Science keeps finding ways to move forward, and lithium trifluoromethanesulfonate, known by many as lithium triflate, reflects that ongoing work. Researchers first synthesized lithium triflate in the latter half of the 20th century, driven by the search for stable and highly conductive lithium salts for novel applications. Battery chemists looked for alternatives to perchlorates, which raised safety alarms due to instability. The urge grew out of practical needs—from the demand for safer, faster-charging lithium batteries to breakthroughs in organic synthesis requiring robust, non-coordinating anions. Synthetic routes grew more efficient, pushing lithium triflate from obscure lab curiosity to mainstay in laboratories and factories. I remember the gradual excitement among fellow researchers, especially those working through the 1980s and 1990s; the adults spoke in hushed tones about replacing problematic salts with this new candidate that ticked both the boxes of safety and performance.
People in science and tech often cross paths with lithium triflate because it solves problems other salts can't shake. The compound has become a favorite in the electrolyte mix for lithium batteries and as a versatile reagent in the synthesis of complex molecules. As lithium-ion technology advanced, the need for reliable, highly soluble, and chemically inert lithium salts grew. Here, lithium triflate stands out by trading volatility and reactivity for stability. Looking back, anyone tinkering with electrolytes noticed how older salts like lithium perchlorate required delicate handling and sometimes put projects at risk; lithium triflate changed the landscape by offering more forgiving properties.
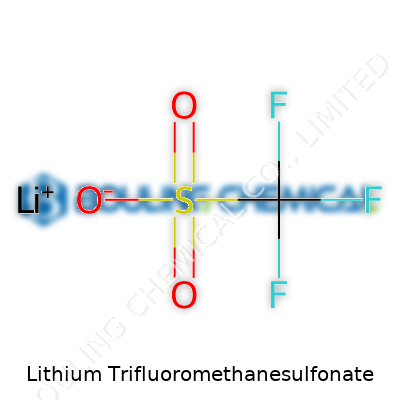
Lithium triflate presents as a white, crystalline powder. It has low hygroscopicity, resisting clumping from moisture, making it easier to store and work with in less controlled environments. Its chemical formula, LiCF3SO3, explains a lot: the trifluoromethanesulfonate anion gives the compound its outstanding thermal and chemical stability. The salt dissolves well in polar solvents, including water and organic polymers. That’s not just a detail on a sheet—handling lithium triflate in the lab means less fiddling to get it into solution, translating to time saved and fewer ruined experiments.
Manufacturers provide high-purity lithium triflate, with purity grades suited for either research or industrial processes. Labels usually note the precise mass fraction of lithium and triflate ions, moisture content, and batch-specific trace impurity data. In the real world, this means researchers can focus on their application instead of worrying about batch-to-batch inconsistency. For battery engineers and synthetic chemists, purity isn't just a nice-to-have; trace contaminants cause unwanted side reactions and performance drops. Reliable sourcing keeps labs and factory lines running smoothly and safely.
Technicians often produce lithium triflate by reacting trifluoromethanesulfonic acid with a lithium source such as lithium carbonate or lithium hydroxide. The acid and base combine in aqueous or alcoholic medium, followed by steps to remove solvent and purify the resulting white solid. I recall a few hours in graduate school spent around a flask, watching crystals fall out. The method sticks in your mind not for its bells and whistles but for its clarity—each ingredient does its part and gets out of the way, making this an accessible synthesis for most well-equipped facilities.
One reason lithium triflate draws attention lies in its chemical resilience. Its anion, the triflate group, is large and spread out, reducing its power to interfere with other chemical players. This makes it a preferred background electrolyte in electrochemical research and a friendly companion in organic synthesis. The lithium cation, on the other hand, participates in forming new bonds and facilitating reactions with carbonyl groups, among others. In battery formulations, lithium triflate’s stable nature translates to less breakdown of solvents and fewer unwanted byproducts at the electrodes, bolstering both performance and cycle life of advanced batteries.
Depending on the label, you’ll spot lithium triflate under several names: lithium trifluoromethane sulfonate, lithium triflate, or LiOTf. Chemical catalogs and safety documents often provide all the common names to steer users clear of confusion. That consistency matters—folks who work in global teams or order supplies across borders rely on knowing precisely what sits in each container. Recognizing each synonym keeps projects—and sometimes entire research programs—moving without avoidable mix-ups.
Lithium triflate’s safety profile draws interest from both regulatory bodies and on-the-ground scientists. Unlike lithium perchlorate, which can explode under the wrong spark or pressure, lithium triflate offers a more forgiving profile. That being said, appropriate care remains a must. The salt can irritate eyes, skin, and respiratory systems, especially in concentrated forms or fine dust. Workplaces emphasize handling it in ventilated areas, using gloves, and wearing goggles. Material safety data sheets spell out these measures based on practical incidents rather than theoretical risks. In my own experience, a few extra minutes spent on proper setup saves days lost to cleanups or worse, medical visits. Fire departments and first responders appreciate the lower hazard score lithium triflate brings to battery manufacturing plants and labs.
The sheer range of lithium triflate’s applications tells its story better than many technical reviews. Modern lithium batteries often include lithium triflate as a go-to electrolyte salt—its wide electrochemical window and chemical stability support repeated charging and discharging cycles, even under tough conditions. Researchers in organic synthesis appreciate how lithium triflate stays out of complex reaction pathways, letting them push for cleaner yields in challenging transformations. The pharmaceutical sector also picks up on its properties—certain drug syntheses lean heavily on lithium triflate to keep unwanted side reactions at bay. Polymer chemists combine it with monomers to boost ionic conductivity in solid electrolytes, opening doors for flexible batteries and wearable electronics. The consistent theme: lithium triflate adds value by underlining performance and safety rather than dominating the chemistry.
Work on lithium triflate hasn’t slowed down. Teams in universities and industry labs keep experimenting with new solvent systems and polymer matrices, hoping to unlock even higher conductivity and stability without the risks attached to other lithium salts. The race for safer, longer-lasting, and faster-charging batteries in electric vehicles turns research curiosity into commercial urgency. Proprietary electrolyte blends often feature lithium triflate, with engineers probing the fine details: how it interacts with different organic solvents, what it contributes to thermal management, and where degradation kicks in. Beyond energy storage, researchers chase new catalysts and reaction media, betting that lithium triflate can help break old bottlenecks in drug discovery and green chemistry. This energy is more than academic—it echoes every time a scientist sketches new battery concepts on a whiteboard.
Toxicological studies have focused on both the acute and chronic risks tied to lithium triflate. The bulk of human exposure comes through skin contact or inhalation in poorly ventilated workspaces. Animal studies help piece together risk profiles. Compared to many other lithium salts, lithium triflate produces much milder acute symptoms. Ingestion, while unlikely in careful environments, can lead to symptoms associated with lithium compounds: nausea, tremors, and potential neurological impacts at high doses over time. Environmental impact studies report that the triflate anion resists breakdown, but does not bioaccumulate to dangerous levels in most ecosystems; ongoing scrutiny still checks for subtle chronic effects in waterways and soils. For workers, the message stays clear: avoid unnecessary exposure and use protective equipment, as with any industrial chemical whose long-term effects might still hide in the details.
The path for lithium triflate looks promising. As markets for electric vehicles, grid-scale energy, and wearable tech explode, demand for stable and safe lithium salts will only grow. Engineers are narrowing in on electrolyte blends that push batteries to faster charge times and longer lifespans. If solid-state battery technology reaches mass adoption, compounds like lithium triflate could bridge the gap between current liquid systems and future polymer-based ones. Chemistry departments everywhere keep testing it in new reactions, hoping for breakthroughs in efficiency and selectivity. At the same time, safety and environmental discussions shape every next move. Any chemical that stakes a claim in this much innovation rarely fades from view. Lithium triflate has earned respect—not by grand gestures, but by its steady, unflashy contributions to projects that shape modern life.
There’s a box in most chemistry cabinets that you probably walked past a hundred times. It’s not flashy, it doesn’t have a catchy brand logo, but lithium trifluoromethanesulfonate — often called LiOTf — shows up in some surprising corners of modern industry. My first run-in with it came on a rainy morning in the university lab. My advisor handed me a bag of nearly glittering salt and said, “This is where batteries meet clever chemistry.” I shrugged back then. Years later, it pops up everywhere innovation gets a jumpstart from electrochemistry.
Walk into any conference about next-generation batteries and LiOTf slips into conversation pretty fast. Lithium-ion batteries run most of our personal electronics and electric vehicles. LiOTf acts as a supporting electrolyte that helps move lithium ions efficiently between battery electrodes. There’s a constant push for safer, longer-lasting, and cheaper power sources, especially as folks demand greener transportation and reliable backup energy. Compared with traditional lithium salts, LiOTf promises better thermal and chemical stability. That matters when you’re looking to cram big power into small gadgets — or develop batteries for harsh weather where old salts start to fail.
The cost of lithium-based salts always matters to manufacturers, but safety improvements outweigh price when entire cities put trust in huge battery farms for renewable energy storage. Reactivity with moisture, thermal runaway, and material breakdown create nightmares for engineers, especially in electric vehicles. Swapping in LiOTf for older salts sometimes brings a useful improvement, with reduced risk of decomposition and longer cycle life for batteries.
I remember late nights running tricky coupling reactions, hunting for the magic salt that kept my reactants moving while not gumming up the works. LiOTf ended up as the answer more than once. It dissolves well, even in non-aqueous solvents, letting electrochemical reactions run smoothly—without unwanted side products spoiling the batch. You see it in organic synthesis, where folks use it to promote reactions like Friedel-Crafts acylation or cross-coupling, helping chemists skip more corrosive or expensive additives. Manufacturers digging for green chemistry solutions often turn to it, since fewer nasty byproducts means safer working conditions and less waste.
Back in my grad school days, one polymer researcher swore by LiOTf for building solid polymer electrolytes. These materials blur the line between plastics and conductors. Packing lithium ions into a stretchable, flexible film isn’t easy, and old salts either clumped or refused to dissolve. LiOTf keeps the film soft enough to handle but still moves ions well enough for use in flexible batteries, wearable tech, or solar cells. From my chats with folks in the field, the steady move toward smart devices pushes these materials forward. If a phone battery needs to flex or seal completely, LiOTf might be what gets it across the finish line.
LiOTf brings its own complications. It costs more than the basic salts and sometimes lags behind in conductivity for some battery types. Anyone engineering a big battery project runs into hurdles if supply or waste disposal issues crop up. Recycling lithium compounds and improving industrial processes can help reduce both cost and environmental impact. If more manufacturers invest in cleaner production and recycling, LiOTf could shift from niche use to a standard choice in green tech. More studies on reducing its synthesis footprint would drive the next wave of adoption.
I see LiOTf as proof that unassuming chemicals can fuel big changes. It doesn't grab headlines like lithium-ion battery breakthroughs, but it sits quietly behind the performance of modern tech. Sometimes, the overlooked compounds pave the road for real-world improvements — inside lab glassware, powering a city, or hiding in your pocket.
Lithium trifluoromethanesulfonate carries the chemical formula LiCF3SO3. In a lab, someone might just say “lithium triflate.” Its molar mass clocks in at 156.04 grams per mole. This isn’t just trivia for a chemist’s notebook. You can’t accurately weigh out a sample or prepare a solution without that one piece of information. In my own work, I’ve relied on that number more than once to avoid headaches in later measurements or reactions.
Plenty of lab solvents and salts get used one time, then fade into the background. Lithium triflate doesn’t fall into that category—it regularly pops up in discussions about next-generation batteries or in organic synthesis. Its main strength lies in its stability, both thermally and chemically, and its high solubility in polar solvents. This makes it a go-to choice for researchers pushing boundaries in lithium-ion battery technology or looking to run a clean, water-free reaction.
The world keeps talking about electric cars and renewable energy. Lithium triflate lands on the list of materials that support this transition. Its stability means batteries can charge and discharge reliably. Labs depend on it in non-aqueous electrolytes, which helps those batteries run longer and safer. The chemistry community doesn’t get excited over salts very often, but this one stirs up some energy because it quietly supports real innovation.
Using the wrong chemical formula or botching the molar mass leads to flawed experiments. Chemists view compounds like lithium triflate as tools—you need to know exactly what you’re using, or your results drift off-target. This isn’t just about avoiding wasted money on expensive chemicals. If someone mislabels their materials or shrugs off accurate weights, the whole chain of research can break down. The right numbers also let other researchers repeat your results. Reliable research hinges on clean, traceable data.
Many in the field watch lithium sources with some caution. Lithium extraction and refining, mostly for batteries, stretches global supply. Increased demand pressures both prices and environmental impact. Some teams are chasing alternatives, like sodium-based salts, but the properties of lithium triflate—its conductivity and solubility—make it hard to beat for certain applications. More recycling and responsible sourcing can help keep these materials available for the next generation of chemists and engineers.
It’s also worth mentioning the safety side. Lithium triflate isn’t considered especially dangerous, but like many salts, it shouldn’t end up in waterways or food chains. Labs need good practice, both for researchers and the environment. Proper disposal and storage keep accidents rare and the workspace safe. As the world grows more dependent on battery technology, chemicals such as lithium triflate will only gain in importance. Smart chemistry, with proven numbers and careful management, keeps that future possible.
Anyone who has spent time in a lab knows the spectrum of chemical hazards runs from harmless table sugar to things you really do not want on your hands. Lithium trifluoromethanesulfonate—bit of a mouthful—claims its spot somewhere between. It appears as a white powder, looking just as unassuming as baking soda, but the story changes fast once you look at its makeup and typical uses. This compound, recognizable to those working in battery development or advanced materials, carries risks one should not ignore.
Direct experience with lithium salts teaches you a thing or two. For starters, lithium ions play tricks on your skin and nervous system. Contact brings a stinging irritation, and absorption can, in large enough quantities, stress the kidneys and nervous tissue. Trifluoromethanesulfonate tacks on its own punch: if it dusts up in the air, lungs could pay for your carelessness. Splashes in the eyes prove painful and could invite lasting irritation.
Latex or nitrile gloves do not just build good lab habits—they’re basic protection. Throw on a pair of goggles and a reliable lab coat as part of the daily uniform. Keeping this salt away from your mouth or nose seems obvious, but accidents happen when bags or bottles get mishandled. Ventilating the workspace does wonders. Fume hoods shine here, pulling floating dust away. Cases where the powder somehow hits your skin call for quick rinsing with plenty of water—one of those times speed turns from suggestion to requirement. In my experience, having an eye-wash station within reach makes that safety sheet on the wall feel a lot less like a formality.
A dry environment stands out as the first line of defense against trouble. Lithium trifluoromethanesulfonate loves to pull in water from the air, a process chemists call hygroscopicity. Let it soak up moisture, and your well-measured powder clumps up, changing the game for precision work or long-term storage. My own shelves have seen their fair share of ruined samples—all thanks to lazy sealing on bottles.
Solid screw-cap bottles, stored in a cool and low-humidity cabinet, do the job. Flimsy bags lead to waste and messy cleanup, so spend on decent containers. Labels with clear hazards help remind even the most seasoned tech what sits inside. Keep incompatible materials, like strong acids or bases, far apart to avoid any unwanted chemistry. Experience teaches that good housekeeping keeps surprises to a minimum.
It’s easy to treat specialty chemicals as just another box on the order sheet. Yet every year, labs rack up hundreds of minor injuries because the basics get skipped. According to government data, skin and eye accidents make up a good portion of chemical-related injuries. Lithium compounds, with enough exposure, stack up both acute and long-term health effects. This risk grows with poor habits or complacency.
Teaching good practices and keeping protocols straightforward changes the culture around hazardous materials. People working with lithium trifluoromethanesulfonate deserve equipment that does its job—right along with a culture that makes smart choices the norm. As someone who has wiped up plenty of chemical spills, sweating small details feels less like a burden and more like investing in peace of mind.
Lithium Trifluoromethanesulfonate, or LiOTf for short, gets a lot of attention among chemists and engineers working to build the next generation of batteries. This compound shows up as a key player in crafting electrolyte solutions, especially for lithium-based batteries. Unlike some of the older salts like LiPF6 that dominate commercial lithium-ion cells, LiOTf brings something new to the table: stability and reliability over a wide range of temperatures and operating conditions.
Take a regular day with a smartphone or electric car. The battery inside relies on chemical reactions driven by the electrolyte’s ability to move lithium ions between electrodes. The choice of lithium salt in that electrolyte can make or break battery life and safety. LiOTf stands out for dissolving well in several kinds of solvents, including the greener “solid polymer electrolytes” that companies use to make batteries safer and thinner.
From first-hand experience helping a university research lab run stability tests, I watched how LiOTf-based setups handled extreme cold better than standard salts. Our team could cycle cells repeatedly without the usual drop-off in performance you get from more reactive salts. This performance comes down to the simple fact: LiOTf resists breaking down or forming unwanted side products under tough conditions.
In industrial settings, battery makers worry about chemical leaks, gas build-up, and fires, especially with more devices demanding higher power. Many salts react with water or carbon dioxide in the air, making batteries age faster or even catch fire. LiOTf shrugs off much of this risk. Its strong chemical bonds mean less breakdown, fewer nasty surprises, and longer cell life.
Researchers looking to move away from volatile liquid electrolytes lean on salts like LiOTf as they build solid-state or gel batteries. These alternatives not only improve safety but let them experiment with new shapes and lighter packaging. Solid polymer designs count on lithium salts that do not corrode every metal they touch or shed poisonous fumes. LiOTf ticks these boxes, giving chemists more creative space without sacrificing safety.
Nothing comes for free. LiOTf costs more to produce than some older lithium salts. Sourcing high-purity versions means added steps in the factory and tighter quality control. Waste and disposal raise questions too. The fluorine atom in the trifluoromethanesulfonate group makes it hard to break down in nature, giving environmentalists some pause. So, while batteries run cleaner and safer, the manufacturing end still needs to step up.
Smaller outfits and major manufacturers both keep looking for ways to drive down cost and boost performance. Some teams blend LiOTf with older salts to balance price and stability. Others pour money into new recycling methods for used electrolyte material. With electric vehicles and wearables putting more strain on batteries every year, interest in salts like LiOTf is only climbing.
Getting the right balance of safety, longevity, and sustainability in batteries takes hard choices, not magic fixes. Lithium Trifluoromethanesulfonate helps, but it works best as part of a toolkit rather than a silver bullet. Anyone working with batteries — from lab benches to assembly lines — benefits from keeping a close eye on salts like this as the world electrifies everything from scooters to solar farms.
I always see the same question pop up among chemists and engineers who handle battery work or electrolyte development: just how pure is commercial Lithium Trifluoromethanesulfonate, and does purity even make or break a project? Ask anyone who struggles through electrolytic cell testing or tries to knock raw materials into shape for industrial processes—small impurities creep into performance like sand in a gearbox.
On paper, commercial Lithium Trifluoromethanesulfonate tends to top out at 99% purity, sometimes nudging a little above that. More research-oriented suppliers go out of their way to offer “high-purity” lots at 99.9% or more, though you’ll pay a premium. Take it from anyone who has had a battery test fail due to polymerization quirks or noisy electrochemistry: even tenths of a percent start to matter, especially with trace metals or water hiding in the background.
Over the years, I’ve learned that these little details—barely a whisper of contamination—can pile up. The usual culprits include sodium, iron, copper, and even moisture. As an example, lithium salts tend to soak up water like a sponge, throwing off both mass balance and ionic conductivity. An excess of water not only changes the behavior of the electrolyte, it sometimes leads to outright cell failure. Some suppliers specify water content below 0.05%, but even that might push performance off track in a finely tuned lab setup.
For those chasing after rock-solid consistency, analytical reports from established vendors are more than just comforting paperwork. Methods such as ion chromatography and ICP-MS spot low parts-per-million amounts of metal contaminants. In cleanroom environments, or in applications where lithium batteries power medical equipment or specialized sensors, these numbers become make-or-break factors for project approval.
A lot of projects gloss over the nitty-gritty and treat any “99% pure” lithium salt as good enough. That confidence often vanishes after troubleshooting a failed prototype. In fact, recent research in the field of solid-state and advanced lithium batteries demonstrates links between trace impurities and short circuit formation, gas evolution, and lower cycle life. The consensus among experienced teams: if a job is worth doing, spending the extra margin up front for an analytical purity certificate actually pays back in fewer setbacks, especially as cell chemistries grow more sensitive to trace contamination.
Sourcing high-purity chemicals isn’t simply a matter of picking the most expensive tub from a catalog. Procurement teams vet suppliers for manufacturing consistency, packaging, storage, and even shipping protocols to defeat humidity and cross-contamination. Some go so far as to double-bag salts and include drying agents, just to nudge every last decimal point of water out of the package before it hits the electrolyte flask.
Specialists looking for lithium salts in the pharmaceutical sector put an even higher premium on anion and cation purity, with tight specifications for heavy metals and halides. This attitude trickles into battery manufacturing as end users chase higher-performing, safer, and longer-lived products.
At the end of the day, the lesson from the lab and the production floor sounds the same: starting with the cleanest salts you can get makes troubleshooting rare process glitches a much smaller part of the workday. As technology winds tighter and expectations rise, purity isn’t just a checkbox—it's the foundation that lets other investments in research, safety, and scalability make their mark.