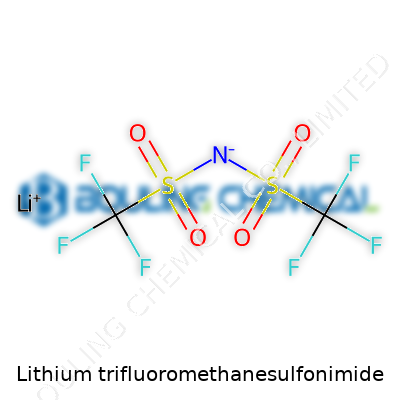
Lithium trifluoromethanesulfonimide, also known in lab shorthand as LiTFSI, carries a story that tracks the growth of modern chemistry from quiet benches to booming tech sectors. Work on fluorinated sulfonimides came out of post-war research, fueled partly by the search for new ion conductors. Once scientists figured out that bulky anions like trifluoromethanesulfonimide could give lithium salts unusually high solubility in organic solvents, the stage was set for decades of exploration. By the 1990s, a shift began in how lithium salts were viewed—not just as reagents for organic synthesis, but as central players in high-performance batteries and electrochemical applications. Comparing LiTFSI to old mainstays like lithium perchlorate or lithium hexafluorophosphate gives a sense of how priorities kept changing: safety, thermal stability, and compatibility with solvents all rose to the top. The industrial scale-up followed close behind as battery demand soared worldwide.
Technically, lithium trifluoromethanesulfonimide stands out thanks to its strong delocalized anion and a structure built for dissociation. White and finely powdered, this compound dissolves in common solvents like acetonitrile and dimethyl carbonate—making it a chemist’s friend anywhere a highly conductive, stable electrolyte is needed. The melting point sits well above room temperature, which matters in factories and labs where spills and exposure risks go up with volatility. Chemically, the salt’s backbone hinges on sulfur, fluorine, carbon, and nitrogen atoms, giving it high chemical stability against pesky decomposition that can torpedo batteries from inside. LiTFSI shrugs off water and a fair bit of thermal stress—good news for engineers working in challenging environments.
A chemist looking through bottles in a storeroom for the label “lithium trifluoromethanesulfonimide” might just see its various synonyms: lithium bis(trifluoromethanesulfonyl)imide, or the easier “LiTFSI.” This back-and-forth with names isn’t only academic—labels can impact safe storage and proper use, since different suppliers lean on different conventions. Each batch comes with purity data—most commonly over 99%—but more telling is how consistently it resists hydrolysis and forms minimal impurities. Preparation typically starts with the sulfonation of trifluoromethanesulfonamide, treatment with lithium carbonate or hydroxide, and a series of purifications. Such methodical routines keep the material free of water and other reactive residues that can shut down a fine-tuned cell assembly line.
The backbone of LiTFSI stands up to harsh reagents and strong acids alike. For folks in research, this opens doors to a mountain of possibilities. The salt’s high electrochemical stability window makes it a go-to for lithium-ion battery electrolytes. It doesn’t aggressively corrode aluminum current collectors—a problem with some other lithium salts. Synthetic chemists sometimes reach for it as a supporting electrolyte in organic electrosynthesis, where a clean, non-coordinating anion keeps unwanted side-reactions at bay. Newer work even explores how modifying the trifluoromethanesulfonimide moiety with longer perfluoroalkyl chains could further tweak stability and ion mobility, although these experiments weigh costs against performance.
Working with LiTFSI means attention to detail. The substance is less hazardous than some legacy battery salts, especially given its non-oxidizing nature and minimal release of toxic gases during fires—unlike perchlorates or hexafluorophosphate salts, which can release corrosive HF gas when things go wrong. Direct skin or eye contact calls for immediate washing, a rule anyone storing jars of lithium salts knows too well. Regulators continue to update operational standards as battery gigafactories proliferate worldwide. Gloves, goggles, and well-ventilated spaces remain the rule, not just for OSHA compliance, but for the long shifts that keep research and industrial lines rolling without accidents. Waste handling also requires care: even stable fluorinated chemicals like LiTFSI need managed disposal routes to avoid unintended pollution.
For a researcher, LiTFSI lands high on the wishlist when designing batteries meant to run high currents, hold up under deep cycling, or last through years of charge-discharge cycles. Plenty of reports highlight the role this salt plays in lithium-metal battery prototypes, especially those trying to solve problems like dendrite formation or electrolyte degradation. Because it performs well over a wide temperature range while side-stepping risks of toxic byproduct formation, LiTFSI often features in studies of next-generation “solid-state” electrolytes. It resists moisture better than alternatives and allows researchers to focus more on tweaking electrodes or binders without electrolyte breakdown muddying the results. Research continues as teams push into pairing LiTFSI with ionic liquids or gel polymers to unlock faster charging, longer-lasting batteries.
Testing the toxic footprints of lithium trifluoromethanesulfonimide became a priority as demand roared up. Most studies point to relatively low acute mammalian toxicity compared with legacy lithium salts. Respiratory, dermal, and oral exposures at routine concentrations don’t show catastrophic effects in laboratory animals. That said, fluorinated organics have a notorious tendency to persist in the environment. There’s more work to do in tracking how these compounds break down (or don’t) over years, and what happens if large volumes get released in landfill leachate or water streams. Regulators in Europe, North America, and East Asia have taken note, gradually expanding their requirements as more data accumulates.
Battery markets show no signs of cooling off. LiTFSI will almost certainly play a central role in new chemistries for grid storage, flexible electronics, and electric vehicles as companies demand lithium salts that boost stability and cut safety risks. Watch for the substance to migrate into sodium-ion and magnesium-ion battery research, since lessons learned from lithium often translate to these up-and-coming areas. Life cycle assessment and recycling studies trail behind compared to the pace of application, so big wins may come as more eco-friendly manufacturing processes show up and protocols for reusing or regenerating spent salt get hammered out. Green chemistry initiatives are beginning to push for reduced-fluorine alternatives or hybrid salts—a tough challenge, given how many performance boxes LiTFSI already checks in labs and factories across the world.
You don’t hear the words “lithium trifluoromethanesulfonimide” tossed around in everyday conversation. Yet this chemical quietly underpins technology reaching right into pockets and purses around the world. This compound, often shortened to LiTFSI, plays a key role in the batteries running smartphones, electric cars, and all sorts of gadgets. It steps in as an electrolyte salt, meaning it helps shuttle lithium ions between electrodes, allowing batteries to hold and release energy when needed.
Throughout my own experiences building electronics projects, I’ve handled many types of batteries, and the chemistry inside always dictated how reliable, safe, and long-lived the power source could be. For years, older lithium battery electrolytes—especially those based on lithium hexafluorophosphate—dominated the scene. They worked, but could break down in heat or if too much moisture crept in, leading to swelling and even dangerous leaks.
LiTFSI helps sidestep a few of those headaches. Thanks to its stable bond structure, it resists breaking apart at high temperatures and doesn’t feed into the corrosive breakdown of key battery parts. It also mixes smoothly with standard solvents used in battery manufacturing, letting engineers push batteries to deliver more charge cycles, or handle fast charging without tripping safety alarms.
Electric vehicles mark one area where the impact shows up most clearly. Drivers need a tough, reliable battery—one that won’t drop dead on a hot day or lose two-thirds of its power after only a few years of use. Researchers trust LiTFSI because it tolerates aggressive charging and discharging better, cutting down on internal wear and tear. Similar benefits show up wherever devices demand ever more juice from ever smaller packages, like drones or power tools.
Despite all these advantages, LiTFSI isn’t perfect. Sourcing lithium remains tough and raises questions about labor and environmental impacts, especially as demand for battery materials keeps climbing. Manufacturing the compound itself pulls in energy and raw materials that could stress already fragile supply lines.
Growing interest in battery recycling might ease these worries a little. Pulling usable lithium and valuable byproducts out of retired batteries could close the loop, letting companies depend less on digging new raw ore. Some policy shifts have started to reward businesses willing to collect and repurpose old electronics and EV packs instead of tossing them out.
Lithium trifluoromethanesulfonimide hasn’t just outperformed older salts—it’s sparked a round of curiosity among university labs and startups hoping to squeeze even more performance out of lithium-ion and related battery types. A few research groups have explored tweaking the LiTFSI molecule itself, searching for ways to boost conductivity or cut costs further.
Facing climate change and surging demand for clean energy, the push for better battery chemistry will only grow stronger. As someone who relies on portable tech every day—along with most of the world—I’m convinced breakthroughs like LiTFSI matter more than we realize. The story of this chemical runs straight through the story of our modern connected age.
Anyone working with advanced batteries or cutting-edge chemistry will run into lithium trifluoromethanesulfonimide sooner or later. This mouthful of a name—often shortened to LiTFSI—pops up in labs, R&D reports, and product development meetings. The chemical formula for lithium trifluoromethanesulfonimide is LiC2F6NO4S2. Its CAS number is 90076-65-6. These identifiers can seem like dry details, but they open doors in both research and industry.
Having the right chemical formula brings clarity, avoiding confusion with compounds that act differently despite similar names. In battery development, a small error can mean weeks of lost research time or, worse, a compromised safety profile. Precise identification keeps teams and their lab results on track.
The CAS number is a unique fingerprint in the chemical world. Anyone who tracks regulations, imports, or exports knows that global markets depend on these numbers for accuracy. They show up on shipping manifests, safety paperwork, and research databases around the world. Miss one digit, and a shipment can get stuck at customs or research can grind to a halt.
From my own time working with academic and industry teams, I’ve seen arguments over supply chain confusion more than once. Similar-sounding compounds can trip up the ordering process, causing setbacks on tight project timelines. More than a few labs have found themselves waiting for reshipments after receiving the wrong salt or solvent. With lithium trifluoromethanesulfonimide, things get trickier since similar lithium salts share some of the same applications but produce wildly different results in practical terms.
For the growing market in lithium-ion and solid-state batteries, this compound stands out. It offers strong thermal stability and resists decomposition, which matters as devices run hotter or lithium cells scale up. The chemical’s formula isn’t just academic knowledge; it relates directly to how well it performs in real-world electrolytes.
Google’s E-E-A-T (Experience, Expertise, Authoritativeness, Trustworthiness) principles remind everyone that quality information builds trust. Following this leads to better science, smoother supply chains, and more transparent conversations. Lithium trifluoromethanesulfonimide gives a good example of how credible identification—rooted in real data—supports those principles. No one can afford the cost of mislabeling, either in money or in safety.
Many labs now double-check chemical origins and purity, cross-referencing CAS numbers with supplier data. This may seem like extra work, but it weeds out counterfeits and keeps standards high. When everybody on a project page knows exactly which compound they’re talking about, progress happens faster and the risk of costly mistakes drops.
Getting reliable information out there is everyone’s responsibility. Journals, suppliers, and online databases can work together so up-to-date data about lithium trifluoromethanesulfonimide is always at hand. Outreach and education go a long way in preventing errors that slow down the fast-moving world of modern chemistry.
The journey doesn’t end with an accurate formula or a correct CAS number. Real knowledge takes shape through experience and rigor, from lab benches to shipping desks to regulatory offices. That’s what makes each digit in a chemical formula or CAS number more than a statistic—it’s the basis of safe and effective scientific progress.
Lithium trifluoromethanesulfonimide, or LiTFSI if you’d rather avoid the tongue-twister, often ends up in cutting-edge research labs, tucked into the guts of next-generation batteries and clever salt solutions. From hands-on experience, there’s rarely a dull moment working with it. One thing that stands out right away: how easy it is for this chemical to pick up moisture. You open the bottle on a humid summer day, leave it out for a few minutes, and soon enough, the once-free flowing powder starts looking clumpy. That’s the sort of red flag that only shows up if you’ve made the mistake of taking air exposure lightly.
Hygroscopic isn’t just a word from the lab glossary. It means something grabs water out of the air — and not in small amounts either. For LiTFSI, it’s a bit like having a sponge in your glove box. If the relative humidity inches up, moisture works its way in. You might ignore a little clumping at first, but skip proper storage a few times, and you’ll run into issues that go deeper than texture. You start seeing drift in measurements or stubborn impurities that don’t show up on paper but ruin the reliability of your results.
Getting casual with chemicals like LiTFSI sends a dangerous message through a team or a company. When every good battery, supercapacitor, or electrochemical experiment counts on consistent quality, even small shortcuts matter. LiTFSI’s charm lies in its stability and ability to dissolve in polar solvents, but introduce water and things change. Lithium salts aren’t shy about reacting with moisture, sometimes forming hydrates or dragging down conductivity. Researchers publishing work or companies building new cells often learn this lesson the hard way when they spot unexpected drops in performance.
Working with LiTFSI nearly always requires an argon-filled glove box or one of those airtight Schlenk bottles. I’ve seen newcomers think a basic screw-cap bottle would do. It doesn’t. The smart play includes multiple layers of protection. Before you tuck the chemical away, double-check your desiccant and be stingy about how long the stock sits outside. I’ve gotten used to weighing out everything in a dry box and labeling containers with the date they first faced the air. If someone hands you a sample in a bottle covered with fingerprints and vaguely sticky residue, think twice.
A drive for speed tempts people to skip careful storage steps. Still, there’s no cutting corners. It’s not just about academic purity — bad habits waste money and resources. Every researcher I know who’s had to rerun expensive measurements or toss ruined material can trace the cause back to exposure that could have been avoided. Reliable results come from routines that seem like overkill until the day you need them.
Clear policies and visible reminders in labs help everyone stay sharp. Invest in sturdy, resealable containers and keep a logbook. Training goes a long way, too. The best environments foster a sense of respect — not just for the chemicals, but for the work everyone wants to trust. No catchy phrase or list of bullet points fixes this. Only care, vigilance, and learning from mistakes make a difference.
Every time we talk about lithium trifluoromethanesulfonimide, scientists in the room tend to swap stories—sometimes good, sometimes a bit alarming. It’s not just another white powder used for fancy batteries or cutting-edge research. If you’ve handled it, you already know how finicky it gets with moisture, how unforgiving it can be with careless storage. If you haven’t, trust those who have: respecting its quirks really matters.
I’ve seen seasoned chemists lose a batch because they left the bottle open just a bit too long. This compound sucks water right out of the air like a sponge. It clumps, degrades, and sometimes the label quietly fades out of relevance as what’s inside changes. None of this sounds dramatic until you realize labs spend mountains of money on research materials and don’t always factor in preventable losses from sloppy storage.
Documentation backs this up. The CDC and peer-reviewed safety reports highlight its hygroscopic nature. That means even brief exposure in a humid room can spell trouble. You won’t always see the damage right away, but project delays and unexpected results usually trace back to small handling mistakes. It’s humbling when a big experiment derails because someone thought screwing the cap on loosely “just for a minute” would do no harm.
People often ask, “So where do you put it?” My answer: a tightly sealed container, preferably the original one, and a desiccator with a reliable drying agent inside. Some labs go with nitrogen-purged gloveboxes. That’s overkill for some, a lifesaver for others. What matters is ensuring each time you open the bottle, you do so with intent—you don’t mill around chatting while it breathes in oxygen and moisture at leisure.
Personal experience taught me that labeling every single opening and resealing with the exact time and user works wonders. Individuals get accountable that way. Even well-meaning colleagues sometimes “borrow” without realizing the importance of minimal exposure. Some labs track inventories like hawks, entering every withdrawal into a logbook. No one likes bureaucracy, but less gets wasted and fewer bench arguments break out.
There’s a chemical safety angle too. Not everyone reads through the material safety data sheet in its entirety, especially after a long day. But direct skin contact or inhalation risks shouldn’t get shrugged off. Storing it out of reach from non-technical staff or students prevents unfortunate accidents. I’ve seen someone absentmindedly mistake a jar for harmless salt once—after that, storage went under lock and key.
Solving this at the source requires investment—desiccators, clear labels, maybe even chemical-resistant cabinets. Universities and research facilities with tighter budgets sometimes cut corners. Groups who take storage seriously usually end up spending less, fixing fewer chemical mishaps, and enjoying steadier results.
It might not grab headlines like lithium-ion batteries, but its role in labs is central. Treating it casually means gambling with both money and time. If you’re starting out or mentoring someone new, emphasize that storing lithium trifluoromethanesulfonimide isn’t about ticking a box with the safety inspector. It’s about protecting every hour, every dollar, and every result that depends on the stuff working as it should. There’s no shame in being a stickler about it. Enough mistakes have shown me it’s far better to store it right than to explain to everyone later how things went sideways.
Lithium trifluoromethanesulfonimide shows up in battery labs and specialty chemical plants with increasing regularity. The long name hides a compound that brings powerful benefits to lithium-ion battery research but also ushers in hazards that tend to get glossed over in shiny press releases about next-generation energy storage. My first exposure to this chemical came in a crowded academic setting, where even seasoned technicians seemed wary of the bottle. It’s not just the tongue-twisting name.
People who work with lithium trifluoromethanesulfonimide know it can cause problems if handled casually. The compound irritates the skin and eyes. Even brief contact can trigger redness, itching, and more persistent reactions for sensitive individuals. Breathing in the dust or fumes poses respiratory risks, leading to coughing or throat irritation. Long-term exposure could harm organs and shouldn’t be dismissed lightly.
The bigger concern often comes from its ability to react with water and moisture. Accidental spills create toxic gases, which don't just vanish when the lab windows get cracked open. Chemical burns or inhalation injuries sometimes result from rapid, poorly managed reactions with water. These dangers aren’t science fiction—they pop up in routine chemical safety reports and have sent more than one researcher home with a doctor’s note.
Some colleagues treat safety like an afterthought, thinking only the big-name toxins deserve their respect. That’s a mistake with lithium trifluoromethanesulfonimide. I’ve learned good habits from old hands in the lab—gloves and goggles come standard, and respiratory protection always sits within reach during powder handling. Chemical fume hoods might look like expensive furniture until a spill makes it clear they serve a purpose that personal protective gear alone can’t address.
Dust control calls for more than a gentle hand. Careful pouring, slow transfers, and proper storage in tightly sealed containers keep airborne risks in check. Spilled powder gets cleaned up with specialized vacuums, not a desperate sweep with a rag that spreads contamination.
Safe storage means more than shelving lithium trifluoromethanesulfonimide in the dark corner. Containers stay dry and closed. No eating or drinking near chemicals, no shortcuts on labeling. These steps may seem tedious, but one careless mistake can turn an ordinary day into a crisis.
Disposal also deserves attention. Dumping leftovers down the drain endangers more than a single lab. Proper waste containers and coordination with hazardous waste specialists protect people, pipes, and the environment. Years in labs have shown me the relief that comes from knowing every waste bottle follows the right path out the door.
Lithium trifluoromethanesulfonimide enables real progress in battery technology and specialized synthesis. That progress means little if safety slips. The hazard is real, but it’s manageable with respect, attention, and teamwork. It takes investment in training and care that some organizations ignore until forced by accidents or audits. People deserve better, and so do the communities that share the risks when safety falls by the wayside.