My appreciation for chemistry often leads me down rabbit holes of fascination, and fluoroboric acid tells a story worth sharing. It didn't arrive on the industrial scene by accident. Chemists working in the late nineteenth and early twentieth centuries recognized the value of complex acids as they sought alternatives to hydrofluoric acid for tasks requiring less aggression but equal persistence. The emergence of fluoroboric acid, or tetrafluoroboric acid, as a distinct entity happened as researchers looked for acids that could drive reactions without corroding everything in sight. The discovery grew out of curiosity and a practical need for acidity without the notorious dangers tied to other fluorinated acids. Chemical explorers quickly found themselves leaning toward this stable proton donor thanks to its manageable reactivity and solubility.
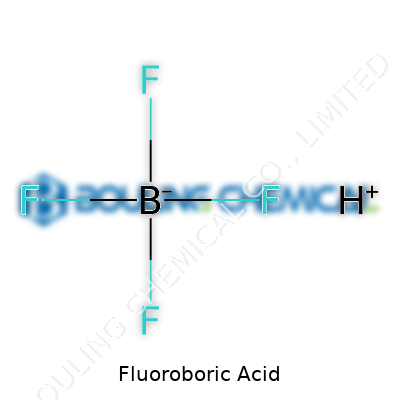
Diving into what sets fluoroboric acid apart, you have an inorganic acid with the formula HBF4. Most chemists encounter it as a clear, colorless solution—sometimes emitting a subtle sharp odor, which immediately reminds you to take care. The acid forms by dissolving boron trifluoride in water or by reacting boric acid with hydrofluoric acid. People often overlook its versatility, but for years, it has quietly influenced sectors ranging from electroplating to pharmaceuticals. What makes it stand out isn't just its acidity but how it releases protons in solutions where other acids would introduce problematic anions or metals.
Handling fluoroboric acid brings a distinct experience. The liquid burns through organic matter rapidly, though not as aggressively as some other mineral acids. Its boiling point and density depend on concentration, but the solution remains fairly mobile and transparent over a range of temperatures. Water solubility runs almost unchecked, which simplifies processes but demands respect for potential contamination. I recall early laboratory tasks where we measured pH with caution, aware that it interacts both violently with bases and gently enough to let complex ions persist. Many underestimate how this acid breaks down in water, yielding hydronium and tetrafluoroborate ions—a combination that pushes unique chemical equilibria. The acid doesn't oxidize or reduce many substances, granting stable performance in complicated reaction mixtures.
Technical documentation for fluoroboric acid does not just exist to check boxes. It sticks out as a testament to how careful you need to be when moving from bench to bench in a factory or lab. Concentrations vary, with purity judging whether it's destined for tech-grade etching or high-purity synthesis. People label cylinders and bottles with hazard pictograms signaling corrosiveness and toxicity for good reason. Flammable storage? Not an issue. Yet, its capacity to corrode metals, glass, and skin keeps it on the hazardous materials list. Every handling protocol I've come across calls for acid-resistant gloves, protective goggles, and sturdy containers—not because the acid is exotic, but because routine breeds inattentiveness, and one wrong move might cost dearly.
Lab synthesis tends to drift between simplicity and challenge. One classic route involves bubbling boron trifluoride gas into cold water, avoiding contact with the air to prevent hydrolysis. I remember fumbling with glassware under a fume hood, learning the critical importance of dry gas lines, as even small leaks let humidity turn BF3 into boric acid slush. Industrial setups take those lessons to heart, running closed reactors with monitored temperatures. Others take the wet chemistry route, using boric acid and hydrofluoric acid, combining the two in cautiously controlled stages. Either path, the acid doesn't wait for mistakes—over-addition or quick mixing sends fumes and can shatter fragile apparatus.
In reaction terms, fluoroboric acid has a way of making or breaking your experiment. It’s well known for acting as a non-coordinating acid; it gives up its proton but the tetrafluoroborate ion sticks to itself, rarely interfering with sensitive catalysts or organometallics. In electroplating, especially for tin and silver, this selective action translates into smooth, bright coatings, not spattered messes or dull surfaces. Organic chemists use it as a proton source where stronger acids like sulfuric would chew up reactants. I’ve seen it used to make stable fluoroborate salts by simple neutralization—important for pharmaceuticals and specialty materials. Its staying power in solution, plus the inertness of its counter-ion, solves problems no other acid can.
People don’t always call it fluoroboric acid. You'll find references to tetrafluoroboric acid, HBF4, even hydrofluoroboric acid in older patents. Commercial suppliers print HBF4 solution, often adding the phrase ‘in water’ to avoid confusion with powdered salts. In daily work, nobody mistakes its function, though variations in name sometimes slow down those just getting started. The alternative labels mainly serve to distinguish between concentration levels or to flag the presence of stabilizers, which crop up in large-volume shipments.
There’s no getting around the need for relentless safety discipline. Fluoroboric acid brings together corrosiveness and the hidden risk of toxic fluorides—especially as inhaled vapor. Guidelines from professional societies and occupational agencies stress the obvious: full skin coverage, eye protection, mechanical ventilation. What stuck with me after a minor laboratory splash years ago was just how quickly this acid can do damage, even at moderate concentrations. Having calcium gluconate gel nearby, knowing first aid protocols, and using plastic piping rather than glass or metal has become second nature. Rules about disposal aren’t just red tape—they reflect real risks of fluoride pollution, with local authorities watching waste streams.
Fluoroboric acid’s influence spreads far beyond academic labs. In printed circuit board (PCB) shops, it strips copper cleanly, prepping surfaces for delicate etching. Electroplaters depend on it for baths that lay down tin and silver with near perfection, jobs where old-school acids would pit the surface or leave odd residues. Dyes and pigments manufacturers lean on it too—its unique action stabilizes intermediates that would otherwise break down, letting them control shades and consistency. In pharmaceuticals, HBF4 salts matter because the counter-ion won’t react or destabilize sensitive drugs. Even the oil and gas sector has called on this acid for certain cleaning and scaling procedures, owing to its reliability where organic solvents stumble.
Every time I scan new chemical journals or patent filings, there’s some twist on using fluoroboric acid. Research groups have shifted from simple reactions to complex catalytic cycles, finding that HBF4 can tip equilibrium just enough to unleash hard-to-make molecules or sharper selectivities. Material scientists now use it for crystal growth, targeting battery electrolytes or next-generation magnets where impurities spell the difference between success or scrap. I’ve seen ongoing development of greener production routes, less reliant on gaseous fluorides, driven by both safety and cost incentives. Chemists tinker with its use as a supporting acid in superacidic systems, and bioengineers look for ways to harness it for protein modification and purification. Every advance points toward a broader, deeper range of uses.
Safety isn’t only about stopping spills. Toxicologists have taken a close look at fluoroboric acid for decades, balancing its utility with its danger. Inhalation or skin contact with concentrated solutions can burn tissue in minutes, and the fluoride can penetrate wounds, stripping calcium from bone. Chronic exposure issues emerge in high-volume workplaces, as even vapor can chew up lungs and contribute to chronic illness. Regulatory bodies monitor it as part of a cluster of hazardous acids. Over the years, advances in detection, personal protection, and neutralization treatments have cut down accident rates. Still, mishandling or poor training keeps risk on the table, which industry responds to with ongoing research into less hazardous alternatives and improved first-line treatments.
Chemistry keeps finding new roads, and fluoroboric acid stands to keep pace, not fade into the background. As electronics and specialty materials surge forward, the need for reliable acidic media without metal contamination only rises. Environmental concerns push producers and users to refine synthesis and disposal, squeezing every drop of efficiency and safety out of the process. The flourishing field of green chemistry might cut back its footprint by uncovering substitutes, but for now, HBF4 refuses to be written off. Its place in catalysis, material processing, and certain branches of metallurgy seems assured, at least until new discoveries manage to meet the same blend of power, selectivity, and practicality. For those of us working with it, vigilance and respect remain the best tools on hand, even as tomorrow’s chemists look for ways to outdo what today’s can only dream.
Working around chemicals, some names stick out more than others. Fluoroboric acid is one. This stuff isn’t just a curiosity for lab benches; it matters far more in industrial settings than most people realize. My experience in industrial cleaning showed me that this acid’s punch comes with benefits—and baggage. If you’ve ever seen a metal part stripped of its grime in a blink, you’ve probably witnessed fluoroboric acid doing its thing. Still, too many folks don’t realize where this acid turns up, or why safety needs a front seat every time it’s used.
Heavy machines and electrical parts don’t clean themselves. Fluoroboric acid gets called in where ordinary scrubbing fails. In metal cleaning, brightening, and electroplating, it works fast and leaves surfaces in better shape for bonding. From automotive shops to electronics plants, it pulls rust and oxide like no supermarket cleaner can. Removing tough scale from machinery parts keeps factories running. If the parts come out dirty, coatings peel away and performance drops. Over years, I watched how missed cleanup jobs cost companies tens of thousands in losses. There’s little room for shortcuts in maintenance, so the right chemicals matter.
So much of today’s tech would stall out without reliable electrodes. Making them isn’t about luck; it needs acids that strip away imperfections. Fluoroboric acid lets companies prepare exact surfaces for metal coatings in industries as wide-ranging as aircraft and circuit boards. Copper and tin electroplating depend on this acid for speed and results. Smooth flow in the tank means finished products pass stricter quality tests. It might sound distant, but everyone who depends on a laptop or car can thank these behind-the-scenes chemistry steps.
I’ve seen people underestimate the dangers. Undiluted, fluoroboric acid is unforgiving on skin or lungs. Protective gear isn’t optional; it’s the only safe way to handle this acid. There are stories—sometimes tragic ones—where a spill or careless splash led to months of recovery. Regulatory agencies agree: strict handling and storage are non-negotiable. The acid also presents long-term waste concerns. Disposal falls under hazardous waste laws for a good reason. Pouring it down a drain causes real harm, both to pipes and the local water supply. Most industrial firms create entire protocols for disposal, and sidestepping those rules does real damage.
Technology doesn’t stand still, not even in the world of acids. Newer options try to match the cleaning power of fluoroboric acid with less risk, but few replace its specific advantages in certain jobs. The pressure is on for chemical manufacturers and engineers to develop choices that clean and prepare just as well, but without the intense hazards. In my time, I’ve seen some progress. Enclosed systems and recycling units lower exposure risks and shrink waste footprints. Training programs have grown sharper, forcing everyone entering the field to respect the danger and respect the results. One thing’s clear: as demand for high-quality metalwork rises, attention to both safety and smart chemistry grows right along with it.
Most people never think about fluoroboric acid unless their job involves chemistry labs, industrial cleaning, or metal plating. This stuff sounds obscure, but it packs a punch. Having spent time in industrial settings, I remember the uneasy feeling working with acids that don’t just burn at the touch—they reach the bones and keep going if you treat them carelessly.
Fluoroboric acid stands out because it brings both hydrogen fluoride and boron trifluoride to the mix. Even diluted, it chews through skin, corrodes metal, and can fill the air with dangerous fumes. Breathing those fumes, even just once, can cause lung damage. Getting a drop on your glove eats through fast, and if it slips onto your skin, pain might not show up right away. That false sense of security often leads to bad injuries later on. Chronic exposure doesn’t announce itself loudly; it creeps up, making teeth brittle, bones ache, and the immune system sputter.
No one wins a prize for rushing through safety steps, and every old-timer in the field has a horror story about cutting corners. Working with fluoroboric acid calls for the best gloves—nitrile or neoprene work well—and a sturdy chemical apron. Splash-proof goggles protect the eyes. Respirators fitted with acid gas filters feel uncomfortable, especially in cramped spaces or during long shifts, but you get used to them once you realize how much trouble a careless breath could cause. Many labs and workshops use fume hoods to pull toxic vapors away, and you can tell the experienced hands from the rookies by the way they never work outside the hood.
Every bottle or drum of fluoroboric acid earns its own ventilated, acid-proof spot away from flammable chemicals and metals. Storing acids sounds like something nobody would mess up, yet stories keep cropping up where someone stacked bottles on a wood shelf or forgot to check for leaks. Once a container starts leaking, fumes work their way into the air in minutes, turning a simple spill into a building evacuation.
Spills and splashes seem inevitable in busy workplaces. Panic can do more harm than good. I still remember a small spill that turned serious because someone grabbed paper towels instead of reaching for the acid-neutralizer. Neutralizing fluoroboric acid usually takes special chemicals, not just baking soda or sand. If anyone gets exposed, the first move—always—means flushing the affected spot with lots of cold water, not wiping or dabbing. For inhalation, fresh air helps, but nobody should try to tough it out; heading straight to a medical professional matters.
Companies look for ways to cut costs on training, but real safety shows up in habits, not handbooks. Thinking back, I learned the most not from printed guides, but from coworkers who carried scars and didn’t like to talk about them. Regular, hands-on drills stick in people’s minds better than warning posters. Knowing emergency contacts, spill-response, and quick decontamination routines lets people react without losing time in a real emergency.
There’s no clever shortcut with dangerous acids. Trusting your senses, respecting what the substance can do, and staying strict with safety gear keeps you and your coworkers out of the ER. Fluoroboric acid does important work in the right hands, but ignoring its hazards never ends well. Staying vigilant grows from everyday habits, not a checkbox on a safety sheet.
Anyone who has spent time in a lab knows the feeling of respect mixed with caution that comes from working with strong acids. Fluoroboric acid brings an extra level of concern, not just because of its acidity, but also because of its potential to react with many common materials. Looking at its uses in metal cleaning, electroplating, and chemical synthesis, this acid finds its way into more workplaces than many realize. Yet, treating it as a regular acid can lead straight to disaster.
It’s easy to assume that most acids find a home in glass or plastic bottles. But fluoroboric acid laughs at shortcuts. It can break down common plastics like polyethylene and rubber, causing slow leaks and weakening containers. Even glass doesn’t offer a true sanctuary, especially over time. Laboratories that have seen the aftermath of a weak seam or a cracked jug know the mess it makes: acid eating through shelves, damaging floors, and putting people at risk. Acid-resistant materials such as PTFE or Teflon hold up better, but those upgrades come at a cost. Some labs try to save money with second-best containers and pay in safety instead.
Moisture in the air might not sound like a big deal, but fluoroboric acid soaks it up and gets even more aggressive. Any open container or poor seal means fumes escape, raising both health concerns and corrosion risks. That sharp, biting odor tells you instantly that something isn’t right. Beyond nose-level discomfort, inhaling those fumes damages the lungs and eyes. In my own experience, just catching a whiff near a poorly closed bottle confirmed how easy it is for things to go wrong, even if you think you’re being careful.
Shoving fluoroboric acid on a shelf near other chemicals encourages trouble. Mixing acids and bases isn’t just a textbook mistake—real-world accidents happen fast. Acids shouldn’t mingle with solvents, oxidizers, or flammable materials, as even trace fumes set off dangerous reactions. Acid-resistant storage cabinets made of coated steel or specialty plastic keep the acid isolated. Labels matter more than most people admit, since a faded or missing one sets the stage for confusion when someone new takes over the lab. I’ve seen storage rooms where poor labeling left coworkers guessing what was in old bottles. That’s not just inconvenient; it’s outright dangerous.
Institutional wisdom gets forged in sweaty, anxious moments when procedures fail. Training new staff helps, but policies alone do little if equipment falls short or managers ignore warnings. Investing in top-quality containers and acid storage cabinets beats any cost-saving effort. Regular inspection schedules—checking seals, looking for leaks, and reviewing labels—catch problems before they become emergencies. Emergency kits and eye wash stations near acid storage aren’t optional; they’re a direct response to hard-earned lessons about what can go wrong.
Most chemical safety regulations urge strict storage, sometimes with legal teeth. Insurers and regulatory bodies take a hard look at how dangerous acids get stored, and for good reason. A single spill doesn’t just threaten one person; it risks evacuating a building and calling in hazmat teams. Even the best training can’t undo a poor storage setup, so it falls to leadership and experienced workers to insist on safe, dedicated storage. In the end, respect for fluoroboric acid pays back more than it costs.
Anyone who’s spent time working around plating shops or chemical storage rooms has seen the clear, almost watery liquid labeled as fluoroboric acid. Chemists and engineers know it as HBF4, but outside science labs, few grasp how its concentration and purity shape its dangers and usefulness. Most people see a drum with a hazard sticker, not a complex blend with huge variation in quality.
What ends up shipped under the name fluoroboric acid varies a lot. In my years working with plating and electronics, I’ve come across drums marked anywhere from 45% up to roughly 50% HBF4 by weight. That number hides a lot – not just how strong the acid is, but also how much water, and sometimes even trace metals, ride along as impurities. For most commercial work, what people call “fluoroboric acid” hovers between 48% and 50%. Anything below makes the process sluggish, while anything higher brings serious safety risks and costs for storage.
If you’ve ever inhaled the vapors at full strength, the difference between a 45% solution and a 50% one isn’t subtle. The more concentrated forms punch harder in lab tests and industry tasks but require well-trained staff and proper gear. Many companies actually go for a slightly lower concentration to reduce splash risk and corrosion, even if it means slower etching or digesting.
Inconsistencies in product purity have real world effects. Contaminants like iron, silica, or heavy metals can mess with plating results, producing unpredictable layer thickness and even damaging circuit boards. High purity fluoroboric acid, often labeled “reagent grade,” costs more, but it lets labs and manufacturers trust their numbers in delicate experiments or high-end hardware.
Across my career, working with lower-grade material led directly to projects stalling—especially in electronics. Folks mixing up batches for sensitive uses can tell you stories of how one tiny hitch in purity suddenly made gold or nickel deposition act weird. Purity isn’t just about avoiding failed parts. Dealing with unknown impurities complicates waste treatment and environmental reporting, piling up headaches for teams already working to meet strict local rules.
It’s not all downside. The industry’s push for better transparency has helped. Tech companies now press suppliers for detailed certificates of analysis, confirming not just acidity, but impurity levels down to parts per million. This lets procurement teams judge what’s okay for mass production versus what’s fit for a pilot batch in a research center. Some regulators step in too, requiring checks on every delivery.
What stands out is the shared demand for clear standards. Whether prepping acid for automotive parts or etching silicon wafers, clearer labels and stricter batch testing reduce risks. Teams won’t always get pure, nearly-anhydrous acid – but having solid data makes it possible to gauge if the drum fits the day’s job. Companies investing in closed transfer systems, improved storage, and staff training keep people safer, and minimize stray reactions that cause injuries or expensive downtime.
For anyone working hands-on with chemicals, the little details—percentages, impurities, even the reputation of a supplier—are the line between success and costly error. Legislation and science keep moving the bar higher. There’s still a gap in public understanding, but inside the industry, few overlook that what matters isn’t just what’s in the drum, but how closely what's inside matches what’s promised on the label. Safer work and more reliable results depend on that honesty.
Anyone who’s handled harsh chemicals knows there’s no room for guesswork. Fluoroboric acid isn’t a name folks toss around in daily conversations, but in plenty of shops and plants, it’s as real as the concrete floor under your boots. The thing about it: this acid doesn’t play nicely with just any material. Its uses in plating, etching, or electronics show how important it is, yet its reactions tell you to slow down and ask the right questions.
I remember the first time someone tried pouring a potent acid into a steel drum. Results didn’t end well. Fluoroboric acid, with its strong acidic punch and knack for forming aggressive fluorides, chews through most metals except a few special types. Regular carbon steel and ordinary stainless steel? You only get pitting, corrosion, and a headache no one wants. So, you reach for plastic or glass. Containers made from high-density polyethylene (HDPE), polypropylene, or glass make the grade. There’s a good reason top-tier labs and plating shops pick these: the acid doesn’t eat them, and you don’t wake up to a dissolved workbench.
It feels like common sense, but nobody who relies on fluoroboric acid just mixes it on a whim. Tossing alkalis or ammonia into the mix and things turn ugly—dangerous fumes, severe heat, sometimes even violent reactions. Add bleach or strong oxidizers, and you risk toxic gases. While it plays an important part in making electroplating electrolytes and prepping metal surfaces, mixing without a plan leaves people exposed to real health hazards. That’s why anyone certified to work with this stuff gets more training than the average chemistry textbook can offer.
Fluoroboric acid doesn’t just threaten materials; it leaves a mark on living things too. Skin burns, eye irritation, chronic lung issues—each case reminds us that the right gloves, goggles, and ventilation systems matter as much as the acid itself. Relying on facts from peer-reviewed journals and regulatory bodies, responsible workplaces make sure all exposure guidelines get followed closely. If something spills, neutralizing with lime or calcium-containing products prevents bigger headaches.
People working with fluoroboric acid know the risks but don’t just throw up their hands. Industry groups call for better labeling, clearer safety data, and more thorough ventilation controls. New storage solutions, double-walled containment, and digital inventory tracking make it harder for accidents to go unnoticed. Even the best advice doesn’t always stick unless folks see the consequences up close, so learning from near-misses often drives safer habits.
Years around plating tanks and chemical storerooms teach you there are no shortcuts. If you’re planning to buy, store, or use fluoroboric acid, look past the surface. Ask about chemical compatibility, check safety gear, and know that real expertise isn’t just a set of rules but a commitment to everyone’s well-being. The best workplaces don’t just avoid disasters—they make sure no one leaves with regrets. That’s the honest truth behind compatibility.