Nitrogen trifluoride has roots that stretch back to the days when people first explored new fluorinated compounds for industrial uses in the mid-20th century. Scientists originally wanted to find safer and more efficient etchants for the booming electronics field, especially as silicon chips started shaping an entire generation’s idea of technology. In the hunt for alternatives to harsher and less manageable fluorine sources, nitrogen trifluoride entered the picture, offering benefits that seemed to overshadow early concerns. By the time the 1990s rolled in, semiconductor manufacturers began treating this compound like a staple ingredient in chip fabrication, etching, and chamber cleaning.
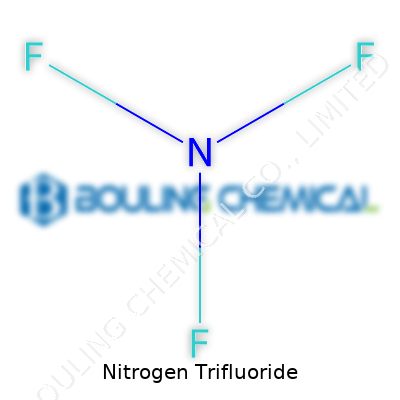
Most folks outside fabrication plants will never encounter nitrogen trifluoride up close, but its impact kicks in during every smartphone upgrade and solar panel install. Looking like a colorless gas, nitrogen trifluoride shows up packed in sturdy cylinders, ready for strict industrial processes. Chemically, it’s made from one nitrogen and three fluorine atoms, tightly bonded for notable stability under normal conditions, though it packs a punch during high-energy reactions. The stuff’s reactivity sparks to life in heated plasmas—the kind that help strip away unwanted material from wafers or clean complex machinery parts without leaving much behind.
Just as with any specialty material, fine technical points decide where nitrogen trifluoride fits. Its boiling point at a chilly -129.1°C keeps it as a compressed gas in normal handling, and its dense vapor ensures good delivery in closed-loop production setups. It’s heavier than air and comes with Tag numbers and chemical labels that workers learn to spot quickly. Factories track everything from purity to residual moisture, making sure even slight contaminants never spoil a production run. This control matters because even minor mishaps with nitrogen trifluoride can trigger hazardous breakdowns, so regulators step in and set firm exposure guidelines.
Preparation usually starts with the fluorination of ammonia or other nitrogen compounds, using tough chemical processes under tightly controlled conditions. Some methods rely on electric discharges to persuade otherwise stubborn precursors to come together, producing nitrogen trifluoride as a gaseous product. Technicians then pass the raw material through purification lines, scrubbing out by-products and water vapor—since both can cause dangerous side reactions or equipment corrosion down the road. In research, people constantly tinker with reaction conditions to boost yield and limit carbon or hydrogen-containing waste gases.
When engineers switch on plasma tools filled with nitrogen trifluoride, the real show starts. The high-energy environment breaks apart each molecule, letting those wild fluorine atoms rip away at surface debris, silicon oxides, or tough contaminant films. In chemical vapor deposition and etching, this approach allows remarkable precision, shaping nanometer-sized patterns that define modern electronics. Scientists sometimes modify nitrogen trifluoride with other gases in search of even more selective, efficient, or less polluting reactions—always balancing performance with practical safety.
Nitrogen trifluoride carries more than just a chemical badge—workers in the field recognize it by synonyms like Trifluoroamine or NF3, marked right on supply tanks or shipment forms. There’s not much guesswork in handling it, but paperwork demands exact labeling, especially with increasing international trade and safety rules. Some regions place it under greenhouse gas monitoring or strict reporting requirements, given its environmental profile.
For anyone handling nitrogen trifluoride, the biggest challenge never comes from daily operations but from the rare cases of leaks, unplanned releases, or improper disposal. The gas itself proves nearly odorless and hard to spot with the senses. If it gets loose in enclosed places, it can push out air, raising suffocation risks. Even more alarming, accidental exposures to plasma or open flames set off toxic breakdowns, releasing fluoric acid or noxious oxides. Strict protocols shape every phase of its handling—automatic leak detection, positive-pressure suits, and round-the-clock training keep injury rates low, but there’s always pressure on operators to keep skills fresh.
Nitrogen trifluoride mostly stays behind the scenes, but devices powered by semiconductor wafers, flat-panel displays, and high-performance solar cells wouldn’t take shape without it. Manufacturers rely on this fluorinated compound for cleaning tools with a sharp edge but gentle touch, stripping away waste without damaging what matters. The stuff’s unmatched ability to clear out stubborn films supports faster production, lowers tool downtime, and raises average yields—all pieces that help keep costs from spiraling. Its role spills over into specialty optics, research-grade lasers, and even some energetic rocket propellant work, though chipmaking still draws the biggest share.
Research labs across the globe dig into making safer or greener alternatives, largely because nitrogen trifluoride possesses a troublingly high global warming potential—over 17,000 times that of carbon dioxide by weight over a century, according to the Intergovernmental Panel on Climate Change. Teams explore less persistent cleaning agents, recapture technology, and process tweaks that cut down on emissions at the plant. Progress doesn’t always move as fast as the demand for cleaner chips and safer factories, so a lot rides on bridging the gap between laboratory findings and factory floor reality. Some researchers push for plasma techniques that demand less NF3 input or for reactive recycling units that reprocess leaked gas before it escapes into the atmosphere.
Animal studies—and a few rare incidents among industrial staff—have shown that inhaling or being exposed to nitrogen trifluoride’s by-products can cause severe respiratory distress, burns, or even chronic lung ailments. Regulatory bodies keep close eyes on air monitoring data, especially in high-volume countries. Nitrogen trifluoride, due to its environmental persistence, also draws attention as a long-lived greenhouse gas that slips through cracks in international agreements unless strictly capped. This pressure sparks calls for more transparency and real-time emissions auditing, with advocates pointing out that small leaks accumulate over millions of annual wafer runs.
Nitrogen trifluoride’s place in the chemical landscape hangs in the balance as people weigh its unique technical benefits against a growing stack of regulatory, environmental, and workplace health challenges. The high-tech world has made a commitment to keep supply safe and emissions low, but governments and advocacy groups are likely to keep pushing for safer substitutes and tighter controls. Growth in clean energy, electric transport, and next-gen computing only expands the playing field—keeping the chemistry labs busy and forcing manufacturers to rethink decades-old habits. In my own experience covering cleanroom upgrades and process audits, companies often see investments in green abatement as a win for PR as much as for compliance. Nitrogen trifluoride may hold its spot for years, but every major player knows that what gets etched today shapes the rules—and risks—of tomorrow’s tech economy.
Fluorine in a tank always spells caution to folks who handle chemicals for a living. Bring in nitrogen trifluoride—NF3—and the air starts humming with a different kind of energy. NF3 isn’t famous in homes or even at most workplaces. Out in the world of high-tech gadgetry, though, NF3 plays a role that many would call indispensable. Flat-screen TVs, computer chips, and solar panels often owe a nod to this clear, colorless gas, even if consumers can’t taste, see, or smell its presence.
If you’ve ever peered into the bright face of a television or smartphone, you’ve indirectly witnessed the work of nitrogen trifluoride. Semiconductor fabrication eats up a big chunk of NF3 every year. The gas acts like a meticulous housekeeper, scrubbing away leftover material from complex chip-making equipment. Instead of opening up machines and cleaning them by hand, workers let the gas flow in so it can clean on a molecular level. This keeps the machines running smoothly and the chips they produce free of defects.
Large panel displays for televisions and smartphones use plasma etching to shape electronic circuits. NF3 punches above its weight in these operations, creating reactive agents that break down stubborn deposits. When I worked with designers in the electronics field, many argued this process made possible the improved precision and shrinking scales that keep smartphones thin but powerful.
Solar tech looks greener at first glance, but making high-purity silicon for solar panels involves steps that don’t exactly scream “eco-friendly.” Factories rely on the molecular cleaning power of NF3 to keep furnaces and chambers spotless, so impurities don’t sneak into finished wafers. The irony never failed to strike me—an industrial chemical helps fashion a renewable energy future, even as it earns its own environmental warnings.
The conversation around nitrogen trifluoride swings from awe to alarm. NF3 isn’t harmless for the climate. As a greenhouse gas, its ability to trap heat beats even carbon dioxide by thousands of times over a century. The EPA flagged this problem because rising levels in the atmosphere match the growth in electronics production.
In my experience talking with engineers, they lose sleep over gas leaks or venting because industry regulations can lag behind technology. NF3 lasts a long time aloft, so even small releases can matter. Tracking and containing every molecule becomes both a moral issue and a technical challenge.
Solutions always start with honest math and cleaner alternatives. Some companies invest in abatement systems that destroy leftover gas before it escapes into the air. Other researchers hunt for replacement chemicals with less global warming potential or invent processes that need fewer chemicals for cleaning. I’ve heard some critics claim these fixes cost too much, but heavy investments now may buy a safer future. The stakes run high—the screens and panels lighting up our world shouldn’t darken prospects for the planet down the line.
People sometimes treat chemicals like ordinary tools, but nitrogen trifluoride carries real risk. Once you’ve seen what an aggressive oxidizer can do, caution stops feeling like overkill. Working around fluorine compounds, I learned fast that even the smallest mistake could have nasty consequences—nose and lung irritation, coughing fits, sometimes symptoms that keep you home for days. A release in a poorly ventilated area turns fast from a minor leak to something you can’t ignore: breathing trouble sets in, warnings start to feel personal, and panic rises if people aren’t trained or prepared.
Regular gloves or basic eye protection fall short with gases like this. Vapor can slip past lightweight barriers and latch onto moisture: skin, eyes, the lining of your lungs. The pain lingers. Chemical-resistant gloves—think nitrile, not latex—and safety goggles with full-face shields become non-negotiable. I’ve seen co-workers grab the right equipment only after stories of hospital visits floated through the breakroom. It shouldn’t take a close call to make safety feel real.
A good lab coat won’t stop a high concentration, but it buys precious seconds. Aprons made for corrosives, strong enough to resist rotting, make all the difference. A good pair of shoes, no exposed ankles—those details suddenly matter when spills splash, even if they’re rare.
Some shops rush to buy the right gear and stop there. Gases build up, more so than you’d think, in blind corners and low spots. If you can smell anything sharp or odd in the air after opening a cylinder, it’s time to admit the hood or local exhaust runs too low. I always push for ventilation that goes beyond bare minimum codes, asking for fresh, working airflow—not just a decorative fan. People should not count on instinct when the consequences include throat burning or worse.
Nobody starts out memorizing what to do if a valve sticks open or a tube slips off the fitting. Refresher courses turn emergency plans into muscle memory. Real stories get shared—one time a click-fit broke loose, another when someone failed to double-check connections. People remember to recognize the faintest whiff and to read the warning labels every time. Getting in the habit of storing every gas cylinder securely, chained up tight, upright, with all valves sealed, that routine should feel as automatic as locking your front door.
Don’t store cylinders near acids, oils, or anything that reacts with strong oxidizers. If a cylinder gets warm to the touch in storage or during transit, stop and check. Leaks won’t wait for a convenient time. Emergency showers, eyewash stations, and fast evacuation plans work only if they are kept close, not buried across the building.
Nothing replaces a culture that encourages people to speak up. If someone points out a loose connection or asks for extra checks, listen. Risk shrinks in places where people look out for each other and double down on the details. Protect your lungs, your skin, and the folks who work beside you. Nobody is too experienced to overlook one small slip.
Nitrogen trifluoride holds a special place in the electronics and solar panel industries. It gets trusted in plasma etching and cleaning, but its impressive efficiency comes with weighty safety challenges. Storing this compound isn’t just about following some rules written in a dusty manual. It’s about putting experience, science, and respect for danger into practice—something I learned firsthand when I visited a facility handling specialty gases.
Anybody who spends time with hazardous chemicals knows the importance of sweating the small stuff. Nitrogen trifluoride sits among those chemicals that can give seasoned workers pause. On its own, it doesn’t burn easily, but the real risk comes if it ever leaks. In air, under the right conditions, it can break down and release toxic or reactive byproducts. I remember a conversation with a plant manager who told me, “One missed step, and the cleanup isn’t just a mop and gloves—you’re calling the hazmat team.”
Storing nitrogen trifluoride means selecting tanks and pipes that do more than just hold pressure. Most teams I’ve seen go with high-integrity, corrosion-resistant materials—think stainless steel or a strong nickel alloy. Regular carbon steel won’t cut it because NF3 is an oxidizer, and it doesn’t take well to weak points in metal. Seals and valves get special attention too. It’s not a case for shortcuts or “good enough” parts. The cost of high-grade hardware pales in comparison to cleaning up a leak or replacing weakened containers.
Nitrogen trifluoride turns from gas to liquid under pressure. In hotter climates or during long storage, temperature swings can spike pressure inside a cylinder. Somebody in a control room has to watch for these changes, making sure the tanks don’t creep past their safe pressure limits. I’ve seen digital monitoring systems that alert staff the moment temperatures rise; it’s a far cry from handwritten logs or guesswork. Once, a maintenance tech showed me a pressure-relief valve that saved his team from a costly accident after a heat spike—technology and vigilance made the difference.
I’ve been inside storage rooms that felt more like wind tunnels because of powerful ventilation systems. That’s by design. The slightest leak gets handled as soon as possible, and airflow helps prevent vapors from collecting. In some places, I saw alarm systems and gas detectors hardwired into the building’s mainframe; if the monitors pick up anything unusual, everyone gets out, and emergency protocols kick in.
No safety guideline beats a team that knows what it’s doing. At every site I’ve visited, staff run drills, talk through worst-case scenarios, and carry personal gas monitors. Seasoned operators pass on lessons to new hires—not just where the valves are, but which wildcards can catch you off-guard. It’s always obvious when management invests in real training.
Nitrogen trifluoride isn’t leaving critical industries anytime soon. Those entrusted with its storage carry a heavy responsibility. The industry depends on strong materials, tight monitoring, powerful ventilation, and training that doesn’t get stale. With new technologies, safer storage becomes more reliable—but none of it works without a culture that puts people before easy shortcuts. In my experience, caring for safety is always the best investment, and it shows in every valve, every log, and every shift.
Folks might not hear the name nitrogen trifluoride too often, but this gas has gained attention because of its growing use in electronics manufacturing. In places like chip factories and display assembly lines, nitrogen trifluoride cleans surfaces with less fuss than older chemicals. Companies turned to it for efficiency. Still, few people talk about what happens after the workday ends and these gases head into the atmosphere.
Nitrogen trifluoride doesn’t pop up around neighborhoods or street corners. It mostly stays in industrial settings. With strong ventilation and controls, workers usually avoid direct exposure. If leaks do happen, breathing it in can irritate lungs, throats, and eyes. At high levels, oxygen gets pushed aside and, as I found out researching for a community training, symptoms get serious—think headache, dizziness, and even suffocation in extreme cases. Safety experts recommend monitoring and quick response plans in these settings. Regular checks save lives, especially with invisible gases.
Nitrogen trifluoride doesn’t get the headlines that carbon dioxide or methane do. Still, the environmental numbers can get a person worried. This gas traps heat in the atmosphere about 17,000 times more than the same amount of carbon dioxide. What’s shocking is not just how potent it is, but also how long it lingers—measured in centuries, not decades. Scientists started raising alarms in the early 2000s as emissions grew with the electronics boom. The numbers are small in comparison, but with climate change, every bit counts.
I’ve seen companies work hard to follow chemical regulations and trace emissions. The problem: no easy way for the public to find out just how much nitrogen trifluoride finds its way outside. Accurate tracking lagged behind its rise in use, which lets some releases go unnoticed. Environmental groups and climate researchers keep asking for tougher tracking rules—more data, posted in plain sight. Studies show that even small emission leaks add up worldwide.
Factories can swap out nitrogen trifluoride, but these substitutes usually cost more or clean less efficiently. Some engineers point to better recycling and destruction technologies at the end of pipework. Others suggest using less NF3 in the first place, urging manufacturers to rethink cleaning cycles or switch production methods. I’ve spoken with a few process engineers who worry about the tradeoffs. Green alternatives take time, money, and, most of all, a shift in corporate priorities. No one wants to drive up prices, but sometimes, the climate bill comes due later.
Nitrogen trifluoride doesn’t feel real to most people, but its fingerprints stretch from our gadgets to our atmosphere. Strong regulations close loopholes. Companies that lead on transparency set the standard for the rest. Policy moves like reporting requirements and research into safer chemicals help. The rest of us can keep pushing for accountability, using our voices and choices as consumers. As with any tough problem, it takes more than just the right equipment on the factory floor to solve it.
Nitrogen trifluoride doesn’t get much attention outside of chemistry or manufacturing circles, but it plays a much bigger role in the products we use daily than most people realize. With the chemical formula NF3, this compound combines nitrogen and fluorine in a way that creates a colorless gas under standard conditions. It hardly smells—the scent’s faint and a bit musty—though what matters more is the industrial impact and environmental concern wrapped into each of those floating, invisible NF3 molecules.
NF3 stands out for its stability and reactivity. That might sound odd—how can something be stable and reactive at once? In the tank, it's not likely to explode or degrade on its own, which makes storage and transportation less dramatic compared to pure fluorine gas. But inside plasma chambers, especially in places like flat-panel display or semiconductor factories, NF3 releases fluorine atoms at just the right moment. Those atoms scour surfaces clean, removing stubborn residues and prepping microchips for the next step.
At room temperature, NF3 behaves as a gas, just like any classic industrial feedstock. It's not heavy like some refrigerants and doesn’t pool. Its boiling point is around -129 degrees Celsius, so you won't see it turning into a liquid outdoors except in some arctic experiment. Don’t be fooled by its mild-mannered appearance—expose it to electrical discharge and the molecule cracks open, turning into a source of highly reactive fluorine.
Nitrogen trifluoride holds another distinction: it’s a greenhouse gas, and an extremely potent one at that. Data from the Intergovernmental Panel on Climate Change (IPCC) gives it a global warming potential much higher than carbon dioxide. Just one kilogram of NF3 in the air can trap heat tens of thousands of times more effectively over a century than a similar amount of CO2. Unlike the more infamous greenhouse gases, NF3 flies below the radar, partly thanks to its low emission levels relative to overall industry. Still, its use has grown along with the electronics sector, putting it firmly on the radar for those tracking climate impacts.
This isn’t fearmongering—numbers collected from environmental monitoring back up the rise in atmospheric NF3 over the last decade. Researchers have noted that neither regulations nor industry self-reporting fully capture the emissions tied to the compound’s life cycle. If we keep cranking out new displays, solar panels, and chips, efforts to minimize leaks and capture unused gas during production become more than a technical detail—they become an ethical responsibility.
More efficient capture systems at industrial sites could cut down on waste. Some companies already use recycling units for NF3, keeping as much material as possible in a closed loop. New plasma cleaning gases are also under investigation, many designed to provide the industrial benefits without the same environmental risks.
Fact-based oversight won’t stifle innovation; it creates trust. It’s not just about legal compliance or scoring environmental points. My experience working with scientists has shown me that people innovate fastest when they’re honest with each other, and when rules push them toward solutions rather than just checking boxes. If we care about microchips that drive our phones, and we want more tech that doesn’t quietly cook the planet, then taking a close look at compounds like nitrogen trifluoride makes a lot of sense.