People love to talk about innovation, but real breakthroughs rarely come on purpose. Polyfluoroethylene, better known as PTFE or by the household name Teflon, is the poster child for unexpected discoveries. In 1938, a chemist trying to make something for refrigeration stashed away cylinders of tetrafluoroethylene, only to crack them open and find a slippery white powder building up inside. What seemed like another day gone wrong ended up transforming entire industries. Shortly after, PTFE’s weird slipperiness and resistance to just about everything drew the attention of engineers, cookware designers, and space agencies. World War II put PTFE to work in the Manhattan Project, and its initial secrecy only added to the mystique. Over the decades, companies found ways to make it in larger amounts, drop the price, and dream up new uses, from frying pans to heart grafts.
PTFE seems almost boring at first glance—a white waxy solid, often pressed into sheets, rods, or coatings. It doesn’t corrode, barely reacts to strong acids or bases, and shrugs off sunlight and solvents. People use it in places where metal rusts, rubber degrades, or less hardy plastics melt down. Its best-known trick remains its nonstick surface, which made cooking eggs possible with little more than a pan and a nudge. But the story stretches far beyond kitchens. Chemists count on it for labware. Electricians wrap it around wires inside jet engines, where heat and friction would tear up anything else.
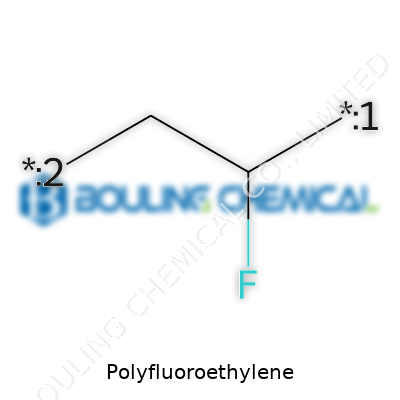
What sets PTFE apart comes down to its unique structure. Each carbon atom in the backbone grabs hold of two fluorine atoms. This makes a shield, blocking almost anything from breaking through. PTFE stands up to temperatures up to 260 degrees Celsius. It doesn’t dissolve in the usual solvents like acetone or alcohol, and even strong acids glance off its surface. Water, oil, and most sticky things roll right off. Its coefficient of friction is one of the lowest for any solid material, so you find it in places where moving parts need to slide smoothly without wearing down or sticking.
PTFE goes through quality checks for melting point, tensile strength, and purity. The melting point hovers around 327 degrees Celsius. Engineers tend to care about specific gravity, tensile strength, and elongation at break, since these numbers let them predict how the material will behave under load. PTFE isn’t the easiest plastic to mold, so it’s sold in the form of sheets, rods, tapes, and finely divided powders. Labels reference its batch, grade, and, for some uses, compliance with standards from groups like ASTM or FDA—there’s no single one-size-fits-all standard. Cost can get high for medical or food-grade types, especially when extra steps for traceability and purity factor in.
Manufacturing PTFE starts with tetrafluoroethylene gas. Chemists spark off a polymerization reaction, linking the molecules together into long chains inside pressurized reactors. This step isn’t for amateurs, since the gas is explosive under the wrong conditions. Once polymerized, the material looks like a chalky white powder. From there, it’s compressed and sintered, almost like how people make ceramics, to form usable shapes. Cutting and machining PTFE creates shavings and dust, which need collecting to avoid waste or contamination. Some companies have developed techniques to add fillers like glass fiber or graphite, boosting mechanical strength or tweaking properties for special jobs.
PTFE comes from regular chemistry but resists almost all chemical changes after synthesis. Standard acids and bases slide off harmlessly. At high heat, PTFE eventually breaks down, releasing nasty fumes containing fluorinated compounds. Modifying PTFE usually happens on its surface. People bombard it with radiation, treat it with alkali metals, or graft other groups onto it to create a bond where glues or inks will stick. The vast bulk, though, stays inert. Chemists have also spun off related plastics—FEP, PFA—by tweaking the recipe to make the material easier to process or more transparent.
Most people know PTFE as Teflon, a DuPont trademark. Chemists might call it polytetrafluoroethylene, and technicians abbreviate it as PTFE. Dozens of companies produce it under their own names—Fluon, Hostaflon, Polyflon—but the recipe stays basically the same. In catalogues, sometimes you see other trade marks, but they all point back to the same slippery polymer at the core.
Anyone working with PTFE runs into safety conversations. The powder version creates a risk for lung exposure, especially in processing plants. Finished PTFE itself doesn't pose much health risk at room temperature, but once it gets heated above 260 degrees Celsius—like in overheated nonstick pans—tiny fumes can come off and cause polymer fume fever, sometimes called “Teflon flu.” Birds turn out to be extremely sensitive, so pet owners learned the hard way. Industry keeps to guidelines about working temperatures, ventilation, and dust handling to keep things safe, and worker protections follow from long experience with plastic processing. Codes from groups like OSHA and NIOSH provide the regulatory backbone.
PTFE quietly keeps the world moving. In chemical plants, pipelines and valves rely on PTFE linings to fend off acids and bases that would destroy metal. Food processing equipment needs clean, nonstick surfaces that resist bacteria and aggressive cleaning chemicals. Wiring in airplanes and computers depends on PTFE’s stable insulation. Surgeons turn to grafts, surgical patches, and catheters made with PTFE because they don’t rot or trigger immune responses. Lubricant sprays based on PTFE reduce wear in gears and locks, even in salty environments where other greases attract dirt. Each use has its own story—sometimes replacing expensive metals, sometimes letting less skilled operators handle equipment with fewer errors.
PTFE keeps scientists and engineers up at night, not because of what it already does, but because of what remains possible. In labs, the focus shifts toward new versions that perform better at extreme temperatures, last longer under stress, or take on novel functions. Composite materials, mixing PTFE with nanomaterials or specialized fibers, open doors for aerospace or electronics. R&D teams explore eco-friendlier manufacturing, searching for ways to recover or reuse PTFE scrap, given its near-indestructible life in landfills. Medical researchers want to fine-tune biocompatibility, hoping to create implants that last decades without body rejection. Electronics manufacturers look for coatings that both insulate and disperse heat, especially as devices shrink and power densities rise. Patents suggest the pace won’t slow down soon.
Scrutiny grows sharper every year around chemical safety, and PTFE can’t dodge the spotlight. As a finished product, PTFE remains stable at low-to-moderate temperatures. Problems come into play with the breakdown products, either during high-temperature mishaps or from the manufacturing process itself. Perfluorooctanoic acid (PFOA) once helped make PTFE but lingered in groundwater and bodies, raising alarm about cancer risk and immune system effects. Research continues, and regulatory authorities in the US and Europe have cracked down on related compounds in production. The industry has moved to replacements, but watchdogs track health studies closely. Workers in factories face the highest risks from dust or fumes, making ventilation and safety gear a must. For consumers, normal use at cooking temperatures stays well below the danger threshold, with incidents linked mostly to overheating or misuse.
PTFE stands out among plastics because it already earned a permanent spot in a ridiculous range of technologies. Looking forward, the issue isn’t simple growth—it’ll involve dealing with environmental baggage. Recycling PTFE poses one of the hardest problems, since it just doesn’t break down naturally. Collapsed landfills and ocean microplastics have people demanding answers, sparking research into chemical recycling, melting down and reusing the polymer, or finding biodegradable cousins. On the performance side, new blends and nanostructured coatings promise better properties for electric vehicle batteries, solar panels, and medical diagnostics. Governments and consumers expect more from manufacturers in the way of transparency, stewardship, and safety. The next breakthrough probably won’t look like slicker frying pans, but rather cleaner manufacturing and smart recycling, pushing the industry to not just celebrate what PTFE does, but how it fits into a changing world.
Polyfluoroethylene doesn’t roll off the tongue, and most folks recognize it by its better-known brand name: Teflon. In kitchens, that simple non-stick coating makes a huge difference. Morning eggs glide off the surface, pancakes don’t cling, and cleanup takes a few quick swipes. Someone with messy kids at home — or anyone tired of chipping food off old pans — quickly learns to appreciate the reliability of this coating.
Yet, the use of polyfluoroethylene stretches far beyond frying pans and baking sheets. That slick surface comes from a marvel of chemistry, with carbon-fluorine bonds resisting heat and chemical reactions. Factories and laboratories depend on gaskets, seals, and tubing that won’t corrode or stick. Electricians rely on high-insulation wire coatings that don’t break down in rough environments. Medical devices and even some implanted materials call on this plastic because bacteria and bodily fluids won’t cling to it easily.
No doubt, the toughness of polyfluoroethylene helped reduce waste. Non-stick cookware often lasts longer. Machines in chemical plants stay running with fewer leaks and fewer breaks. Workers avoid exposure to harsh chemicals that once splashed through weaker seals. In my own kitchen and garage, damaged old tools and cookware used to pile up regularly. Switching to Teflon-lined pans and PTFE-taped plumbing definitely stalled the steady march to the trash can.
There’s a flip side: that same strength and resistance makes polyfluoroethylene persist in landfills and elsewhere in the environment. These plastics won’t simply disappear. Investigations in the past decade have raised concerns about the chemicals used in making non-stick coatings, especially as some linked compounds — known as “forever chemicals” — stick around in groundwater and even the human body. In some places, research on alternatives gained real momentum as the public learned more about these issues.
People ask if there’s a safe middle ground. I believe real progress starts in the kitchen, workshop, and lab. Using quality over quantity, not tossing pans at the first scratch, and keeping coatings out of contact with high flames or metal tools keeps Teflon from degrading too fast. Manufacturers have begun swapping out the most harmful chemical steps. Some restaurants have already changed their cleaning and cooking methods, limiting chipping and overheating of non-stick wares.
Recycling offers another avenue, yet these plastics are tough to break down and reuse. A few tech companies have started researching ways to recycle polyfluoroethylene by grinding and processing it at high temperatures, hoping to turn old coatings and gaskets into new products without polluting the air or water. Switching to alternatives — like ceramics or stainless steel in certain applications — also helps, particularly where the extreme resistance of polyfluoroethylene surpasses what’s actually needed.
Not every tool or gadget needs the highest-tech plastic out there, but polyfluoroethylene gives people more freedom to try, fail, and try again without worrying about every scratch or spill. We need to learn both from its strengths and its missteps, finding responsible ways to use and eventually replace coatings and parts that stick around long after they’re needed. For now, the story of polyfluoroethylene is about choices: making the best of its usefulness, while staying honest about the challenges it leaves behind.
Many folks hear “polyfluoroethylene” and “Teflon” thrown around in conversations about cookware or chemical safety. Sometimes, it gets confusing, especially when marketers label things with fancy names. I can tell you after years of writing about science in everyday life—Teflon and polyfluoroethylene are basically two sides of the same modern story, although the details matter.
Polyfluoroethylene is usually shorthand for polytetrafluoroethylene, which most people see as the plastic that keeps your eggs from sticking to the frying pan. Teflon, on the other hand, is the trademarked name that DuPont gave to their own version of PTFE (polytetrafluoroethylene) back in the 1940s. Over time, “Teflon” became the word folks use for nonstick cookware, coatings on wires, and even waterproof clothing. The stuff itself—a slippery, tough, heat-resistant polymer—stays the same, but who makes it, how pure it is, and what additives go into it can vary a lot.
Sometimes the difference between a brand name and a chemical name means nothing to most people, just like a tissue being called a Kleenex. Still, these labels get confusing when safety is on the line. Headlines about “forever chemicals,” which sometimes include forms and byproducts of PTFE, make it even more important to untangle what goes into our kitchens and lives.
Here’s the thing—to earn the trusted Teflon name, DuPont (now Chemours) had to follow strict manufacturing rules. Not every pan labeled “PTFE-coated” is held to the same standard. Bad coatings might chip, degrade, or let loose more particles at higher temperatures. A chipped or overheated nonstick surface can release fumes, and research has linked these to polymer fume fever in rare workplace accidents. Keeping pans below 500°F and discarding scratched ones heads off most issues.
PTFE by itself does not break down into dangerous stuff under normal cooking, and it’s considered stable and non-toxic in everyday kitchen use. The worries aren’t about PTFE but about chemicals used to make it—specifically, older generations of perfluoroalkyl substances (PFAS) like PFOA. Companies have phased out the worst offenders, but PFAS pollution, bioaccumulation, and water safety remain top concerns. Scientists have found these substances in drinking water and wildlife, which has sparked community health debates and new laws in several states.
From my own experience talking with chemists and public health experts, transparency comes out on top. Shoppers want to know if their cookware is safe, and regulators want fewer dangerous chemicals in soil and water. Accurate labeling—saying “PTFE” and “Teflon” only when they mean what people expect—is a good first step. Meanwhile, companies investing in safer manufacturing and clearer communication can help restore public trust.
Nonstick pans do make life easier, but folks shouldn’t have to play chemist just to fry an egg safely. Keeping pans away from metal utensils and high heat can head off most trouble. Proven alternatives—ceramic, cast iron, stainless steel—cut down on chemical worries for those who prefer old-school cooking. But for a well-made Teflon-lined pan, decent care still brings years of trouble-free breakfasts.
In the end, clear knowledge always beats marketing gloss. Whether you call it polyfluoroethylene, PTFE, or Teflon, it pays to look for real information, not just the brand stamped on the handle.
In daily life, you don’t notice how much depends on plastics until one rips or melts away before the job is done. A certain class of plastics—fluoropolymers—steps up where most common plastics give out. Polyfluoroethylene, which many folks recognize through its well-known brand Teflon, brings a reputation earned by its stubborn resistance to nearly everything thrown at it. This isn’t just a fluke. It takes some wild chemistry for a plastic to survive strong acids, boiling water, and the abrasion of years of scraping.
Most plastics gain their properties from carbon chains bound by hydrogen. Polyfluoroethylene flips that script. Each hydrogen trades out for a fluorine atom. Fluorine really grips onto carbon, forming a bond that most chemicals fail to bust open. Chemists talk about this as one of the tightest bonds found in synthetic polymers. Because of this, polyfluoroethylene doesn’t play along with reactions that chew up other plastics. Acids like nitric or sulfuric acid, which normally chew holes in standard plastics, don't get much of a foothold here. Even in a splash of lye or a bath of harsh solvents, the fluorine shell shrugs it off.
This chemical stubbornness leads to its famous temperature resistance. Kitchens depend on nonstick pans that can take high heat, but in industry, it’s more than breakfast eggs—pumps, hoses, and valves face hot acids, aggressive solvents, and caustic brines, and only polyfluoroethylene reliably stands in their way. It pushes back against melting or softening, too, only giving way at temperatures higher than what most cooktops reach.
Polyfluoroethylene’s predictability saves countless hours and prevents equipment failures. Factories avoid downtime, labs push the limits of chemical research, and surgeons trust coatings on syringes and guidewires. In my own experience rebuilding a leaky water filter that handled aggressive cleaning solutions, the solution was always the same: swap out the tired standard seals for polyfluoroethylene. Only then would my repairs actually last. The material simply laughs off what would destroy lesser plastics.
With these impressive properties come some challenges. Bonding polyfluoroethylene to other materials frustrates even experienced engineers—the low surface energy that makes it so slippery also thwarts adhesives and coatings. Its extreme stability means it sticks around in the environment for a long time, raising reasonable environmental concerns. As more research documents traces of fluorinated chemicals in groundwater and food sources, the chemical’s durability turns from a benefit into a debate about how much long-lasting material society actually needs to introduce.
One path forward involves recycling and reusing polyfluoroethylene instead of single-use discards. Supporting research into biodegradable alternatives can help, but for now, no substitute matches the suite of chemical properties this polymer brings. In applications where nothing else can last or keep people safe, polyfluoroethylene remains irreplaceable. More responsible use seems like the sensible path. Save this plastic for jobs where its strengths matter, invest in better handling at the end of its life, and keep pushing for chemistries that work as well—without the baggage.
A lot of folks know polyfluoroethylene by its brand name, Teflon. Cookware with a nonstick surface has changed how many people make breakfast. Pancakes slip from pans, eggs crisp at the edges without extra oil, and cleanup feels like magic. Yet, questions about the safety of this wonder coating keep bubbling up. Is it really a safe bet when it touches the food we eat?
Chemists developed polyfluoroethylene in the 1930s. They marveled at how little anything would stick to it or break it down. By the late 20th century, it had become a staple in kitchens and factories. The FDA approved its use for food contact, but that doesn’t always end the story.
Studies show that pure polyfluoroethylene is stable under most cooking conditions. It doesn’t react with water, oils, or the acids you find in tomato sauce. The material’s carbon-fluorine bond ranks among the strongest in chemistry. Food doesn’t pick up bits of Teflon, and researchers have struggled to detect any leaching of the polymer itself under typical cooking temperatures.
Anyone who overheated a nonstick pan knows the smell that fills the kitchen. Once polyfluoroethylene reaches temperatures above about 260°C (500°F), it can start to break down. That’s when toxic fumes show up. These fumes have sent pet birds and even people to the doctor with headaches and chills. In the real world, most home cooks don’t crank burners high enough for this to happen. Yet, stories are common of scratched pans and scorched surfaces.
Beyond the polymer, attention has shifted to chemicals used in the manufacturing process. People may have heard of PFOA, which stands for perfluorooctanoic acid. Though separate from polyfluoroethylene itself, PFOA used to be part of making nonstick pans. Studies tied PFOA to potential cancer risks and other health effects. Manufacturers have phased it out in North America and Europe, but questions remain about what new substitutes could do.
Regulators keep a watchful eye. The U.S. FDA lists certain grades of polyfluoroethylene as safe for direct food contact. European regulators hold similar positions. Still, modern eaters deal with a landscape where trust feels hard to earn and easy to lose. People want clear answers about what lines their pans, bakes their cookies, or touches their kids’ lunch.
Scratched nonstick pans should head for the recycling pile. Avoiding high heat keeps the risk of fumes low. Leaving birds out of the kitchen helps, too—a lesson some learn too late. Stainless steel and cast iron pans offer a solid alternative and skip the chemical debate. Yet, for many, the ease of nonstick cooking keeps polyfluoroethylene in the cupboard.
Keeping count of how many years a pan lasts and swapping it out once the coating starts to peel is one way to err on the side of caution. Manufacturers release new coatings without key chemicals like PFOA, but questions about long-term safety don’t disappear overnight. Strong science makes a difference, but open communication and honest labeling might matter even more for peace of mind.
Any facility worker or lab tech who has handled polyfluoroethylene knows it’s one of those high-performance plastics that fills a rare gap—remarkably stable, tough under pressure, trusted anywhere extreme chemical resistance is a daily demand. For decades, labs, manufacturers, and even home tinkerers have been putting it to work because it tolerates harsh surroundings where most plastics call it quits.
Handling this stuff comes with real responsibilities. One slip-up—improper storage, a contaminated container, ratty gloves—and everything from gadget reliability to worker health takes a hit. Taking shortcuts with how you keep polyfluoroethylene around might not cause chaos immediately, but over time you’ll see equipment issues, process slowdowns, or even health complaints cropping up. That’s not theoretical: anyone who’s worked for longer than a season in a manufacturing setting knows small lapses get expensive fast.
Keep polyfluoroethylene cool and dry. Leave it near heat or in direct sun for extended periods, and you’ll risk the plastic breaking down in ways you can’t undo—something you might not notice until that critical valve or gasket fails months later. Humid conditions cause materials around it, especially paper or cardboard packaging, to degrade. Mold and mildew follow, and now there’s more than just plastic failure to worry about. I’ve seen more than one storage room get musty and suddenly a batch from last quarter is fit only for the trash.
What about dust and contamination? Those who cut corners on packaging quality or let inventory sit open might end up with dust or oils on the surface. That creates issues for both processing and applications, but sometimes you only catch it after a critical product test fails. It’s far easier to keep everything covered, sealed, and off the floor than to spend the next morning scrubbing everything before shipping out an order.
Every job should focus on safety, but polyfluoroethylene isn’t just another plastic pellet or film. At high temperatures, it can release fumes you’d rather avoid—fumes proven to cause flu-like symptoms and, after high exposure, much worse problems. Ask anyone who forgot about adequate ventilation or skipped a mask during a maintenance run. Respirators, basic gloves, and eye protection matter, especially if the job involves any cutting, welding, or thermal work.
Label every container clearly and keep materials separated from solvents, acids, and especially from equipment that gets hot during the workday. In shared workspaces, confusion leads to mistakes. Keeping training up-to-date means less guesswork in the storeroom and fewer emergency calls after hours.
Facilities and workshops don’t need high-tech gear to avoid trouble. Use covered bins, maintain basic ventilation, and keep stock organized so nothing gets misplaced and potentially forgotten. Track shelf life by rotating inventory. Store bulk shipments off the ground and out of sunlight. Hold regular training—not just one-and-done sessions. Tie good habits to the company’s bottom line: fewer repairs, lower replacement costs, happier staff.
Every workplace has its quirks, but respecting the basics—clean, cool, dry, well-managed stockrooms—pays off in productivity and safety. Polyfluoroethylene isn’t magic, but if you treat it right, it’s a long-lasting tool in your back pocket. The best shops I’ve seen don’t rely on luck; they build routines and keep everyone accountable from stocking shelves to tearing down production lines.