N-Methylpiperazine has roots that reach back to the middle of the twentieth century, tying together the progress of organic chemistry and pharmaceutical research. Chemists in the 1950s and 1960s set out to modify piperazine rings, in part to improve drug solubility and to add new function to established skeletal frameworks. After its synthesis, N-Methylpiperazine found a seat at the table in a range of R&D labs, arising as an essential scaffold for medicinal chemists. Long before computers did the modeling, library after library of potential drug molecules started to feature this compound for one simple reason: it lends itself to endless molecular creativity. Patent records and journal literature from the latter half of the twentieth century trace its steady adoption from a chemical curiosity to a foundational building block, one valued both for its versatility in the lab and for the way it paves the way to new ideas.
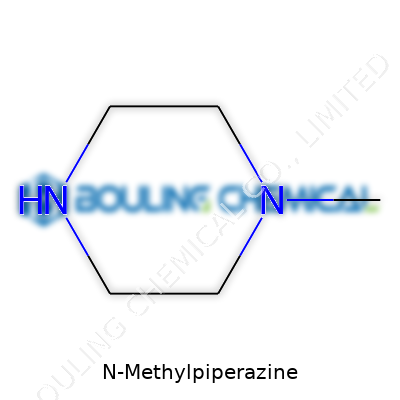
At its core, N-Methylpiperazine is a small, nitrogen-rich heterocycle—a ring structure with four carbons and two nitrogens, plus a single methyl group attached to a nitrogen. This compact architecture comes with a unique blend of chemical reactivity and physical stability. Labs focused on either pharmaceuticals or high-performance industrial polymers routinely use N-Methylpiperazine as a cornerstone intermediate. My own experience in bench chemistry underscores how much easier it becomes to introduce or mask nitrogen atoms in complex molecules with access to this compound. Its use extends past small-scale synthesis; industrial-scale production runs keep it on hand, especially as robust supply chains started to surface worldwide in the late twentieth century.
N-Methylpiperazine generally presents as a colorless liquid at room temperature, although it can occasionally crystallize in cooler settings. Its strong odor signals the presence of its basic amine functionalities. The chemical structure, C5H12N2, gives it enough water solubility to blend easily into aqueous solutions—a key trait that’s made it an attractive agent for both organic and aqueous synthesis. Its boiling point sits high enough for stability during standard reactions, yet it shows enough volatility that handling needs simple but steady caution. One unexpected gift comes in the way it resists breakdown under basic or mildly acidic conditions; piperazine rings do not hydrolyze or decompose in the way that some open-chain amines might. By taking up protons quickly, this molecule works well for preparing buffer solutions, enhancing both industrial and laboratory utility.
Handling N-Methylpiperazine goes beyond ticking boxes for purity or storage requirements. Chemical suppliers have learned that researchers expect clarity on level of impurities, typical concentrations, and reaction-grade ratings. Labels often specify whether the product supports pharmaceutical synthesis, polymerization, or other applications. From personal lab experience, I know well that overlooked detail—like residual water or unreacted precursor—can make or break a multistep reaction. Safety labels cover its toxicology profile and highlight its potential as a skin and respiratory irritant, so a chemist keeps goggles and gloves close. Storage recommendations tend to include cool, dry settings with tight seals, both to preserve quality and prevent unwanted absorption of CO2 from the air.
Chemists produce N-Methylpiperazine by introducing methylating agents—commonly methyl halides such as methyl chloride or methyl iodide—to piperazine under basic conditions. In a classic reaction scheme, the process calls for careful stoichiometric control to limit the production of unwanted byproducts like N,N'-dimethylpiperazine. Over-alkylation or incomplete reaction eats up precious time and materials in downstream purification, so process engineers tend to optimize batch parameters through temperature, solvent choice, and the gradual addition of reagents. Industrial setups take solvent recycling and waste mitigation seriously, using distillation and extraction techniques to purify the desired product and recover valuable starting materials. These days, research into greener and safer methylation routes continues, as chemists look to sidestep hazardous methylating agents wherever possible.
Once in the hands of a skilled chemist, N-Methylpiperazine offers a generous platform for derivatization. Nucleophilic substitution comes easily at the remaining nitrogen, enabling the attachment of acyl, sulfonyl, or alkyl groups. Functionalization at the ring’s carbons—though trickier—also has its place in pharmaceutical design, as medicinal chemists exploit this ability to tune biological activity. This compound behaves as an efficient ligand for metal-catalyzed reactions, especially in cross-coupling chemistry, where its ability to donate electrons steers reaction pathways. laboratories developing novel polymer technologies often use it as a cross-linking agent, leveraging its dual basic nitrogens to build robust molecular networks. Its reactivity keeps scientists returning to it, both for the familiar recipes of standard synthesis and for the boundless hope of discovery in new chemical landscapes.
N-Methylpiperazine’s name sticks in the mind, though its chemical aliases are equally important to recognize. Registration systems and textbooks reference it as 1-Methylpiperazine or 1-Methyl-1,4-diazacyclohexane. In older literature, the ring sometimes appears under cyclic amine codes or under trade names from chemical supply houses, linking diverse references back to a common foundation. Navigating this trail of synonyms means checking each structure, not just trusting a name. Lessons learned from my own library searches remind me how easy it is to overlook key hits by skipping over an unfamiliar code or IUPAC variation.
No chemistry bench runs safely without respecting the hazards of N-Methylpiperazine. Direct exposure can cause skin irritation or respiratory distress. Accidental spills create slip-and-inhalation hazards, made worse if open flames or static sparks fly. Good shops install exhaust hoods and monitor air concentrations, and training programs drill the safe use of gloves, goggles, and splash-resistant lab coats. Safety standards, shaped by decades of incident reports and regulatory oversight, call for secure storage of both raw materials and waste. Spills demand swift attention, both for chemical containment and for thorough documentation for workplace compliance. Burned into my own habits, these protocols save both lives and experimental results, keeping research on course and labs incident-free.
This compound has made itself at home in several worlds. Pharmaceutical drug discovery uses N-Methylpiperazine both as a building block and as a fragment in candidate drugs targeting nervous system disorders, viral infections, and cancers. Beyond the medical frontier, its structure brings value in polymers and specialty coatings, where it confers chemical resistance and flexibility. In agrochemicals, it serves as a key intermediate for herbicides and plant growth regulators. Water treatment solutions and catalysts in industrial chemistry also benefit, as N-Methylpiperazine can trap acidic gases and balance pH in complex mixtures. Many colleagues in the chemical industry refer to it as a “go-to” choice when robust nitrogen-containing cycles are needed for new formulations or exploratory syntheses, cementing its status as more than a niche reagent.
A look at recent research shows fresh enthusiasm for N-Methylpiperazine in drug design, material science, and catalytic technologies. Medicinal chemists working on central nervous system drugs have built entire compound classes off this structure, in part because it improves metabolic stability and brain penetration. Material chemists tinker with N-Methylpiperazine to make water-softening agents and new types of resins. Environmental R&D efforts look for greener, safer synthetic pathways both to make and to destroy the molecule, aiming to minimize side effects on ecosystems and human health. Academic and industrial labs alike have poured resources into machine learning and combinatorial chemistry, using N-Methylpiperazine as a test bench for new algorithms and robotic synthesis platforms. These developments show no signs of slowing, especially as the tools for chemical discovery keep growing faster and more precise.
Toxicology studies flag the need for measured handling of N-Methylpiperazine. Acute and chronic exposures have been tested in various model organisms, driving the setting of workplace exposure limits and informing medical monitoring of lab workers. Ingestion, inhalation, or skin contact bring pronounced effects—nausea, dizziness, and organ stress. Researchers dig into metabolic breakdown pathways, tracking metabolites and their potential cross-reactions, especially for long-term exposure risks. Waste streams get monitored for environmental persistence, since some amine derivatives linger and disrupt aquatic life. The upshot: this tool works best in hands that stay alert to the risks, keeping build-up in the body and in the environment at bay.
The years ahead promise both new opportunities and fresh challenges for N-Methylpiperazine. Pharmaceutical and agrochemical innovation requires ever-finer control over synthesis and safety, pushing chemists toward greener production lines and more selective transformations. Calls for sustainability mean tighter accounting for lifecycle and environmental footprints, from mining of precursor materials to final waste treatment. Ongoing advances in catalysis and automated chemistry open new routes and scale-up options, pulling N-Methylpiperazine deeper into the tapestry of discovery. Regulatory agencies, responding to updated toxicology data, could place stricter controls on workplace exposures or waste disposal. Engineers are starting to look at the circular economy potential too, recovering and reusing this compound in closed-loop systems. So while the compound’s backbone remains the same, the context around it evolves—reshaping the ways scientists and industries put it to work for medicine, manufacturing, and sustainability.
Everyday folks rarely come across the name N-Methylpiperazine, and that’s probably because it doesn’t show up in grocery aisles or pharmacies. Yet step behind the scenes and it turns out this chemical plays a bigger role than most people know in industries that reach deep into our lives.
N-Methylpiperazine, a small organic molecule, often gets added to chemical reactions to help make medicines work better during their development. In the hands of a chemist, it functions a bit like the reliable wrench in a toolbox. Drug makers routinely select it during the early stages of research because it helps build complicated molecules that target disease with precision, especially when making certain cancer therapies, antiviral drugs, and even some antibiotics.
My experience talking with researchers makes it clear that they appreciate tools that shave down tedious steps. N-Methylpiperazine helps rationalize the process of attaching active groups onto drug candidates—almost like connecting new Lego bricks to elaborate structures. This speeds up drug discovery, gives companies a chance to react to new health threats faster, and, in a big-picture way, keeps the pipeline of new treatment options open when diseases evolve. These developments aren’t obvious to those picking up a prescription, but the speed and flexibility this compound brings show up in what medicines get approved each year.
Outside of pharmaceuticals, N-Methylpiperazine often helps spin up materials for advanced electronics as well. I once toured a facility where chemists trialed compounds for battery development and specialty polymers—both needed in renewable energy tech. Again, the same qualities that help drugmakers—flexibility, reliability—carry over to engineers building more robust devices for tech and energy.
The ability to tweak properties in plastics, adhesives, and specialized resins starts in crowded labs using base chemicals like N-Methylpiperazine. In areas like water treatment or textile finishing, plenty of these same building blocks turn up as components in advanced products. For example, companies involving themselves in mining or purification often lean on chemical tricks to selectively bind and separate metals or impurities—a craft that needs reliable chemistry to keep costs down and yields up. N-Methylpiperazine sits quietly on those shelves, again making the process work.
Chemical manufacturing doesn’t come without responsibility. Research shows that routine handling, storage, and disposal matter a lot. Mismanagement leads to workplace hazards, contamination, or worse. Regulators tend to keep an eye on where intermediates like this travel. There’s good reason: even when companies follow best practices, there’s always potential for leaks and exposure. Proper containment, regular audits, and thorough employee training act as stopgaps. Still, I’ve heard stories from colleagues about accidents caused by simple shortcuts—a misplaced drum, a botched reaction, or improper cleanup. Each incident, even when minor, becomes a lesson that better oversight and transparency matter more than ever.
To keep this chemistry safe, organizations should stay wary of taking on more than they can responsibly handle. Investments in workplace safety, green chemistry programs, and chemical stewardship help avoid costly mistakes and support healthier communities around industrial sites.
The full story of N-Methylpiperazine rarely makes headlines, but its influence stacks up across many corners of manufacturing and medicine. By making chemical innovation safer and more efficient, it supports progress in quiet but essential ways.
Most folks aren’t thinking about chemicals as they go about their day. Still, names like N-Methylpiperazine show up all over industries, from medicine to manufacturing. The chemical formula for N-Methylpiperazine is C5H12N2. To some, that collection of letters and numbers might not mean much. Yet, for chemists, pharmacists, and folks working around new materials, understanding molecules at a formula level often means turning ideas into safe, practical products.
A lot of modern medicines and coatings trace back to the lab benches where chemicals like N-Methylpiperazine were first mixed. I remember talking with a friend in pharmaceutical research. He stressed how even a single carbon or nitrogen atom can change how a drug behaves. N-Methylpiperazine, built with five carbons, twelve hydrogens, and two nitrogens, sits at the center of many transformations. Some cancer treatments, antidepressants, and other therapies wouldn’t see the light of day without the right building blocks.
Science teachers often say molecules are like Lego bricks. You snap together different shapes and sizes to invent something stronger, more useful, or safer. Knowing the correct formula lets chemists predict how a chemical might react, whether it fizzles harmlessly or creates something dangerous. In the wrong hands or handled carelessly, even a common chemical can become problematic. Understanding what C5H12N2 actually looks and acts like cuts down on mistakes and saves lives.
Working safely starts with clear information. When scientists know a chemical’s formula, they map out its behavior and possible hazards. It feels a lot like reading a weather forecast: you get ahead of trouble by knowing what's coming. N-Methylpiperazine’s formula helps researchers set up the right protections in a lab or manufacturing plant. Gloves, goggles, proper ventilation—nothing fancy, just smart steps supported by solid facts.
Mistakes in chemistry can cost money and sometimes much more. Reliable details let companies build processes that keep both their workers and the wider community out of harm’s way. In schools and small labs where funds run tight, clear data, starting with the chemical formula, helps folks handle chemicals responsibly.
News cycles move fast, and people have a right to feel confident that substances in their food, water, and medicines are managed responsibly. Sharing key facts like the correct formula demonstrates transparency. It invites deeper trust between scientists and the public. I’ve seen how a short conversation about what’s really in a compound can clear up confusion or even nip a rumor before it starts. Giving communities access to science, not just experts, benefits everyone.
Industry’s next step isn’t just knowing what’s in N-Methylpiperazine, but figuring out the best ways to handle, transport, and dispose of it. Green chemistry encourages us to look for safer alternatives when possible. Even with a familiar, well-understood compound, people keep asking – can we do this better, safer, gentler on the earth?
Knowing the chemical formula isn’t about complicated trivia. It’s a practical step, making science work for people. Whether someone is stocking shelves or discovering tomorrow’s medicines, every detail matters.
People in labs or chemical plants sometimes come across N-Methylpiperazine. If you don’t work with specialty chemicals, the name probably looks intimidating. Over the years, I’ve learned that the questions matter more than the jargon. N-Methylpiperazine is clear, has a strong smell, and tends to show up in pharmaceutical or agrochemical work.
Stories float around about what it actually does to your immune system, skin, or lungs. Some folks shrug off concerns—“It’s just another lab solvent.” Experience suggests otherwise. Getting a chemical splash on your hands or a noseful of fumes can change your perspective. Skin absorbs more than most think, which means even a small spill could lead to irritation or worse. The strong odor signals something: your nose is warning you. Some evidence points to irritation of skin, eyes, or airways after exposure, and that's not just theory in a textbook—people in real-world settings have felt the sting.
I’ve seen workers rush through a task and skip the gloves. Maybe the job feels routine, or maybe someone once said the risk is low. Then a splash causes redness and burning, or worse, allergic reactions pop up with repeated exposure. Not many have the patience to comb over old studies or safety guidelines, but missing information doesn’t mean there’s zero risk. The chemical industry tracks incidents because people sometimes get hurt, and the same ingredient in the wrong hands can cause trouble.
Inhalation is another real threat. Fumes can build up in closed rooms if the air isn’t moving. The U.S. National Library of Medicine lists N-Methylpiperazine as a harmful substance if inhaled or swallowed. In my time helping students run experiments, I’ve stressed the same rules every week: use a fume hood, and don’t skimp on face protection. An extra few minutes of setup keeps lungs and eyes clear. Ignoring these basics creates stories no one wants: headaches, breathing issues, and sometimes lasting health problems.
Solutions start with recognizing danger. The biggest leap comes from culture, not just gear. Fact sheets from NIOSH and the European Chemicals Agency spell out the risks, but nothing replaces someone refusing shortcuts. Chemical safety is about habits—labeling bottles, checking gloves for holes, and having proper spill kits nearby. Teams with good habits don’t see as many “accidents” because they never treat risk as an afterthought.
Sharing stories is just as important as reading rules. Colleagues who talk frankly about fumbling a beaker or feeling the telltale redness do more to keep others alert than laminated posters ever can. Sometimes the safer solution is substituting a less hazardous ingredient when possible. Ongoing reviews of chemical inventories, routine training, and frequent air monitoring all add up.
Trusting old wisdom puts people in danger if facts change or surprises turn up. N-Methylpiperazine might help build a medicine, but it doesn’t care who uses it. Honest sharing of actual incidents—good and bad—broadens perspective. Policy and practice work best hand in hand. Relying on expert guidance from agencies, maintaining strong habits, and facing every chemical with due respect—this approach has served generations of researchers, and it still does now.
N-Methylpiperazine shows up in more chemical supply rooms than most people realize. Chemists know it for its role in making everything from pharmaceuticals to additives. Yet even folks just stocking shelves have good reason to care about how it’s handled. From real experience in small labs and bigger facilities, mistakes are rarely about complicated theory. Usually, they trace back to someone cutting corners or ignoring old warnings about proper storage and safe handling.
This compound, a clear liquid with a musty odor, brings its own quirks to the storage game. It's hygroscopic, which means it slowly pulls moisture out of the air. That moisture can ruin its usefulness for sensitive reactions. The vapor and liquid also act as irritants — touch or inhale too much, and you’ll remember it. People who handle drums or bottles of the stuff know gloves and goggles are about more than ticking off safety boxes. There’s little room for mistakes once you splash or spill even a small amount.
I’ve seen many storage closets over the years. The best ones share a few basic habits. Workers keep N-Methylpiperazine inside well-ventilated rooms, away from strong oxidizers or acids. Industrial suppliers often recommend tight-sealing containers, often stainless steel or thick glass, to keep moisture and air from creeping in. Labeling looks boring until you remember how similar a lot of these bottles look. Clear names, hazard warnings, and dated shelf checks can mean the difference between routine work and panicked first aid.
Temperature accidentally gets ignored, but it lines up with longevity. Warm rooms can speed up degradation and risk pressure buildup inside sealed bottles. A stable, cool spot – usually between 2°C and 8°C – works best. I’ve learned not to trust aging refrigerators or forgotten corners of equipment rooms; leaks or condensation add new headaches. Spills on warm surfaces can create nasty vapors fast, so clean, uncluttered storage areas help keep surprises to a minimum.
Easier access to training now means there’s less excuse for risky short-cuts. In too many accidents, someone skipped using proper chemical-resistant gloves or worked with an open bottle out in the open. Breathing masks seem clunky until you see someone start coughing – or worse – from an unplanned splash. Ventilation counts for more than people admit. I follow old habits, like double-checking that fans actually work before pouring or measuring, after seeing make-shift setups let harmful fumes build up dangerously.
Regulatory guidelines from OSHA and the European Chemicals Agency do a good job laying out safety hoops to jump through. Yet the reality always comes back to the habits of those who work most closely with the chemical. Regular refresher sessions and honest accident reports teach more than colored posters. I try to bring up stories of near-misses in every training, because the human element sticks longer than a checklist. People care about stories that involve real risk and real consequences, and those stories seem to do more to change habits than any abstract rulebook.
N-Methylpiperazine won’t become safer just by writing rules. It happens through the unglamorous daily grind of checking bottles, honoring warning labels, and refusing to get lazy about protective gear. Those details add up, making a world of difference not just in compliance, but in the basic safety and well-being of workers everywhere who come into contact with tricky chemicals.
If you’ve spent time working in research chemistry or pharmaceutical supply, chances are you’ve run across requests for N-Methylpiperazine. It’s a common piece in the synthesis toolkit, especially valued by those grinding away in R&D labs or scaling up to kilo-labs. In my years talking shop with chemists and following the movement of fine chemicals, the question of purchasing isn’t simply a matter of hitting “add to cart.” It’s a dance with regulations, sourcing reliability, ethical practice, and real safety concerns.
Most professionals start with big-name scientific suppliers—think the likes of Sigma-Aldrich, Fisher Scientific, TCI, or Alfa Aesar. These suppliers don’t hide their catalogs but they don’t just ship to anyone. To buy N-Methylpiperazine, buyers usually need to show credentials: company affiliation, a verifiable shipping address tied to an institution, and, quite often, a reason why the chemical is needed. Routine as it sounds, this gatekeeping blocks a surprising number of problematic transactions.
Bulk orders play by stricter rules. Chemical vendors treat bulk quantities as a red flag for resellers and regulatory authorities. Suppliers in the United States, Europe, and much of Asia require further paperwork, sometimes including detailed end-use declarations. In all my time navigating these waters, customers who expect to click and pay for bulk chemicals without scrutiny get nowhere. Even university teams face checks for larger amounts.
It’s not hard to find listings on platforms like Alibaba or smaller B2B sites. Here, the vibe is more mercenary. A buyer could theoretically purchase from overseas brokers, especially in regions with a lighter approach to controls. Risk stacks up fast: issues with product purity, mislabeling, and the possibility of something being outright illegal depending on the chemical’s categorization in your country.
Regulatory bodies such as the DEA in the U.S. or REACH in Europe may treat seemingly innocuous chemicals like N-Methylpiperazine as “precursors.” This means purchasing, especially in bulk, sometimes bumps you into regulatory regimes that carry heavy fines and criminal charges for missteps. It’s not something that only worries big pharmaceutical manufacturers. Even lab startups in shared workspaces have landed on government watchlists for failing to dot every last “i.”
Strict sourcing expectations didn’t come about just for tradition’s sake. N-Methylpiperazine, like many intermediates, can be repurposed toward more dangerous ends. It’s fair to say, based on conversations I’ve had with suppliers, that a shadow market exploits these same supply chains. Recently, news broke about bad actors diverting fine chemicals for synthetic drug labs. Even though most buyers are legitimate, this backdrop creates a demand for diligence at every step.
If you need N-Methylpiperazine for a legitimate project, the right answer often starts with openness. Contact known suppliers, provide full documentation, and stick to the regulations. Some organizations now employ third-party audit programs—these add paperwork but let them order what’s needed through pre-approved processes. For start-ups and university teams, working with institutional procurement offices helps avoid compliance surprises down the line.
It’s tempting to look for speed but cutting corners never pays off in specialty chemicals. Building direct relationships with trustworthy vendors goes a long way. Those vendors offer support understanding rules, maintaining records, and following safe practices—important for safety, legality, and protecting one’s own reputation too.