testing, this chemical helps labs spot trouble in water supplies. That’s important if you live downstream of any industry or agricultural operation. Safe water isn’t something to take for granted. Having dependable tools for rapid, field-friendly testing remains essential in my book. Potassium 4-Methoxysalicylate, by helping reveal trace contaminants, gives environmental watchdogs another way to keep pollution from slipping through the cracks.
Working around any reactive chemical means balancing effectiveness with safety. Researchers need to keep track of each new additive’s health and environmental risks. The story of Potassium 4-Methoxysalicylate reads like many specialized chemicals: useful, but always due for more research. Old procedures get new scrutiny, guidelines change, and regulatory bodies like the FDA or EPA step in as more gets learned.
From where I sit, it matters that we never treat these compounds as throwaway details in bigger processes. Each one has ripple effects—good and bad—across science, medicine, and environmental health. More transparency and ongoing studies help everyone, including people outside the chemistry world. Every time another research team shares findings about the safety or effectiveness of compounds like Potassium 4-Methoxysalicylate, it’s a step toward safer labs, cleaner water, and better drugs.
There’s nothing glamorous about lab safety. Gloves and goggles don’t make headlines. Yet every chemist, student, and hobbyist who works with Potassium 4-Methoxysalicylate learns pretty quickly that you can’t get far without paying attention to the basics. Getting lax with your approach to a compound like this only ends one way—trouble. I’ve dealt with enough chemical samples to understand how one split-second mistake can sideline a whole project. So here’s what matters, from an honest, no-nonsense perspective.
Ask anyone who has gotten a chemical splash near their face what stands out more: the sting or the regret. I’ve never seen anyone work with Potassium 4-Methoxysalicylate—used sometimes in analytical chemistry or as a fine chemical—who skips gloves or safety glasses twice. Nitrile gloves keep skin from absorbing dust or solutions, and safety glasses or goggles stop careless eye rubs from escalating into an emergency. A lab coat becomes your best friend because spills do happen, no matter how careful you think you are.
Good ventilation means you control your working environment, instead of letting circumstances control you. It’s easy to underestimate how quickly fine powders like this can become airborne when pouring, weighing, or mixing. I’ve watched minor carelessness turn a corner of a bench into a cleanup headache. I always advise using a fume hood because breathing in any dust is playing roulette with your lungs. At a minimum, work upwind of your materials in a well-aired space. Many colleagues have found that a local exhaust fan pays for itself the first time something goes wrong.
Potassium 4-Methoxysalicylate sits comfortably in the realm of organic salts but that doesn’t make it harmless. Just like any chemical, it reacts in unexpected ways with the wrong solvents or under excessive heat. I learned early to double-check compatibility before dissolving anything. Heat sources get monitored closely. If you aren’t sure, don’t mix it in with acids or strong oxidizers just to see what happens—those stories never end well. Reading up on known incompatibilities means no guessing games with your health or workspace.
Every lab accident I’ve heard about started small—a drop left on the counter, a mislabeled container, someone distracted by a phone. Label everything clearly. Clean as you go, instead of saving it for later. Store Potassium 4-Methoxysalicylate in tightly closed containers, somewhere cool and dry, away from other reactive substances. I keep all powders and solutions locked away if there’s even a chance of curious visitors, especially in shared spaces.
Too many people ignore this crucial step until it’s too late. Have spill kits, eye wash stations, and emergency showers within reach. Learn how to use them without fumbling around. Most chemical exposures don’t become serious injuries, unless people hesitate or panic. I always remind my team that running water washes away most chemical splashes if you move fast.
Potassium 4-Methoxysalicylate demands respect, not fear. Solid habits, common sense, and a willingness to slow down and think—these do more to protect you than any fancy piece of equipment. I trust simple routines over luck any day, and I encourage everyone working with chemicals to do the same. Safety isn’t a formality; it’s the reason you get to keep working another day.
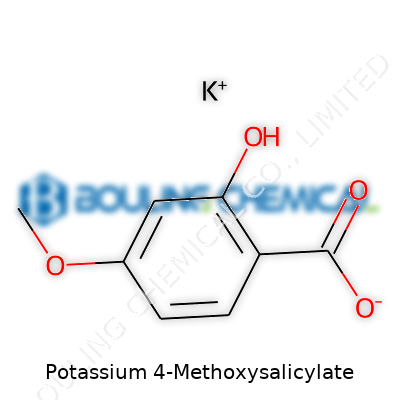
Mention Potassium 4-Methoxysalicylate, and the conversation quickly turns to its chemical formula: C8H6KO4. The structure starts with a salicylic acid base, swaps the carboxylic hydrogen for a potassium ion, and adds a methoxy group at the fourth carbon. This little tweak opens up new properties that regular salicylates just don’t have. The shift from acid to salt form also means a jump in water solubility. In pharmaceutical and lab circles, that feature isn’t just a bonus—it’s a requirement for consistent results. C8H6KO4 might not roll off the tongue, but knowing that formula matters: it shapes the compound’s behavior, usefulness, and impact.
For anyone who remembers hours spent over titration beakers or chemistry homework, that change from hydrogen to potassium isn’t just chemical trivia. In practical terms, potassium salts tend to dissolve easily and help active molecules reach their target faster, whether in research or in industrial use. That matters for labs aiming to get rapid, predictable results each time they run a sequence. Not every molecule likes the bloodstream or a reaction flask. Tucking a methoxy group at the para position (the “4” in 4-methoxysalicylate) affects how the compound reacts with enzymes, interacts with other molecules, and even its shelf life. These details become the difference between a compound that disappears unnoticed and one that turns heads in the lab.
Potassium 4-Methoxysalicylate isn’t just a sum of its atoms. Its stability, ease of handling, and compatibility with water-based systems give it a leg up for some formulations. In the real world, that means less struggle with dissolving powders, less guesswork re-creating results, and fewer surprises in the test tube. Folks developing new treatments want compounds that not only do the job, but do it every time.
Costs enter the conversation too. Sometimes, small changes in a molecular structure translate to fewer steps during synthesis. That’s not an abstract advantage. In the pharmaceutical world, fewer steps mean lower bills, and that can trickle down all the way to clinics in need of reliable supplies.
Chemists put their trust in a formula, and rightfully so. C8H6KO4 has set boundaries—reactivity, melting point, storage needs—that have been mapped out. But getting it wrong isn’t a small slip. Labs need to know exactly what’s in each bottle. Precision in naming and formula guards against mix-ups that could upend experiments or, worse, patient safety. The purity, stability, and batch reliability of potassium salts deserve close attention from researchers, manufacturers, and regulators alike.
Seeking new uses for potassium 4-methoxysalicylate should run alongside deeper safety checks and transparency in the supply chain. Open communication on purity testing and trace impurity levels can put a dent in unexpected setbacks. Ongoing collaboration between the research community and regulatory bodies ensures that compounds like this one stay both useful and safe, not just on paper but in the field.
Getting the formula right means more than a correct answer on an exam. It sets the stage for trust, predictability, and progress in everything from medicine to manufacturing.
People often overlook the simple things when handling chemicals: the way bottles get stacked, the type of container, or how long something sits out once opened. From years of experience in lab environments, ignoring the basics with a compound like Potassium 4-Methoxysalicylate is asking for trouble and can cause headaches for both safety and research integrity.
Storing Potassium 4-Methoxysalicylate gets straightforward once you remember its sensitive nature. This compound has a habit of breaking down if left in humid conditions or under direct sunlight. It holds up best in a well-sealed bottle, kept out of reach from moisture and light.
Drawers, cabinets, even fridges meant for chemicals—these spots do a solid job as storage spaces. Molded plastic vials or glass containers offer dependable protection. Tossing a desiccant packet inside the bottle works wonders at soaking up stray moisture.
Left sitting out at room temperature, especially on a muggy day, you start noticing clumping, discoloration, or even changes in texture. Potassium salts in general don't like soaking up water from the air. Wet or partially degraded material means ruined experiments and inaccurate measurements.
Ideal storage ranges from 2 to 8 degrees Celsius. Science shows a drop in temperature slows chemical changes and keeps unwanted reactions in check. In one published case, leaving bottles at 25°C versus 4°C meant double the loss of quality in a matter of weeks. Adding this step does not feel redundant when you want results to match published data.
Writing the date of receipt and last use straight on the bottle makes a difference. Nobody remembers when that bottle was opened, and no one wants to risk using a spoiled ingredient during an important project. In busy spaces, recycled containers or fading labels create confusion and wasted money.
Clear, updated labels with hazard warnings, lot numbers, and dates keep the workplace organized. Responsible chemical use comes down to easy habits, built from early training or advice from colleagues.
Not everyone handles chemicals in state-of-the-art labs. Sometimes supplies end up at schools, research clubs, or even home workshops. There have been news stories about accidental poisoning or spills simply because bottles got tossed in a junk drawer or left near food supplies.
Storing Potassium 4-Methoxysalicylate high up, away from kids and pets, inside a locked cabinet or box, shrinks these risks. Local regulations often require hazardous materials to stay locked up and properly labeled. It may sound excessive, but quick mistakes often have lasting consequences.
Labs toss out kilograms of solid chemicals each year, largely due to poor storage. Once a material takes in moisture or oxidizes, it’s finished. Upgrading storage spaces pays back in saved cost and time. Simple changes—such as investing in humidity meters, automatic lighting timers, and better shelving—make a clear difference.
Treating Potassium 4-Methoxysalicylate like it matters builds trust in the results. Solid planning up front saves headaches down the line, in science and in safety.
Ask anyone who has spent time in a lab, and you’ll hear the same story: chemicals don’t wait around for you. Open a bottle, and the countdown begins. Potassium 4-methoxysalicylate isn’t rare on chemical shelves, especially among researchers exploring pharmaceuticals or cosmetics. Yet, many overlook just how much shelf life shapes safety and research reliability.
I remember finding an old jar of potassium 4-methoxysalicylate in our shared chemistry storeroom, label faded, sachet half empty. We debated whether it still worked—an argument that took up more time than the quick experiment I wanted to run. This is a reality in labs, small and large.
Official shelf life can differ based on how tightly a container is sealed, the room’s temperature, and how often the chemical sees the open air. For potassium 4-methoxysalicylate, manufacturers often suggest storing it in cool, dry spaces with tightly-closed lids. Moisture and light start chipping away at its stability. Most sources agree on an average shelf life of two to five years in unopened packs stored under ideal conditions—usually 2–8°C and away from direct sunlight. Opened containers, especially in humid places, might not stick to this timeline.
Push past the window, and you risk more than a failed experiment. Potassium 4-methoxysalicylate may lose potency or react differently. In worst cases, old material forms clumps or gives inconsistent results, especially when purity matters for something like a skin treatment study. As a grad student hunting for reproducible results, I learned to trust my nose—and the calendar. A chemical that looks clumpy or smells off doesn’t belong in a test tube.
Ignoring shelf life isn’t just about wasting money. Unstable chemicals might harm users or ruin months of research. With more organizations tightening up safety expectations, expired reagents create compliance headaches. Auditors still talk about the time someone tried to explain away a ten-year-old bottle of organic acid.
Potassium 4-methoxysalicylate works as a mild preservative and has roles in analytical chemistry and cosmetics. Like its cousins, it can absorb water from the air and breaks down if left exposed. Water eventually hydrolyzes the compound, lowering quality. Simple analytical checks—melting point, spectral analysis, and pH studies—tell a clear story when things go wrong.
The U.S. Pharmacopeia and other chemical regulatory bodies encourage strict control of reagent age. Labs must keep logs. My experience matches this: regular tracking cuts down on disasters.
One quick fix: write the opening date on every bottle, and review the chemical cupboard twice a year. Rotate stock to use up older materials first, just like at the grocery store. If there’s any doubt, find the chemical’s certificate of analysis and double-check the manufacturer’s recommendations.
For bigger outfits, inventory tracking tools help spot slow movers and highlight what’s nearing expiration. Staff need clear guidelines on spotting tell-tale signs of age—yellowing powder, strange clumps, or awkward crystallization.
Ignoring shelf life risks safety, research outcomes, and regulatory headaches. Real vigilance comes from everyday habits: clear labeling, mindful storage, and regular checks. It’s about learning, sharing, and keeping the work honest.