The journey of hexafluorobutadiene traces back to a period marked by rapid advancements in both organic and inorganic fluorine chemistry. Early chemists saw the promise of fluorinated compounds due to their thermal stability and chemical inertness. As electronics and aerospace engineers began searching for materials capable of handling high voltages and harsh environments, compounds like hexafluorobutadiene entered the spotlight. Its introduction wasn’t just the result of academic curiosity—industrial demand for etchants in semiconductor fabrication drove investment and research. While early methods relied on multi-step synthesis with yield and purity challenges, persistence paid off as the field learned to tame the stubborn reactivity of elemental fluorine and unsaturated hydrocarbons. This compound, often abbreviated as C4F6, now stands as a testament to decades of hands-on laboratory work and trial-and-error tinkering, as much as the technical innovation that defines the chemical industry.
Just about anyone working in plasma etching or advanced lithography knows hexafluorobutadiene as a specialized tool, rather than a household name. It rarely shows up in everyday life, yet its fingerprints are found in the manufacturing processes behind microchips and flat-panel displays. Its main appeal rests on how it delivers precise results during plasma etching—removing thin films and structures at scales invisible to the naked eye, but critical to device performance. At room temperature, it’s a clear, colorless gas, stored in cylinders at moderate pressure, with a faint, sweet odor. It distinguishes itself from less-fluorinated relatives by greater chemical inertness, making it a favorite in applications where other etchants prove too reactive or messy.
In terms of the basics, hexafluorobutadiene comes with a boiling point just shy of room temperature, making it easy enough to handle as a condensed liquid but often encountered as a gas. Its chemical structure, consisting of a four-carbon chain socked with six fluorine atoms, leads to high density and low flammability. It stands firm against attack from acids and bases, a trait thanks to the stability the fluorine atoms provide. This stability allows it to participate in plasma reactions without falling apart prematurely, but also means it resists chemical degradation in storage. Its low reactivity at room temperature pairs with high reactivity in electrical discharges—exactly what process engineers desire in an etching gas.
In a laboratory or semiconductor fab, you won’t find hexafluorobutadiene sitting around unlabeled. It ships under strict labeling to meet international standards, with hazardous materials codes reflecting toxicity and environmental risks. Cylinders contain pressure and purity details, as trace impurities wreak havoc on chip fabrication. Even small differences in feedstock purity lead to flaws in etched circuitry, driving demand for analytical verification at every stage. Handling practices rely on clear and legible information, warning any user about both immediate risks and the procedures required for safe transfer and usage.
Making hexafluorobutadiene seldom follows a straightforward path. The traditional approach favors direct fluorination of butadiene or partially fluorinated intermediates. Elemental fluorine, notoriously reactive, demands brave chemists and robust lab setups. Specialized reactors cool and dilute the action, limiting runaway reactions that threaten yield and safety. Other synthesis variants employ photochemical or catalytic methodologies aiming for milder conditions and cleaner output. In each case, scrubbing and purification steps remove byproducts and excess fluorine, producing a product stringently clean for industrial needs. Process improvements never cease; as the stakes rise with smaller semiconductor nodes, companies invest heavily in refining these methodologies to cut down waste, energy usage, and costs.
Hexafluorobutadiene keeps a low profile outside plasma processes, and rightly so. Its double bonds, masked by fluorine armor, make most classical organic reactions sluggish or unproductive. Under the right conditions, like strong UV light or in the guts of a plasma reactor, it fragments—producing an array of reactive fluorocarbon species that etch silicon or silicon oxide surfaces with surgical precision. Chemists have tried coaxing it into cross-coupling or polymerization reactions, but the yields and practicality usually lag behind better-suited starting materials. Modification efforts remain a niche pursuit, mostly in the context of fluoropolymer research or attempts to recycle spent etchant streams.
This compound goes by a few names, most commonly hexafluorobutadiene, 1,3-Butadiene, hexafluoro-, and various identifiers used in international chemical inventories. In trade circles, abbreviations like C4F6 serve as shorthand, while researchers may cite more systematic nomenclature or CAS numbers depending on the context. These synonyms cause confusion only rarely, since its use case and safety precautions keep it locked within specialist domains.
Safety isn’t just a checklist item—it’s a daily mindset for anyone using hexafluorobutadiene. Leaks pose an inhalation hazard and, in confined spaces, the risks stack up quickly. Regulations require exhaust ventilation, gas monitoring, and emergency protocols with trained personnel on hand. Protective gear, including goggles and gloves rated for fluorocarbon exposure, stands as the norm, not the exception. The legacy of early mishaps in fluorine chemistry has made industry leaders especially vigilant. Engineers install redundant containment and automated shutoff systems; new hires receive extensive orientation before touching a cylinder. Disposal challenges arise due to high chemical stability—thermal oxidation under controlled conditions stays as the standard route to minimize environmental release.
Most discussions about hexafluorobutadiene circle back to microelectronics and semiconductor fabrication. Plasma etching processes demand gasses that can create fine patterns at the nanometer scale, and this molecule delivers reliable, clean results with minimal collateral damage to delicate structures. It also finds use in the production of flat panel displays, where its unique reactivity profile sets it apart from older etching gases that generate harmful byproducts. Small research streams test its utility in synthesizing advanced fluoropolymers, though widespread adoption faces hurdles related to both cost and process simplicity. Broader industrial usage remains limited, making this compound a specialized solution rather than a commodity chemical.
R&D efforts around hexafluorobutadiene move in several directions. One camp hunts for better, more sustainable synthesis routes, tired of the risks and waste created by direct fluorination. Another segment chases more efficient plasma etching chemistries, targeting lower energy consumption and superior selectivity, with fewer residues left on chips and reactors. Collaborative projects see chemists, physicists, and engineers running trials with new gas blends, looking to push the limits of device miniaturization. Environmental research teams dig into the atmospheric fate and breakdown mechanisms, recognizing that even trace releases in production or disposal cycles can challenge air quality standards. Universities and corporate labs alike face pressure to publish and patent greener, safer process tweaks, turning what might seem a niche product into a proving ground for broader trends in chemical and materials science.
The dark side of progress shows up in the toxicological profile of hexafluorobutadiene. Early animal studies pointed out lung and liver toxicity at high concentrations, findings that underline the need for rigorous workplace exposure limits. Chronic exposure data remains sparse, though repeated inhalation experiments in rodents showed adverse systemic effects. Environmental persistence pops up in regulatory literature as a concern; scientists flagged its potential to resist breakdown and migrate through air or water once released. Monitoring programs search for breakdown products, some of which bear a strong resemblance to persistent organic pollutants. The industry response involves heavy containment, personal protective equipment, and active gas monitoring—not just to comply with rules, but out of a learned respect for the compound’s risks. Calls for more thorough epidemiological and ecological studies grow louder, tracking with rising awareness about chemical exposures and long-term health.
Anyone betting on semiconductor and electronics growth sees hexafluorobutadiene playing a role for years to come, even as process engineers seek alternatives for safety and sustainability reasons. Demand for ever-thinner, more complex devices increases pressure on suppliers to deliver ultra-pure etching gases. At the same time, regulatory agencies tighten rules around fluorocarbon emissions and toxic byproducts, challenging the industry to innovate or face new barriers. Promising research looks at tailored etchant blends, reclaim and recycling technologies, and downstream destruction methods to cut overall environmental impact. The global move toward greener manufacturing pushes all stakeholders to examine every input, and hexafluorobutadiene’s future will hinge on balancing unmatched technical performance with practical steps to limit exposure and environmental release. That tension between performance and responsibility echoes throughout many high-tech industries, forcing both incremental tweaks and bold rethinks.
Hexafluorobutadiene doesn’t grab headlines the way gold, cobalt, or lithium do. Still, its influence runs deep in the world of electronics. This colorless gas pops up in conversations about plasma etching for semiconductors. As someone who relies on digital devices every day, I notice how easily people overlook what goes into making those speedy chips and memory cards. Hexafluorobutadiene plays a key part in carving microscopic circuits onto silicon wafers. It helps manufactures control feature size, reduces material loss, and lets chips run cooler and faster.
Semiconductor fabs treat gases like hexafluorobutadiene almost as specialty tools. I’ve watched engineers handle these tanks with a blend of respect and caution—this is not stuff you want escaping unfiltered into the air. Chlorine and other etching gases have been around longer, but hexafluorobutadiene allows for more precise, less damaging etch profiles. If you’re carrying a sleek smartphone or working at a high-powered laptop, there’s a real chance this gas contributed to the sharpness of those transistors. Devices keep shrinking, from seven-nanometer chips down to three or smaller, and that’s only possible with the right etching chemistry backing it up.
High performance often bumps heads with high risk. Hexafluorobutadiene belongs to a group of chemicals famous for their global warming potential. A big worry with perfluorinated compounds: they don’t break down easily in the atmosphere. I’ve read about strict regulations and extensive filter systems in chip fabs, designed to suck up and destroy these gases before they slip into the outside world. Even so, small leaks or poor waste management can send this compound drifting out, where it sticks around for years. As the semiconductor industry grows, especially in places ramping up chip production, these emissions need attention—just as much as the final product’s speed or memory size.
Over the past decade, companies started exploring alternatives. Some test new plasma gas blends that do the job with less impact on the climate. Others add gas abatement units or recapture systems to scrub exhaust streams before release. There’s room for improvement, and change happens fastest when governments, industry leaders, and local communities push for better answers together. Consumer awareness helps, too. If you care about greener gadgets, it’s worth asking what lies behind the glossy screen.
Technology brings comfort, entertainment, and connection, but also raises questions about long-term consequences. The story of hexafluorobutadiene shows that small details, like an invisible etching gas, shape our future in ways most people never notice. For anyone invested in cleaner technology and responsible manufacturing, understanding these silent contributors is just as important as chasing the latest phone upgrade.
Anyone who’s set foot in a lab knows that familiarity with chemicals can become a silent risk. Hexafluorobutadiene might not be a household name, but in my own years of working alongside chemical engineers and lab technicians, I’ve learned to respect substances that have both industrial utility and significant hazards. People often think protective gear creates barriers or just slows things down, but with a compound like hexafluorobutadiene, skipping steps can bring about emergency room visits or worse.
Hexafluorobutadiene is not something to handle casually. The gas is colorless and might not give an immediate warning if there’s a leak. Inhalation poses real dangers: irritation, dizziness, and lung damage. Since it’s heavier than air, the gas tends to settle in low-lying spaces, which can lead to unexpected build-up. Fires caused by reactive chemicals leave scars, both physically and psychologically, even if the event never hits national news. Personal experience sticks longer than any training video. This is why I never treat safety instructions as fine print.
No one wants to look like an astronaut every day, but gloves, eye protection, and flame-retardant lab coats are more than just a checklist. I once saw a careless slip—someone reached for an unmarked flask and the splash sent a colleague to urgent care with chemical burns. Chemical goggles and face shields guard against splashes, and gloves designed for fluorinated gases block exposure. Respirators with the right filters—approved for acid gases—become essential if ventilation fails. Sometimes, just noticing that a glove has a small tear can make the difference between a routine experiment and disaster.
Proper airflow changes everything when working with hazardous gases. Fume hoods weren’t invented as decorations; they literally pull danger out of the breathing zone. In my lab days, every technician learned how to inspect airflow—holding a strip of tissue at the opening, feeling for suction, not just taking someone’s word for it. If something smells odd or you feel a draft where there shouldn’t be one, shut things down and investigate. Leaks can hide, and it takes only a small lapse for things to get out of hand.
Keeping hexafluorobutadiene in tightly sealed, corrosion-resistant cylinders works only if those cylinders stay upright and anchored. Never store reactive chemicals next to flammable materials or near heat sources. I remember a time someone moved a gas cylinder too close to a steam pipe—luckily, the mistake was caught before any reaction, but nobody ever forgot that training session. Regular inspections and clear labeling make it so newcomers and veterans both know the score at a glance.
Not everyone can memorize the emergency protocols from a safety manual, but drills and hands-on training always pay off. I once attended a simulated leak drill, and the real takeaway came from the adrenaline of trying to reach an exit before the hazard “spread.” Practicing these scenarios cements a respect for the process and builds teamwork. Keeping a spill kit and emergency eyewash stations within sight isn’t paranoia—it’s preparation.
The people I trust most in a lab are those who call out shortcuts and double-check each other’s work. Rewarding safe habits, sharing stories of near-misses without shame, and giving everyone a voice shape a place where safety doesn’t take a back seat. The value of these habits goes beyond compliance—it sets a standard worth living up to. Handling hexafluorobutadiene isn’t just a technical problem; it’s a test of collective responsibility and care for each other’s well-being.
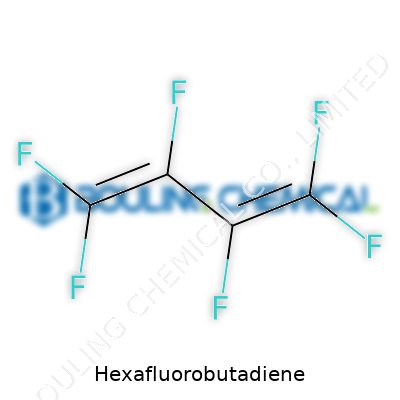
Hexafluorobutadiene carries the formula C4F6. This tells you it’s a hydrocarbon backbone where all the hydrogen atoms got swapped for fluorine. The actual structure unfolds as a four-carbon chain with alternating double bonds, so its name maps out as 1,3-butadiene but cloaked in fluorine. You get a molecule with this arrangement: F2C=CF–CF=CF2. The double bonds don’t just look good on paper. They deliver a lot of reactivity, especially for industries working with electronics and high-performance polymers.
Working in chemical research, the detail that always stood out about hexafluorobutadiene goes beyond the textbook structure. Its all-fluorine exterior sets it apart. These fluorine atoms make the molecule both stable and hard to react with anything less reactive than lightning. This tends to show up in microelectronics fabrication. The gas is popular for plasma etching, the process that carves out tiny patterns on silicon wafers to create integrated circuits. If you’ve ever worked with semiconductors, you know the difference the right etchant makes—from sharper lines to fewer defects. It helps save time and reduce waste, qualities every engineer and technician seeks on the factory floor.
There’s a tug-of-war between performance and safety. Hexafluorobutadiene moves fast in reactive environments but doesn’t play nice with human health. Studies note that inhalation brings real hazards—respiratory issues and the risk of central nervous system damage. The OSHA guidelines don’t mess around when it comes to exposure limits. These reasons mean you’ll only find trained personnel working with it, housed in tightly controlled setups with plenty of extra precautions.
From supply chain consulting work, one lesson stands out: specialty gases like hexafluorobutadiene don’t thrive in fragile systems. Purity demands stretch resources thin. Each cylinder moves through strict controls, adding costs along the way. When disruptions hit—shipping delays, natural disasters, or sudden demand spikes—pricing can leap up and projects stall. This forms a real pain point for manufacturers, especially those scaling up chip production. Every hiccup can mean missed deadlines for the next big device launch.
The environmental footprint calls out for attention as well. Fluorinated gases, especially ones used for etching, make headlines for their impact on atmospheric stability. Their persistence means they can drift into the upper atmosphere and linger, adding to the greenhouse effect. There’s no easy fix here. Engineers keep looking for greener alternatives, lower-impact processes, or improved scrubbing systems to catch emissions before they escape.
Change has to happen at every step. Research labs chase less harmful etchants that won’t gut performance. Green chemistry principles come into play—designing molecules that deliver precision but pose less risk downstream. Companies invest in recovery and recycling systems, capturing fluorinated gases that once would have floated into the environment. And regulators lean on updated science to define exposure limits and emissions targets, all while partnering with industry to keep progress moving.
The tech world lives and dies by the smallest details, from chemical structure to supply chain resilience. Those lessons echo across science and manufacturing: chemical innovation opens doors, but it also brings hard questions about safety, stability, and stewardship. As we keep pushing for smarter solutions, the story of hexafluorobutadiene points to the need for balance between innovation and responsibility—a balance that matters for everyone who relies on the endless march of technology.
Hexafluorobutadiene, a fluorinated gas with high value for specific electronics manufacturing, doesn’t give much room for error. In my own experience working with specialty chemicals, even a small oversight in handling can lead to major risks for workers, neighborhoods, or the environment. That’s why the best operators turn to tried-and-tested routines for storage and shipping, built on a foundation of clear rules—no shortcuts.
Put simply, this isn’t the kind of stuff you stash in just any warehouse. This gas reacts if it slips outside safe boundaries of temperature, so top-performing facilities settle on temperature-controlled steel cylinders certified for pressure and corrosion resistance. Leaky containers or warmer-than-expected rooms aren’t just costly—they’re a danger. Gas cylinders live in ventilated, locked rack systems, clearly marked with hazard signs, far from busy workspaces or smoking zones.
Storing chemicals like hexafluorobutadiene is a team effort. Trained workers keep records of cylinder inspections, shelf-life, and never treat rusty valves or dented tanks as business as usual. If moisture sneaks in, things can go sideways fast. That’s a hard lesson I picked up early, when a supplier’s shortcut led to tiny leaks, making us scramble for replacements and forcing a review of our checks.
Every time a shipment leaves a plant, the risk shifts, demanding sharp focus. The most reliable chemical haulers load approved gas cylinders onto specially outfitted trucks, secured to prevent bumps or tipping. Trained drivers keep a printed manifest with emergency instructions. In tough weather, routes sometimes change to avoid exposure or delay, so there’s less risk of cylinders overheating during a jam.
Local and country regulations draw clear boundaries. I saw one transfer halted because a truck’s paperwork didn’t match the manifest—rules get enforced for a reason. These rules call for placards and labels that announce exactly what’s inside, so if there’s ever a spill or fire, first responders don’t lose a minute guessing.
Neglect or shortcuts don’t just rack up fines, they threaten health and safety. Hexafluorobutadiene exposure can irritate lungs, and a leaking tank can add long-lived greenhouse gases to the air. I’ve watched companies spend more on rapid clean-up after a misstep than they’d ever budgeted for safer storage in the first place. Prevention beats bad press, lost contracts, or—worst of all—tragic accidents.
Safer storage and transport depends on more than good intentions. Regular drills, smart investments in modern detection systems, and giving every worker a clear say all add up. Downsizing risk sometimes starts as a hassle, but in chemical handling, it quickly moves from headache to habit. Knowing the dangers and acting on years of industry wisdom keeps workplaces cleaner, workers safer, and communities out of harm’s way.
Smart organizations never leave safety as an afterthought. They listen to those with hands-on experience and make steady improvements to their systems—from sensors to staff training. Every time I see one of these shipments handled well, it reminds me that with the right approach, even the trickiest chemicals can move through the world without leaving damage behind.
Strong chemicals often make headlines but rarely stay in public conversation unless something dire happens. Hexafluorobutadiene, a specialty gas mostly used by the semiconductor industry, doesn’t draw the kind of attention that, say, lead paint or asbestos has over the years. That doesn’t mean it deserves a free pass—especially since the consequences show up in ways people might miss until they’re staring a problem in the face.
A quick Google search or talk with someone in chip manufacturing makes it clear: accidental exposure to hexafluorobutadiene doesn’t just create unpleasant workplace stories. Breathing in the fumes can trigger coughing, chest pain, or that tight feeling in the lungs. Direct contact stings the eyes, causing them to water and blur, and can burn the skin. I remember chatting with an engineer who once spilled a cylinder while making a maintenance round; even with proper gloves and goggles, he needed treatment for rash and persistent eye irritation. He’s not alone—these stories show that personal protective equipment only goes so far.
The far-reaching effects worry occupational doctors the most because data on long-term exposure to hexafluorobutadiene isn’t as complete as it could be. The available studies make one point clear: repeated exposure raises the risk of chronic lung disease. One journal article described cell changes in animal lungs, suggesting a link to more serious problems over time.
Some animal studies hint that chronic exposure could alter nervous system function or trigger cancer. There’s also the toxic effect on kidneys and livers, organs that often pay the price after years of subtle chemical insults. Scientific consensus remains cautious, but history shows how toxic risks play out decades after workers clock their hours—most people in the 1960s and 1970s missed the signs with other industrial toxins. We’re looking at a similar blind spot here: few long-term studies, and even less information for people living near plants using or disposing of this compound.
Strict regulations help, both for workers and for areas surrounding manufacturing facilities. Still, industry compliance only works as well as the enforcement and daily vigilance of everyone down the line. A lot of workplace stories share a theme—safety measures become more relaxed when productivity pressures hit or budgets tighten.
Transparent reporting stands out as a strong solution. Communities exposed to industrial chemicals deserve regular updates about what’s used nearby. The EPA’s Toxic Release Inventory gives a foundation, but public input and watchdog groups keep accountability front and center. For workers, unions and safety officers should push for annual health monitoring and stronger ventilation systems, not just written warning labels tucked into training binders.
Lower exposure limits also need a re-examination as more data rolls in. Engineering out risk—investing in safer alternatives or closed-loop systems—beats cleaning up messes later. A hospital can treat a burn, but it can’t erase a cancer diagnosis that might show up decades later.
Ignoring hexafluorobutadiene doesn’t make the dangers fade away. Every time workplace shortcuts happen or nearby neighborhoods get left in the dark, the cost gets paid somewhere else—sometimes much later, sometimes in ways that go far beyond the factory gates. We’ve seen industries change only after too many stories surface; learning from those lessons means not letting this chemical stay under the radar.