Hexafluoroisopropanol, or HFIP, entered the chemistry scene in a big way during the rapid expansion of fluorine chemistry in the twentieth century. As researchers searched for solvents capable of breaking through the limitations of older materials, HFIP emerged. Its roots trace back to curiosity about how fluorination can change alcohols, and HFIP’s unique profile comes from packing six fluorine atoms around a central isopropanol backbone. Chemists couldn’t ignore the way HFIP stood apart for dissolving compounds most other solvents rejected. Over time, labs shifted away from legacy alcohols and ether-based solvents for certain reactions and analyses, favoring HFIP’s exceptional solvation and hydrogen bonding abilities. In pharma, materials science, and synthetic labs, that switch speaks volumes. From my own work in organic chemistry labs, hexafluoroisopropanol gained a reputation as a go-to solvent in peptide synthesis, especially when standard alcohols failed to deliver. The learning curve remained steep, but the payoff for mastery turned out to be significant.
HFIP doesn’t look all that intimidating—a clear, mobile liquid with an unmistakable pungent smell. It boils at around 58°C, with a melting point near -4°C. What makes it stand out has less to do with appearance and more with what happens in solution. Sporting a high dielectric constant and both hydrophobic and hydrophilic characteristics, HFIP can dissolve proteins and fluorinated polymers alike. The hydrogen bonding power makes it a favorite for denaturing biological macromolecules, which explains its starring role in protein folding studies and advanced NMR sample prep. Its acidity, with a pKa near 9.3, changes the way it interacts with basic and acidic species—it’s not your everyday alcohol. Across the board, labs learn quickly that the slight change in water content or temperature will shift reaction rates and selectivity. These lessons often come not from textbooks but from the frustration (and eventual discovery) at the benchtop.
Buying HFIP involves filtering through purity grades targeted for research, pharmaceutical development, or materials manufacturing. Most suppliers offer it at above 99% purity, and water content plays a large role in suitability for sensitive applications. Storage containers carry fluoropolymer coatings or specialized glass to hold back its reactivity—aluminum and unlined steel fail within weeks. The bottles require tightly fitting lids and dry storage because HFIP picks up moisture from humid air. Labels warn about flammability and aggressive action toward many plastics. In my experience, mislabeling or sloppy storage led more than one synthetic project astray after traces of water or untracked contamination led to unpredictable results.
Industrial production of HFIP usually draws from hexafluoroacetone and hydrogenation using specialized catalysts and controlled pressure. This reaction calls for real precision—any deviation brings a cascade of unwanted fluorinated byproducts that cost both time and money to remove. For those few labs set up for small-scale prep, most avoid making it from scratch and instead rely on commercial sources. Trying to cook it up without the right gear risks both personal safety and expensive regulatory headaches, not to mention unpredictable yields. Add this to the need for chemical recycling and environmental monitoring, and one quickly sees why most choose to have specialty suppliers do the heavy lifting.
HFIP’s power goes beyond acting as a solvent—it actively changes many reaction landscapes. It’s familiar in rearrangement reactions, oxidations, and as a promoter for photochemical transformations. Its tendency to stabilize carbocations or radical intermediates brings certain rearrangements or cyclizations into play that might otherwise stall. Tattersall’s textbook synthesis of certain steroids, for instance, finds HFIP opening reaction channels not available with either methanol or acetonitrile. The underlying theme comes through experience: running a reaction in HFIP pushes both yields and selectivity in directions not always obvious from theoretical models or initial screenings. That creative control fuels a lot of discovery but also requires lab workers to pay extra attention to safety and waste disposal because of HFIP’s strong solvent action on conventional lab materials.
Within chemical catalogs, HFIP lives under several names—1,1,1,3,3,3-Hexafluoro-2-propanol, Hexafluoroisopropyl alcohol—and many seasoned chemists refer to it simply as “HFIP” to avoid confusion during research planning and procurement. Still, knowing its various tags helps avoid costly purchase mistakes, especially for newer colleagues wading through distributor websites or shared inventories. Miscommunications can and do lead to shipment delays, and sometimes cause work halts in fast-paced settings where time costs real money.
Working with HFIP means taking safety dead seriously. HFIP evaporates quickly, filling the air with vapor that can irritate eyes, nostrils, and the respiratory system. It burns easily and should stay away from open flames or sparks. Gloves resistant to solvents—usually nitrile or fluoroelastomer—provide a bare minimum of protection, and spills need quick cleanup with suitable absorbents to keep workplace air quality in the acceptable range. Fume hoods aren’t optional; they’re an operational requirement. Some colleagues prefer full-face shields for larger transfers, knowing that splashes sting and may cause longer-term skin issues if ignored. Fire departments and hazmat teams usually list HFIP among their top-tier flammable hazards, and labs that follow routine fire drills fare best under review. On a personal level, learning to respect HFIP pays off, since inhalation and skin exposure cause headaches and nausea even at concentrations beneath regulatory thresholds.
Few solvents generate as much loyalty among research groups as HFIP. In medicinal chemistry, its role in peptide unfolding and cleavage reactions stands nearly unchallenged. Polymers researchers rely on it to dissolve stubborn fluorinated backbones that resist nearly everything else. Textile and fiber innovators turn to HFIP to spin synthetic silks and high-performance fibers such as Kevlar. The solvent’s solid track record also includes enzymatic studies, hydrogen bond characterizations, and the isolation of membrane proteins. My own group found HFIP crucial to mapping protein secondary structure using NMR, especially in cases where other solvents left us with only broad, useless peaks. As research deepens, new uses keep popping up, from fuel cell development to new hybrid materials, and more focus lands on designing less hazardous but equally effective replacements.
Academic and industrial efforts focus on fine-tuning both the synthesis of HFIP and its downstream applications. More groups look into how controlled molecular environment in HFIP leads to reproducible reaction outcomes and better understanding of non-covalent interactions. Some funding agencies carve out special calls for safer, more sustainable solvents, so HFIP’s story includes both an appreciation for its utility and pressure to cut down on toxicity and environmental impact. Green chemistry targets come into sharp relief as researchers chase both functional utility and better safety scores. Labs start trialing micro-recycling systems for solvent recovery, evaluating whether HFIP’s short life cycle in the reaction flask really needs to end in waste disposal tanks. Some notable developments include catalyst systems that function in more benign solvents while delivering HFIP-level performance, but the leap to large-scale replacement remains tough.
The story around HFIP toxicity grows more complicated with each new study. Researchers flag issues like acute inhalation effects, chronic organ damage, and environmental persistence. Low-dose exposure causes nervous system stress, and accidental spills in poorly ventilated rooms linger in memory, thanks to coughing fits and headaches that drive home just how strong this compound is. Evidence is mounting that HFIP’s degradation products don’t break down as quickly as once hoped, which pulls in new scrutiny from regulators and green chemists alike. Animals exposed to HFIP show signs of liver and kidney toxicity, which means industry and academia can’t ignore these findings. My own experience working with regulatory teams taught me that even secondary exposure through waste streams triggers questions about long-term aquatic toxicity, putting pressure on organizations to step up monitoring and reporting.
Looking to the future, it’s clear the days of unconstrained HFIP use are numbered. Ongoing research pressures manufacturers to fine-tune production methods and invest in capture, recovery, and recycling tech. Green chemistry guides push for molecules offering the benefits of HFIP with less baggage—better biodegradability, lower toxicity, and reduced environmental footprints. Regulatory agencies grow more vocal in pushing for life-cycle analysis and end-of-life tracking. From my vantage point, plenty of opportunity emerges for chemists willing to bridge the gap, whether that means creating next-generation solvents or developing processes to limit release and maximize recovery of today’s HFIP stocks. Those who’ve spent years wrangling with HFIP’s quirks see the double edge: extraordinary performance, balanced by mounting concerns that demand serious action now rather than later.
Most people never hear about hexafluoroisopropanol at all—unless you happen to spend your days in a research lab or a specialized chemical plant. In those places, folks talk about it like a daily staple, and for good reason. This chemical, which often goes by HFIP, pushes science ahead in more ways than one might guess. The name doesn’t roll off the tongue, but its uses matter to all kinds of innovation.
I first ran into hexafluoroisopropanol as a curious undergrad in the university chemistry lab. You recognize the distinct odor before you see the label. One day we worked with certain polymers, and nothing dissolved them quite like HFIP. In the world of manufacturing high-performance fibers—Kevlar and similar materials—HFIP acts as a go-to solvent. Think about bulletproof vests or the thermal suits astronauts wear. That tough, flexible material starts as a powder. HFIP gets involved in spinning those fibers, making them even and manageable for weaving into life-saving fabric.
This chemical doesn’t just punch above its weight with polymers. Pharmaceutical researchers also put HFIP to work when they want to build complex organic molecules. It helps shape complicated compounds, some of which turn up in hospital IV bags and pill bottles. About ten years ago, a close friend was on a team at a biotech startup; they kept HFIP stocked for this exact reason. It helps certain reactions happen faster, letting new medicines come to market more quickly and reliably. The compound can influence the very way scientists design molecules, especially where every atom counts.
HFIP’s story goes further than material science and medicine. In analytical chemistry, it steps in for researchers using advanced tools like liquid chromatography-mass spectrometry. Understanding proteins creates new opportunities to treat diseases that hit millions of families. Here, HFIP helps scientists separate and identify proteins by interacting with them at the molecular level, offering sharper results and more reproducible data.
Its high polarity, hydrogen bonding, and volatility make it a unique choice in certain reactions. These chemical properties open doors for innovation in fields as diverse as environmental monitoring, microelectronics, and synthetic biology. Anyone who’s tried and failed with common solvents knows the frustration. HFIP offers a solution where ordinary methods stall.
No one calls hexafluoroisopropanol perfectly safe. Handle it wrong, and it’ll harm eyes, lungs, skin. It is a nasty chemical if it’s not treated with respect. Safety goggles and fume hoods become non-negotiable. Measures to reduce human exposure and keep air, soil, and water clean can feel inconvenient, but turning a blind eye to risks leads to stories that never end well. Chemical manufacturers are taking steps to minimize emissions and improve worker safety through automation and sealed systems. Finding greener alternatives becomes important as sustainable chemistry keeps gaining ground.
HFIP’s impact stretches further than a single experiment or research paper. It keeps advanced science moving forward—but that progress relies on researchers using it responsibly. Investing in safer handling, supporting transparency, and encouraging the search for less toxic alternatives is the right way forward. In the meantime, this chemical remains a powerful tool for those building the next generation of materials and medicines—one cautious, well-informed step at a time.
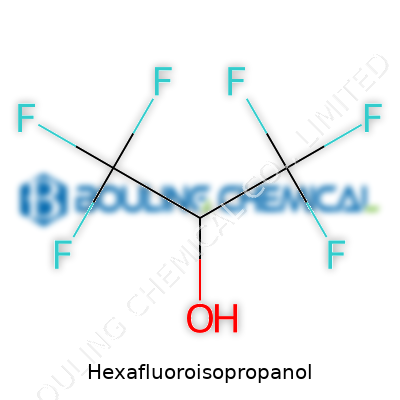
Hexafluoroisopropanol, or HFIP as chemists call it for short, brings a punch to molecular design. The chemical formula, C3H2F6O, might look cryptic on paper, but behind those nine atoms hides a rare versatility. Its structure sets it apart: a central carbon anchors two trifluoromethyl groups (CF3) and a hydroxyl group (-OH). Picture a compact, three-carbon backbone, loaded with fluorine atoms, making this molecule both powerful and a bit unpredictable compared to simple alcohols.
Let’s get real – chemistry textbooks love neat diagrams, but working with HFIP in the lab slaps a whole new appreciation for its unique arrangement of atoms. The CF3 groups beef up the molecule, yanking electron density mightily toward themselves. That makes the alcohol's hydroxyl group unusually acidic, packing a punch more like a carboxylic acid rather than your typical booze (think rubbing alcohol—nothing like this!). This sharp acidity shows up where you least expect it. When you’re dissolving stubborn biopolymers like silk or cellulose, HFIP breaks bonds that most chemicals can't budge.
Years ago, I watched colleagues struggle to dissolve certain polymers until someone pointed to a dusty bottle of HFIP. Like magic, protein powders vanished, and the lab picked up momentum. Now, the textile and pharmaceutical fields rely on those same properties. HFIP gave researchers the keys to synthesize new fibers and peptide-based drugs. It’s a game-changer for companies working on smart clothing, super-strong threads, or even new drug delivery systems.
You can’t spend time around HFIP without noticing its sharp, biting odor—the kind that tells you you’re breathing something you shouldn’t. Its volatility comes with risk, as does its reputation for flammability and potential to cause irritation or worse. Studies point to fluorinated compounds persisting in the environment, and HFIP shares this stubbornness. I’ve seen labs lock down disposal protocols because no one wants these molecules leaching into soil or water, a worry that’s growing louder among environmental scientists.
People in the field need to double down on safe uses and smart alternatives. Labs can upgrade their ventilation and training, but that doesn’t tackle persistent contamination. Some universities have started programs to recover or recycle HFIP after use. Supporting greener synthesis and real accountability in handling this chemical could limit long-term fallout. It makes sense for scientists and industry to join forces, share data, and nudge policy toward safer practices without shutting down the innovations HFIP unlocks.
Innovation rarely runs without friction. The formula and structure of hexafluoroisopropanol pack a punch that few other alcohols match, opening doors in research and industry that once seemed locked tight. Harnessing its benefits while respecting its risks—this is the chemistry that really counts.
Hexafluoroisopropanol, usually called HFIP by chemists, pops up often in labs. It helps scientists dissolve tough molecules and break chemical barriers, so research keeps moving forward. Walk into a research facility, you’ll find this stuff around where people study proteins or create new materials.
You start to wonder about its safety after seeing all the warnings on the bottle: gloves, goggles, don’t breathe it in, keep away from heat. A wiff in the air can irritate your nose and throat. A splash on bare skin stings pretty badly. It evaporates quickly, and a spill fills a room with pungent fumes. Look up accident stories online, you’ll see medical journals warning of anything from headaches to chemical burns.
Long hours working with chemical cocktails convinced me some compounds demand extra respect. HFIP is one of them. The way it’s built — with plenty of fluorine atoms — makes it powerful, but this also has a price. The human body has little defense against these chemicals. Breathing too much can mess with your respiratory system and central nervous system. Prolonged exposure brings risks ranging from severe eye damage to deeper organ injury. A few years ago, a colleague developed a persistent cough after cleaning up a small spill with only a regular mask, which shows how easily exposure happens outside ideal conditions.
On the environmental side, those same fluorine atoms raise different red flags. HFIP resists breaking down in water or soil, so it lingers. If it escapes from a lab sink and flows into local waterways, both aquatic life and the wider food chain might face consequences. Fluorinated chemicals have a reputation for sticking around, like PFAS, sometimes called “forever chemicals.” Long-term buildup — even in small amounts — can affect fish and contaminate drinking water supplies, with effects that nobody fully understands yet.
Companies and research labs already follow strict storage and usage rules for HFIP. In the United States and Europe, chemical safety laws demand that workers wear personal protective equipment and ventilate work spaces thoroughly. There’s still a gap though. Disposal rules sometimes lack detail. Regulators focus on immediate splash or inhalation hazards without always thinking about long-term buildup downstream.
It’s useful to point out that scientists are collecting more data about how much HFIP escapes into the environment and how long it lasts. Agencies keep updating their guidance as new facts come in. Still, without clear rules about what to do with HFIP waste, people sometimes flush dilute leftovers down the drain. This routine habit risks creating bigger environmental headaches years down the road.
Research and industry both rely on HFIP, but nobody wants the health hazards or the mess it can leave behind. Switching to safer solvents would help, but few options work as well for some jobs. We need stiffer disposal rules and more investment in “green chemistry”— safer molecules that get the job done without sticking around in rivers, lakes, or lungs. Regular training and strict lab culture help cut down on accidental exposure. In the big picture, the solution means recognizing that every shortcut—every quick dump of leftovers—has a cost, paid in pollution or illness. Protecting ourselves and the world now means handling HFIP with both caution and care, even if that slows the day’s work.
Ask anyone working in a chemistry lab about Hexafluoroisopropanol and they’ll probably tell you the same two things: the stuff works wonders for dissolving stubborn polymers, and its hazards command respect. It’s not just an abstract chemical formula found in a textbook—it’s a liquid that demands responsible hands. Often, storerooms tuck it away behind extra locked cabinets, and with good reason.
Breathing in its fumes feels harsh. Even brief exposure leaves a noticeable burning in the nose, throat, and eyes. Anybody who has poured it without proper ventilation only does that once. The danger jumps higher if it gets onto your skin; chemical burns happen quickly. Water alone won’t offer a comfortable fix. Toxicity concerns don’t just sit in the pages of technical manuals. The reality hits if you skip gloves or treat ventilation like an afterthought.
The best way to store this chemical isn’t just about putting it on the right shelf. Safety depends on keeping it somewhere cool and away from sources that could spark, like hot plates or light switches. Its vapors ignite more easily than most folks expect. Once, after watching a careless technician store it beside an oil heater, everyone in the lab developed a new respect for the sudden danger those vapors can bring.
Fluorinated solvents sound high-tech; they still pour like trouble if handled with old habits. Over the years, the safest setups I’ve seen involve chemical-rated cabinets with clear signage and sturdy spill trays underneath. That’s not overkill. It shows an understanding that accidents start with small missteps—a leaky cap or a distracted hand, not some Hollywood-style explosion.
There’s a reason experienced chemists insist on full-length sleeves and heavy gloves. One lab I visited relied on thin nitrile gloves, thinking it was enough. A splash burned through quickly. Using PPE isn’t about checking off a requirement; it’s about trusting experience. Chemical goggles make sense, even if nobody ever looks cool in them.
Labeling becomes your safety net after the bottle gets put back on the shelf. I’ve seen lookalike containers cause near-misses, where tired hands reach for what looks like simple alcohol and end up holding something much worse. Clear, bold labels and updated inventories matter every day, not just after an inspection.
Regular checks keep problems small. Spotting corrosion on caps or catching a forgotten spill in a storage tray means acting before trouble gets out of hand. Training shouldn’t stop with the oldest workers—new lab members bring risks if skipped over for “later.”
Good ventilation, reliable personal gear, fire safety plans, and clear communication all make the difference with chemicals like Hexafluoroisopropanol. Safety shouldn’t rely on luck or personal memory. It comes from treating true hazards like real risks and using habits that protect everyone in the room, not just the person pouring from the bottle. Nothing beats direct attention—and the willingness to speak up if something seems off.
Step inside any polymer chemistry lab and you’re bound to find one thing lurking in a cupboard—hexafluoroisopropanol, or HFIP. The bottle always carries stern hazard labels, but ask anyone working with stubborn polymers and they’ll tell you how this solvent makes life a lot easier. I remember struggling for hours to dissolve a chunk of polyamide in the usual suspects: water, methanol, even DMF. HFIP came out, and minutes later, problem solved. That’s not magic. That’s strong hydrogen bonding and a knack for breaking up even the toughest macromolecular tangles.
Go beyond plastics, and HFIP quickly pops up in protein labs as well. Scientists use it for dissolving and refolding tricky peptides and amyloid proteins, such as those linked to Alzheimer’s research. The reliable denaturing power tears apart protein aggregates that other solvents won’t touch, allowing for clean spectroscopic studies. Years back, during my own research, I saw graduate students breathe a sigh of relief after hours of failed attempts to get a peptide sample into solution. HFIP cut the struggle short, and the team moved ahead with their NMR and mass spectrometry analyses.
Polymers aren’t just for plastic bags. They’re embedded in drug delivery, soft robotics, and electronics—all fields that depend on solvents that can handle complex compositions. HFIP stands out for spinning polyamides and biodegradable scaffolds, especially when precision matters, like in electrospinning fine fibers for tissue engineering. Research shows that HFIP gives scientists the power to tailor fiber diameters and morphologies that other solvents just can’t match.
HFIP does more than just dissolve. Around organic benches, chemists value it for its influence on reaction mechanisms. It boosts electrophilicity and stabilizes elusive intermediates, which speeds up reactions or opens up new pathways. Reports in reputable journals highlight how reactions, such as Friedel-Crafts acylations and C–H activations, run with higher yields and selectivity simply by swapping in HFIP. It can mean fewer by-products and less purification headache later on.
None of these strengths mean HFIP is a dream chemical. It’s pricey, volatile, and definitely not the friendliest for waste streams. Environmental rules keep tightening around solvent disposal, and with HFIP’s persistence, no one wants to be careless. In my own lab, we switched over to greener solvents for routine jobs and reserved HFIP only for stubborn cases. A growing movement in chemistry pushes for alternatives whenever possible, and researchers keep working on new solvent systems that blend HFIP’s power with a smaller ecological footprint.
Fundamentally, HFIP serves as a lesson in balance: revolutionary in the right hands, but heavy on safety and sustainability concerns. As industries and universities reflect on their carbon footprint and solvent waste, adopting HFIP responsibly—knowing when it’s truly the best option—becomes part of the daily routine. The chemical’s story really comes down to smart choices, a healthy respect for risk, and keeping an eye out for the next big breakthrough in green chemistry that might finally give HFIP a run for its money.