Perfluoroketone’s story runs parallel to the deepening search for safer and more effective extinguishing agents, especially since the old standbys—halons and CFCs—faced bans because of their ozone depletion risks. In the late 20th century, as global environmental regulations tightened, researchers started poking around the perfluorinated chemical family. What sets this family apart is a backbone built with robust carbon-fluorine bonds, creating a class of substances with excellent chemical stability and resistance to degradation. Discoveries in the 1990s brought perfluoroketones into focus, especially for niche uses. Fire suppression teams and electronics manufacturers looked at these compounds as answers to problems the world didn’t want to ignore: fire safety in sensitive places and environmentally viable cooling fluids.
Perfluoroketone shows up in places with high standards and little room for error. Data centers, heavy industry, and aircraft routinely tap into this chemical's abilities. You’ll most often see it in fire extinguishing systems that protect electronics, because water or old-school carbon dioxide just can't handle the job without causing more harm. This chemical finds a strong audience in those who value clean agent firefighting with minimal cleanup. Word among professionals travels fast when a product works as promised. The same chemical toughness that interests fire safety folks benefits cooling and dielectric applications. People who buy perfluoroketone usually do so with clear intentions, and the market for it reflects a shift in attitude toward environmental responsibility and performance.
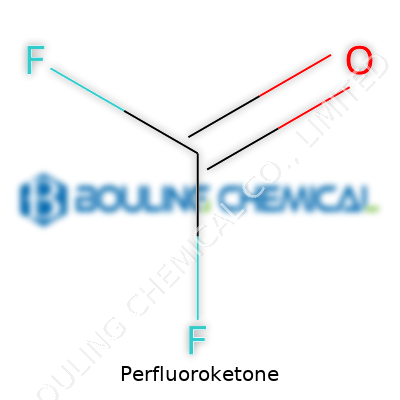
Perfluoroketone stands out for what it doesn’t do as much as what it does. This clear, odorless liquid boils at a low temperature under normal air pressure, and its vapor lingers heavier than air, creating a smooth blanket effect over electrical fires. The famed carbon-fluorine bonds give it high resistance to heat, acids, and light, so it won’t break down under demanding conditions. Unlike halons, it doesn't feed ozone-depleting cycles or readily react with most everyday materials. In my experience working with electronics, the non-conductivity of perfluoroketone offers a sense of calm during high-stakes troubleshooting, especially around sensitive circuit boards.
You won’t find much poetry on chemical drum labels—regulators want clarity, not marketing. Perfluoroketone jars and drums usually display the chemical formula and hazard warnings. Labels mark the product with a CAS number and include GHS pictograms for quick risk assessment. Manufacturers must report concentration and purity, and professionals check data sheets for boiling point, density, and recommended storage. Smaller quantities for laboratories arrive in specialized containers to reduce leakage or accidental exposure. From my own work, those clear, detailed specifications help workers keep risk in check, especially if chemical compatibility plays a role in equipment or container choices.
The synthesis of perfluoroketone starts with a traditional ketone core, then pushes the molecule through a rainstorm of fluorination reactions. Electrochemical fluorination or direct fluorination replaces the classic hydrogens with fluorine, resulting in a transformation that ramps up chemical toughness. Yields run highest when the process happens under strict temperature and pressure controls, using specialized reactors designed to withstand corrosion. Technicians handling the process keep a close watch for byproducts, since incomplete fluorination can introduce impurities. In my own industry interactions, careful monitoring during synthesis makes all the difference between a safe, reliable chemical and one that causes headaches downstream in application.
Perfluoroketone rarely goes looking for trouble in a reaction flask. Its molecules carry little desire to take part in typical organic reactions, thanks to that fortress of fluorine atoms. Efforts to tweak or break apart the molecule demand pretty extreme conditions, such as high energy UV or strong alkali metals, but most users want the structure to stay intact anyway. Some chemists have explored partial substitution or mixing with other perfluorinated compounds to tune volatility or introduce a specific functionality, but most commercial uses stick to the tried-and-true versions due to consistent performance and well-documented safety profiles. For those in research labs, pushing the limits always calls for extra vigilance, since manipulating these stable molecules can steer into unknown territory quickly.
Many in the field call perfluoroketone by shorthand, referencing either its chemical structure—like C5-ketone or dodecafluoro-2-pentanone—or brand names that have entered everyday conversation among specialists. The fire suppression field names products after their application or molecular backbone to distinguish between options. Some call it by the numbers, referencing the length and position of fluorinated chains, but the essential signature stays the same: a perfluorinated molecule with a ketone group sitting at its core.
The rules around working with perfluoroketone reflect the chemical’s unique benefits and potential risks. Its low acute toxicity to humans gives some peace of mind, but inhalation of vapor at high concentrations can still cause discomfort. Industrial settings adopt engineering controls, proper ventilation, and personal protective equipment to reduce the chance of exposure during filling and application. Certified fire suppression systems integrate automatic controls to cut off discharge if room oxygen falls too far, ensuring life safety for workers or first responders. Globally, organizations such as the NFPA and the EPA press for systems that minimize both worker exposure and environmental leakage. In operations I’ve witnessed, training and regular drills keep incidents rare. Waste handling follows guidance that prevents the release of persistent organofluorines into water or soil, as regulatory scrutiny now focuses keenly on these compounds due to PFAS-related concerns.
Perfluoroketone found a foothold in spaces with more on the line than just property. Banks, hospitals, data storage centers, and telecommunication sites invest in fire suppression systems using this molecule because they want to avoid both fire and water damage. Mission-critical applications demand quick knockdown of flames without harming their high-value electronics or risking hazardous residue. Some specialized cooling and dielectric fluids rely on perfluoroketone too, as it delivers thermal management where air or water wouldn’t stand a chance. For teams building electric vehicles or running high-voltage testing setups, the chemical’s nonconductive and stable nature offers an edge. While not exactly everyday stuff, you know perfluoroketone’s present where stakes and consequences run high.
Science continues to dig into perfluoroketone, driven by both opportunity and concern. Teams in academia and industry investigate ways to dial in specific properties, such as adjusting volatility or tweaking the boiling range for harsh or unique environments. Upcoming research projects include methods for recovery and recycling, cutting costs, and reducing waste. Environmental groups have pressed for better lifecycle assessments, who want more data on long-term environmental persistence and alternative breakdown pathways. New analytical tools and detection techniques surface as laboratories strive to track incredibly low concentrations of the compound in air or water. My own run-ins with R&D teams showed a split personality: part optimism for new breakthroughs, part caution about the unknowns that come with persistent chemistry.
Health and environmental toxicity take center stage in the current debate over perfluorinated substances. So far, perfluoroketone ranked low in mammalian acute toxicity compared to its peers, showing minimal effects at common workplace exposure levels. The compound clears from the body faster than some of the notorious long-chain PFAS chemicals, offering some reassurance. scientists keep turning over rocks to identify chronic exposure effects, subtle breakdown products, and the potential for bioaccumulation. Studies in aquatic environments and soil ecosystems still run behind the curve, as the field chases new analytical targets. One lesson from the PFAS controversy: lagging research can turn into big regulatory or public relations headaches if environmental persistence goes unchecked.
The road ahead looks complicated for perfluoroketone. Its promise in performance, particularly for fire safety and advanced cooling, won’t fade soon with major industry investments relying on tried-and-true chemical characteristics. Still, the public conversation about PFAS keeps evolving. Lawmakers around the world look for the next shoe to drop as analysts gather more data and environmental activism pushes hard. More investment will likely go into developing "greener" analogues, creating recycling methods, and tighter regulations on emissions and disposal. Engineers want materials that do their job without leaving a legacy of persistent pollution. Users in critical industries feel the pressure from all directions—reliability, safety, regulatory certainty, and environmental peaks all matter. As research continues, perfluoroketone will need to answer more questions about its full-life impact, pushing the community to innovate or adapt with better stewardship and alternative technologies.
Pulling back the curtain on chemical names, perfluoroketone rarely makes the headlines, yet it keeps plenty of things running safely, often out of sight. I first came across this compound through a friend working in fire suppression for data centers. Her team kept servers cool for hospitals—packets of private patient data, vital imaging records—by using perfluoroketone-based systems instead of traditional water sprinklers. No one wants to fight fire but fry a cluster of servers in the process.
Perfluoroketone shines in specialized fire suppression. In big server farms, sensitive manufacturing plants or high-voltage environments, standard firefighting options don’t cut it. Water damages electronics. Gasses like halons harm the ozone layer and can leave behind a mess or health risks. Perfluoroketone offers a strong answer. It extinguishes fires by absorbing heat and breaking the chain reaction that feeds flames—without flooding the room, and importantly, with minimal impact on the environment or on people.
This molecule shows up in the branded solutions filling fancy glass-walled server bunkers. Reports from industry groups show the plain truth: as organizations ramp up their digital infrastructure, they look for ways to manage fire risk without endangering investments or violating environmental rules. Perfluoroketone steps up: it has a low global warming potential compared to old-school chemicals like hydrofluorocarbons, and doesn’t linger in the environment. Test results after system discharges reveal almost nothing left behind—air levels fall back to normal quickly, so spaces can be occupied soon after use.
Some groups have pressed for tighter regulation on chemical suppressors—prompted partly by what happened with older halon products. Decades back, too much reliance on high-warming, ozone-damaging fire suppression led to phased bans. Today, perfluoroketone doesn’t face those same criticisms, and safety reviews from labs and environmental watchdogs back up its claims. The American Society of Heating, Refrigerating and Air-Conditioning Engineers has even highlighted its safety attributes, especially where human health and quick re-occupancy matter.
Challenges sit on the supply and disposal side. Like most fluorinated compounds, careful handling is a must. Any chemical can cause trouble in the wrong context. Waste disposal processes should keep residues out of groundwater, and production lines ought to follow strict emission controls. It’s fair to say manufacturers still need to meet higher transparency and disposal standards as demand rises. Environmental agencies watch closely, and oversight cannot slack—past mistakes with persistent chemicals offer a clear warning.
For those of us focused on long-term solutions, the story raises ongoing questions about balance. We need fire safety solutions that don’t trade one risk for another. Perfluoroketone looks like a step in the right direction today, but only as long as manufacturers, users, and regulators share responsibility—rigorous monitoring, honest reporting, and updated practices as science advances. More sustainable answers may still emerge. The best way forward is to dig deep, double-check impacts, and support innovation that keeps safety and health up front.
Every year, companies hunt for safer ways to put out electrical fires. The search led to chemicals like perfluoroketone, branded as a greener alternative for fire suppression in data centers and power stations. At first glance, the pitch is impressive. Perfluoroketone breaks down quickly in the atmosphere, doesn’t form persistent greenhouse gases, and has a low global warming potential compared to older firefighting agents. This has pushed it into the spotlight as data centers multiply and regulations on greenhouse gases get tighter.
Perfluoroketone doesn't stay in the air for decades like some older chemicals. Instead, it turns into harmless byproducts within days when exposed to sunlight. Studies put its atmospheric lifetime at under a week. That’s good news compared to chemicals like perfluorocarbons, which linger for thousands of years and trap heat with frightening efficiency.
Companies argue that because perfluoroketone doesn't accumulate, it sidesteps some of the worst problems linked to climate change. The numbers mostly back that up. Direct emissions appear far below regulatory thresholds for most industrial users. Plus, it doesn’t chip away at the ozone layer.
Even though the stats look promising, relying only on them feels short-sighted. Experience has taught me that just because something breaks down quickly in the open air, it doesn’t guarantee zero risk. Fire suppression systems sometimes dump large amounts of chemicals in a hurry. If that chemical makes its way into storm drains or groundwater, there’s not much sunlight to break it down. Small mistakes or spills build up over decades, even if each one seems minor on its own.
Perfluoroketone sits in the family of fluorinated chemicals known for resisting natural processes. Past issues with cousins like PFOS and PFOA have shown how these substances tend to stubbornly stick around in water and soil, impacting rabbits, fish, and even people living nearby. Data shows perfluoroketone doesn’t bioaccumulate as much as those earlier chemicals, but traces still show up in lab results. It makes me wonder if the excitement for newer, 'greener' chemicals sometimes outpaces careful research.
Regulatory agencies don’t give out free passes. They wait for full toxicological data before making sweeping rule changes. That approach makes sense. Chemical companies and users should monitor for unexpected byproducts in local water and soil. Even trace effects on wildlife and people deserve close attention.
One thing that brings peace of mind: using less chemical in the first place. Regular system checks and targeted release zones help avoid the sort of full-room dumps that send gallons of firefighting agents into the environment. Investing in modern leak detection and investing in less toxic alternatives where possible bring down reliance on chemicals that might have hidden costs.
We saw with refrigerants and firefighting foams how slow-moving damage can surprise everyone, even folks who thought they did due diligence. Jumping to say something is 'safe' based on a handful of metrics won’t cut it. Real safety keeps an eye on both lab stats and how things behave in the real world, over years, not weeks.
Companies can do more than point at chemical breakdown stats. Publishing ongoing monitoring reports, investing in environmental research, and staying honest when unknowns pop up helps earn trust. People have a right to know what’s in their water, air, and soil. These steps build confidence—not sales pitches or half-told stories.
For years, the word “fire suppressant” would trigger images of foam, water, or thick chemical clouds billowing out of fixed pipes. Growing up, I’d see fire extinguishers in school hallways and never really think about the chaos they’d actually create if ever discharged indoors. The cleanup would be a nightmare. But in places where electronics rule—think data centers, museum archives, or advanced medical labs—the cost of that mess can dwarf the damage caused by the initial fire itself. That’s where perfluoroketone (often called by its chemical shorthand, FK-5-1-12) shows up.
Instead of smothering a flame with foam or choking off oxygen, this stuff absorbs heat directly at a molecular level. Picture a chemical that moves into a room, soaks up the heat, and drops the temperature of a blazing fire in seconds, fast enough to stop combustion cold. Unlike old-school halon systems, there’s no need to worry about toxic byproducts filling the space or sticking around long after the danger passes. That matters in any place where people might still be inside or where valuable equipment can't just be hauled out and replaced.
A few years back, I visited a local university's computing center after they’d upgraded to a perfluoroketone system. It looked just like any chilled office room until a demo: a mock fire flared, and with a hiss, FK-5-1-12 was released. No big plume, no residue, just air that felt a bit cooler, and equipment untouched. Students working there kept typing, barely glancing up. It's one thing to read about “clean agent” technology; seeing it work in real time turned me into a believer.
Traditional agents like halon fell out of favor for good reason—they wreck the ozone layer and stick around in the environment. Perfluoroketone doesn’t linger in the atmosphere or accumulate in living things. Its atmospheric lifetime measures in days, not decades, compared to some of the old chemical fire suppressants. This makes a real difference in communities trying to shrink their environmental footprint while keeping data or rare art safe.
Water-based suppression works for warehouses and homes, but let water loose on racks of servers, and repairs skyrocket. Perfluoroketone leaves no puddle, no sticky residue, and doesn’t short out electronics. Spraying it won’t destroy archival documents or ancient paintings either. That appeals to anyone entrusted with preserving irreplaceable records and equipment.
There are costs, literally. FK-5-1-12 systems can run pricey, and refilling after discharges means ongoing expense. But in places where an hour of downtime means millions lost, or data loss can’t be undone, the outlay often earns its keep. Cleaner composition also means less regulatory headache compared to legacy halons.
No single fire suppressant works everywhere, and perfluoroketone won’t replace water for your kitchen fire anytime soon. But in the zones that power our digital lives, clean agents deserve a closer look. Proper fire risk assessments, robust system designs, and training go further than just picking the right agent—it's about adapting to new threats and using tools that fit our needs without wrecking the world we live in.
Perfluoroketones probably don't show up in everyday conversations, but these chemicals have quietly found their way into many parts of high-tech life. Used as coolants in electronics and clean agent fire suppressants, they offer performance nobody argues with. Seeing these substances pop up in more systems and workplaces, the conversation about long-term effects on health just can't be brushed aside.
My initial curiosity hit when a friend mentioned headaches and breathing discomfort after repeated contact with electronics cooling fluids. I started reading and found the environmental persistence of fluorinated chemicals had folks in chemistry and medical circles worried. Unlike garden-variety chemicals, these compounds linger in air and water for a long time before breaking down—if ever.
So, where does trouble show up for people? First, no one has a perfect map of every health consequence. Still, decades observing relatives who worked with advanced cooling systems in industrial settings made me cautious. Exposure happens through inhaling fine mists, accidental skin contact, or even touching contaminated surfaces. Short-term exposure might lead to irritation in the eyes, nose, or throat. A few studies reported headaches or coughing, much like my friend’s complaints.
The bigger picture starts to look more serious as exposure increases. Research suggests that high concentrations could affect liver function, impact kidney health, or even mess with cholesterol levels—similar to older cousins like PFOA and PFOS. These legacy chemicals have well-documented histories: connections to cancer, immune system problems, even changes in hormone levels in exposed workers. While perfluoroketones aren’t as widespread as those, the lack of long-term human data doesn’t translate to safety.
Most folks in the electronics or firefighting industry don't get warnings about possible low-level chronic exposure. These days, health isn’t just about avoiding acute poisoning. It’s about recognizing that even a practically invisible vapor can set someone up for health trouble years later. Knowing the way these molecules cling to surfaces and resist being washed away raises real questions about secondary exposure at home or in water supplies.
Families living near disposal sites or manufacturing plants have a right to be concerned. When you see reports linking fluorinated compound pollution to thyroid disease or reproductive issues in wildlife, it’s not hard to draw connections and wonder if the next headline will involve neighbors down the road.
Change won't come just by blaming users or workers. Companies need to place more focus on routine monitoring of air quality and strict control of chemical leaks. Workers should get clear guidelines―not just a hazard warning tucked inside a manual. In my experience, honest conversations about what’s being used on-site make the biggest difference in building trust.
Doctors and nurses in occupational clinics should get training about the early symptoms of fluorinated chemical exposure. Real progress would mean industry leaders start putting more dollars toward finding safer alternatives, and regulatory bodies setting exposure thresholds that actually reflect today’s realities, not old assumptions.
Perfluoroketones aren’t some abstract danger from a chemistry textbook. They’re already here, in tools we rely on every day. It makes sense to open our eyes, demand transparency, and push for health protections before stories like my friend’s become the norm.
Getting to know the way chemicals behave isn’t just a science project — it keeps people, the workplace, and the environment safer. Perfluoroketone stands out in labs and data centers, especially as a firefighting agent and cooling liquid. Its handling often gets pushed aside because people assume it’s less risky than legacy alternatives. That’s not the full picture.
Years back, halons got the boot over ozone and climate troubles. Perfluoroketone rolled in as an option for demanding fire suppression jobs. Unlike old-school halons or even hydrofluorocarbons, this stuff breaks down in the lower atmosphere. Environmental impact drops, and so does risk of long-term persistence. That earns it points but can trick folks into treating it like water or mineral oil. That’s a shortcut to trouble.
Most perfluoroketone on the market sticks to being a low-boiling, non-flammable, colorless liquid. In reality, it can evaporate quickly at room temperature. This calls for containers with tightly sealing lids, resistant to chemical corrosion. Metal alloys that stand up to fluorinated compounds beat out plastics that can degrade or react; glass isn’t always practical, but stainless steel works.
Temperature swings matter, since heat will boost evaporation. Dry, cool storage spots cut loss and keep unwanted vapor from building up in closed spaces. Everywhere I’ve worked, we’ve kept stocks away from sunlight and ignition sources, just to be sure. Vapor isn’t explosive, but large releases in closed rooms can displace oxygen and complicate air quality.
Perfluoroketone has a low toxicity profile compared to a pile of legacy fire suppressants, but that doesn’t mean handling can be sloppy. Leaks create slippery patches, raising injury risk, especially in data centers or mechanical rooms. I’ve seen a minor spill turn into a scene from a slapstick movie—people sliding or falling—because staff didn’t have the right absorbents handy or didn’t know the liquid would spread this far, this fast.
Personal protective gear matters, too. Splash goggles, impervious gloves, and long sleeves will prevent cold burns or skin irritation. Good ventilation rules the day, especially if someone is pouring or transferring product. Most facilities have local fume extraction; that extra attention means workers aren’t left breathing solvent vapor. Quick cleanup tools—absorbent granules, chemical-resistant mop kits—belong right next to storage racks.
Some operators risk dumping old perfluoroketone with regular trash or washing it down drains. That’s reckless; local regulations in most places block that outright, and rightfully so. Licensed chemical disposal outfits convert waste into benign substances, or carefully incinerate it. I’ve learned that clear labeling and strict usage logs cut down waste, let teams spot leaks fast, and keep containers from piling up unused until someone gets careless.
I’ve noticed training gets more earnest as companies grow serious about long-term environmental and health outcomes. Regular drills help spot slip-ups or improve the layout of chemical storage zones. Sharing these hard-earned lessons at safety meetings keeps mistakes from repeating.
Safe handling and storage of perfluoroketone depends on consistent attention to detail. This isn’t paranoia—it’s common sense shaped by past accidents and a drive to keep both people and the planet healthier. A little extra care up front always beats regret on the other end.