Chemistry often appears out of reach, but take ethylene sulfate. It first emerged in scientific records during the mid-20th century, at a time when chemists searched for new routes to efficient organic synthesis. Interest picked up as researchers noted its ring structure could open doors to both industrial chemistry and laboratory-scale applications. Over the years, ethylene sulfate’s story fits right in with the growth of specialty chemicals. Advances in organosulfur chemistry during the 1960s and 1970s added momentum. Laboratories began turning out ethylene sulfate through several improved methods, shifting from academic curiosity to a building block in more meaningful processes. By the time large research consortia started examining lithium-ion battery electrolytes, ethylene sulfate’s profile kept growing as a niche additive. This development speaks to the evolving role of tailored synthesis in modern chemical industries—always on the lookout for molecules that bring something new to the table.
A closer look at ethylene sulfate brings out its character. It shows up as a colorless, mobile liquid at room temperature. Industrial chemists and research scientists find themselves drawn to it because of its cyclic sulfate structure: a three-membered ring including two carbon atoms and a sulfur atom double-bonded to two oxygen atoms. This structure means ethylene sulfate’s chemistry opens avenues for ring-opening polymerizations, as well as modifications in the synthesis of pharmaceuticals and fine chemicals. Some see it as a stubborn but rewarding raw material—hard to handle for bulk use, but valuable for precision synthesis. Its tightly wound ring carries significant strain, which means it reacts distinctly with nucleophiles and can serve as a versatile intermediate in making specialized organic materials.
On the bench, ethylene sulfate stands out for a few reasons. Its boiling point, ranging near 115 °C at reduced pressure, signals moderate volatility, while its density and water miscibility create distinct handling needs. Chemists know that its ring system holds potential energy, making it susceptible to attack by reactants like amines, alcohols, or even water under controlled conditions. Ethylene sulfate dissolves in polar solvents, letting researchers blend it into a wide range of reaction setups. In day-to-day work, care always matters, given the tendency of such strained rings to undergo exothermic ring opening, sometimes giving off hazardous fumes. Recognizing these features drives the protocols labs and factories develop whenever this chemical appears on the workbench.
Labels on chemical drums rarely tell the whole story, but for ethylene sulfate, specifics matter. Containers carry hazard warnings for severe eye and skin irritation and the need for ventilation during use. Technical literature stresses purity levels for different applications: electronic-grade materials often demand contaminants below parts per million, while alkylating agents in synthetic chemistry can handle slightly less stringent standards. Containers are almost always tight-sealed to keep out moisture, reflecting how small amounts of water can provoke slow degradation. Chemical supply catalogs rarely list vast quantities, since most demand falls inside the specialized sector, but every shipment comes with clear data on batch origin and storage demands: cool, dry, and away from sources of heat or sparks.
Lab-scale production of ethylene sulfate often tracks back to reactions between ethylene glycol and chlorosulfonic acid, with controlled addition of base to coax out the cyclic product. In industry, similar steps follow, with continuous-feed reactors allowing for better control over reaction rates and thermal management. Process development teams shoulder the job of minimizing by-products and maximizing yields: excess chlorosulfonic acid can push toward over-sulfonation or unwanted sulfation at non-cyclic positions. A well-tuned batch quickly becomes a lesson in heat control and careful addition—watching the color and exotherm gives experienced chemists early warnings if something drifts out of the expected range.
The reactivity of ethylene sulfate brings out the creativity in organic chemistry. Opening the sulfonate ring with nucleophiles leads to diols, hydroxy-sulfonates, or more complex derivatives when other reagents get involved. Pharmaceutical researchers see an opportunity here: one route moves through ring opening to create unique sulfonate-protected alcohols, which have become building blocks for more complex synthesis. In the polymers world, ethylene sulfate’s cyclic structure turns out to be a useful initiator or monomer precursor. Laboratories experimenting with ionic liquids also study its behavior in basic and acidic environments.
You rarely find just one name for a chemical. Ethylene sulfate travels under a few banners. Sometimes it's called 1,2-ethylenesulfate or ethylene oxathiolane-2,2-dioxide. Literature from older Soviet or German research efforts may use subtle variations, often aligning with local nomenclature habits. Most global suppliers settle on “ethylene sulfate,” but savvy researchers check chemical abstracts and synonyms, since databases sometimes mix it with close relatives—like ethylene sulfonate or derivatives with similar ring structures.
From my own work in chemical warehouses and academic labs, ethylene sulfate never gets treated casually. Glass bottles come with secondary containment in case of leaks. Safety data sheets mark risks related to eyes, skin, and—in the worst-case scenario—acute exposure to vapors. Fume hoods become non-negotiable. Not enough people acknowledge the fire hazards with strained-ring organosulfur compounds, so training drills at the start of every major project matter. Emergency eyewash stations and respiratory protection sometimes save the day. Modern labs build protocols around small-scale handling, spill containment, and waste deactivation, conscious of the compound’s role as both a building block and a hazard.
Ethylene sulfate’s influence extends most noticeably in specialty organic synthesis, but broader uses touch batteries, pharmaceuticals, and polymer chemistry. In lithium-ion battery research, adding small amounts to electrolyte blends shown higher cycle stability—an innovation that made headlines among electric vehicle engineers. In drug discovery, its ability to modify or protect alcohols lets medicinal chemists shape complex molecules with fewer side-reactions. Laboratory supply companies sell it to researchers probing ring-opening mechanisms and sulfonate group transfers. Its activity as a sulfonating agent gives process chemists another tool for pushing synthetic frontiers. Unlike workhorse chemicals, ethylene sulfate never becomes just a commodity; it stays tied to smart, targeted innovation.
The literature around ethylene sulfate sprawls across decades, with university consortia and private companies investigating everything from fundamental structure to practical use. Analytical chemists use NMR, mass spectrometry, and crystallography to track reaction intermediates. Battery technologists emphasize its oxidative stability and influence over solid electrolyte interface formation, citing real-world improvements to energy density and recharge cycles. In the past ten years, new patents describe ways to stabilize ethylene sulfate in reactive environments, limit decomposition, and extend its shelf life. University research often blends practical chemistry with theoretical modeling, especially as demand grows for battery chemistries that combine safety and performance. The most intriguing developments often come from underfunded corners of the research world—teams that take risks on out-of-fashion topics, then spark renewed industry attention with unexpected results.
A transparent discussion about any specialty chemical starts with what’s known about toxicity. Compared to some organic sulfates, ethylene sulfate hasn’t drawn the high-profile studies that, say, industrial solvents have. Animal data and in vitro work point toward local irritation and potential for systemic toxicity at higher doses, with standard mutagenicity panels returned in long-term studies. Regulatory guidelines emerge from this literature, focusing on limiting occupational exposure and preventing spills in water systems. Compared with more famous hazardous compounds, ethylene sulfate draws less widespread concern, possibly because its use often gets managed by experienced professionals in specialty settings. Even so, anyone handling or transporting this material follows protocols shaped by research on compound accumulation, bioavailability, and safe disposal. Newer safety studies, especially those tied to clean-tech battery scale-up, highlight the importance of not letting older, looser standards slip back into practice during high-pressure production surges.
Looking forward, ethylene sulfate’s story continues to unfold. Pushes for cleaner energy and advanced electrochemical storage have pulled it from the back shelves into the laboratories shaping the next generations of batteries. Synthetic chemists still find untried transformations as ring-opening routes develop. Challenges remain: improving scalability for industrial-size battery facilities, developing greener synthesis routes, and tightening exposure limits to prioritize worker and environmental health. A few promising teams investigate biocatalytic synthesis to reduce hazardous by-products, while others look at new derivatives inspired by ethylene sulfate’s core structure. The intersection of regulatory attention, battery innovation, and creative synthetic chemistry keeps this compound moving. For those invested in advances around electrochemistry and advanced synthesis, tracking ethylene sulfate’s place in tomorrow’s toolkit isn’t academic—it directly shapes the future of technology as much as the habits of the chemists who work with it.
Ethylene sulfate doesn’t show up in the headlines or get mentioned much outside chemistry labs, but this colorless, almost odorless liquid plays an outsized role in shaping some of the most vital technologies on the market. Spend any time digging into the chemistry behind batteries—especially the kind found in electric cars—and you’ll notice ethylene sulfate appearing more and more in research and industry discussions. So what exactly does it bring to the table?
I remember the days when a cellphone battery barely lasted until dinner. Lithium-ion batteries changed that. Ethylene sulfate is one of the unsung heroes in this revolution. As an additive in electrolytes—the liquid part inside batteries that lets ions move between electrodes—this little molecule helps fix some of the biggest headaches in making batteries safer and longer-lasting. One use that keeps popping up is in helping form a thin, protective layer on battery electrodes. This barrier keeps unwanted side reactions from cutting a battery’s life short, and helps batteries pump out steady power across years of recharging.
Ask anybody in electronics manufacturing, and the need for safer batteries comes up instantly. Fires linked to short circuits or overheating brought headlines and product recalls. Ethylene sulfate can play a part in reducing these risks. By improving the chemical stability inside batteries, this compound reduces the chance for dangerous reactions, making today’s battery packs more robust when faced with high temperatures or heavy usage. Research shows batteries using electrolytes blended with ethylene sulfate typically see less capacity loss over time. This boosts confidence in devices that need to run reliably, from medical gear to grid-scale storage systems.
Chemical production usually leaves a mark on the environment. As use of ethylene sulfate expands in energy storage, it raises fair questions. What happens once a battery dies? The truth is, chemical engineers still face hurdles in recycling complex battery mixtures. Ethylene sulfate’s stability helps batteries last longer, which means less waste. Still, developing better recycling systems for battery chemicals ranks high among the next set of challenges. Companies and scientists have started exploring closed-loop recycling, where recovered electrolyte components can find new life in fresh batteries, though real-world systems remain rare. We’ve seen similar struggles with plastics, and public pressure keeps rising for green solutions.
Most people never see the chemicals powering their everyday tech. Yet the details—right down to the role of something like ethylene sulfate—make a real difference in safety, performance, and the planet’s health. Learning about these choices opens up conversations about what we demand from the companies making these devices. It’s not just about longer battery life; it’s about pushing for smarter, cleaner approaches from start to finish. Support for research into advanced battery chemistry, smarter manufacturing, and robust recycling should matter to everyone who owns a phone, car, or backup power pack. The story of ethylene sulfate proves that chemistry, often forgotten in the background, quietly reshapes daily life.
I know what it feels like to have your hands in the middle of a busy lab or plant. Sometimes, curiosity draws you toward new substances. Ethylene sulfate carries its own set of risks, and forgetting that even once can bring trouble. This isn’t just a bottle to set next to the bleach; it’s a volatile compound, used in research and industry because of its reactivity. One slip, a splash, or breathing in fumes that don’t belong in your lungs, and you could spend the next few hours dealing with problems you didn’t see coming.
You won’t find comfort if ethylene sulfate lands on bare skin or splashes in your eyes. Inhaling any vapor is just as dangerous. Direct contact leads to chemical burns, irritation, or far worse damage. Eyes, especially, are at risk—no one wants to gamble with sight for a shortcut. I’ve seen colleagues skip goggles only to pay for it with days off work and doctor’s visits. Inhaling it might give you a headache at best. At worst, respiratory tissue gets inflamed and recovery drags out.
Long sleeves, tight cuffs, gloves made for chemicals, and quality goggles—these aren’t extras, they’re the baseline. The best professionals keep a habit of double-checking PPE before handling ethylene sulfate. Lab coats keep splashes off your arms and torso. Gloves protect the thinner skin on your hands. Goggles or a face shield mean your eyes and face have an extra layer between them and a mistake. Each item should be in good condition and replaced at the first sign of wear.
It doesn’t matter how skilled you feel—good ventilation has always been my favorite layer of protection. Fume hoods whisk away vapors before anyone has a chance to breathe them in. Never use ethylene sulfate in a closed room. Storing the bottle isn’t a job for a cluttered bench corner; it needs a tightly sealed container, away from heat, flames, and unsecured organic materials. More than one fire or small explosion started because someone thought “just for now” was safe enough.
Familiarity breeds carelessness. The only times I’ve seen real scares happen were when routines slipped or shortcuts sounded tempting. It’s tempting to believe nothing will go wrong, but ethylene sulfate’s reactivity punishes arrogance fast. Spills should get neutralized at once with proper absorbent materials—and not with your bare hands or a rag pulled from a drawer. Always have a spill kit ready. If a fire catches as a result of mishandling, standard extinguishers can make things worse. Use those specifically rated for chemicals, and know exactly where one is before you even begin.
Reading safety sheets only gets you halfway there. Practice and clear protocols teach you what hard copy cannot. Routine drills, up-to-date training sessions, and close observation of experienced hands shape safe habits fast. If a coworker looks unsure, there’s no shame in pausing everything until everyone feels confident again. I’ve seen new colleagues learn faster surrounded by folks who take questions seriously instead of brushing them off.
No experiment, no deadline, no shortcut justifies risking health for the sake of speed or convenience. If respect for ethylene sulfate slips, the danger quickly becomes personal. Real safety comes from knowledge, preparation, and purely stubborn consistency in protecting yourself and those around you.
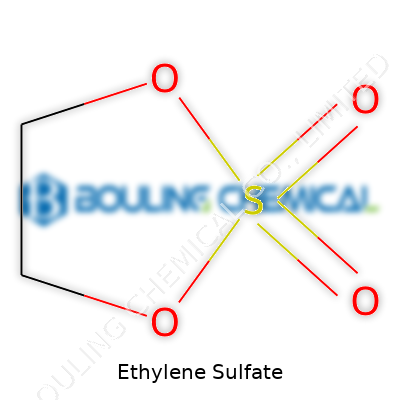
Ethylene sulfate deserves some attention for its role in chemistry labs and emerging applications in modern technology. This compound stands out because it brings together a simple structure and some serious chemical punch. Ethylene sulfate features a three-membered ring at its core—think of it as a triangle built from two carbons and a single oxygen. That ring belongs to the ethylene group, and the trick here is that this ring links with a sulfonyl group, forming a five-membered cyclic sulfate. Put together, the chemical formula comes out as C2H4O4S.
Drawing this out, you get a five-membered ring (a dioxathiolane), with sulfur right at the center, tied up with two oxygens and a vinyl group made of ethylene. Chemists write its structure as:
Ethylene Sulfate: C2H4O4SThe cyclic form means that the sulfur atom holds hands with two oxygens and a carbon chain, looping all the atoms into a tight, reactive ring.
A lot of folks shrug at chemical structures, figuring those curly lines don't change much. That’s not true with ethylene sulfate. Its cyclic form locks lots of energy and reactivity into the molecule. Squeeze together three or more atoms into a small ring and you’ve got strain—the chemical equivalent of winding up a spring. The sulfur atom inside this ring gives the molecule unique chemistry you won’t find in a simple open-chain alternative.
I remember my first real reaction with a strained ring compound in university—it crackled and fizzed faster than anything else we had in the lab. That stuck with me. Ethylene sulfate behaves in the same way: its ring strain makes it a useful building block, especially in organic synthesis and battery research.
Lately, I’ve been seeing ethylene sulfate pop up in surprising places. Researchers started looking at it as an electrolyte additive in lithium batteries. The idea isn’t exotic: its high reactivity lets it help form solid electrolyte interfaces (SEI) inside the battery, which slow down unwanted chemical side-reactions and help boost battery run time and safety. Peer-reviewed studies, including several from leading academic labs, back up this benefit, showing that ethylene sulfate can cut down gas formation and improve battery cyclability.
Describing it as a chemistry “helper” undersells it a bit—it actively changes how these battery cells live and die. When new discoveries promise cheaper, lighter, and more reliable storage for renewable energy, it’s never about one superstar element, but about the right tools at the right time. Ethylene sulfate shines because its ring structure is reactive enough to create strong, uniform SEI layers without causing a cascade of side reactions.
There’s a catch worth sharing. The same molecular energy that makes ethylene sulfate useful brings risk. Mishandling can lead to toxic byproducts, especially if the compound breaks down in the wrong way. Most commercial labs use strict controls—including ventilated hoods and careful handling protocols—to keep chemists safe.
As industry relies more heavily on lithium batteries and other advanced technologies, demand for raw materials like ethylene sulfate will only rise. Thoughtful regulation and shared best practices will go a long way to keep people protected while letting researchers tap the molecule’s unique benefits. The balance between practical use and safe handling never goes away—but with knowledge, vigilance, and shared experience, this molecule promises new chapters in chemistry’s story.
Storing industrial chemicals always brings a certain weight—some folks see the drums and tanks as just another line on a spreadsheet, or think protocols serve only to satisfy distant regulators. Ethylene sulfate snaps that illusion in an instant. This isn’t a substance for improvisation or afterthought. Anyone who has worked with battery manufacturing or handled specialty solvents probably knows the bite of mistakes here: spills, breathing issues, or even chemical fires after just a few minutes of negligence. Seen enough to know these are real risks, not just legal language on a package insert.
What separates ethylene sulfate from less volatile chemicals? Its flammability. If a container leaks or stays unsealed near heat-producing machines, it won’t just evaporate—vapors can ignite. Fire departments handle enough preventable incidents each year where a poorly labeled drum finds its way next to a space heater or a light fixture with faulty wiring. The right habit is to treat this compound with the respect saved for anything that burns readily. Store it away from direct sunlight, off heat sources, and in a place where only those who understand the chemistry have access.
Years spent in facilities handling solvents taught me that even a few percent of extra humidity in the storage room can change everything. Moisture doesn’t just spoil drums, it often causes slow chemical changes invisible to the naked eye. Before you know it, containers begin to bulge or leak, and crews face unexpected, expensive cleanups. Using tight-sealing, corrosion-proof drums isn’t just a checklist item; it’s common sense if you’ve ever untangled a sticky spill from the floor or had to explain lost inventory to management.
A good ventilation system isn’t just for emergency response. After spending time in a plant where air moved properly, headaches vanished and accidental exposures dropped to zero. The difference shows in health records and turnover rates. Vapors build up quietly in storage areas, lingering until someone swings open a door and suddenly breathes them in. Investing in fans and vents, even when it seems costly, benefits the whole team. Keep carbon filters and regular airflow maintenance on the budget, not just on paper.
Actual incidents often trace back to overlooked details—a label peeling off, a replacement worker who never saw the safety video. I’ve seen rookie mistakes domino through entire departments because one drum got “temporarily” parked in the wrong spot. Clear, tough labels and periodic training sessions aren’t bureaucracy run wild—they save real money, and sometimes lives. Mixing ethylene sulfate among other solvents or acids never ends well, so mark zones and lock them down. Even if a routine feels obvious, walking new hires through the correct procedure should stay a non-negotiable part of onboarding.
Guidelines for chemical storage don’t stay static, and keeping up matters. Agencies adjust standards as new research appears or incidents highlight overlooked problems. In my years, the best-run outfits dedicated a few hours each quarter to reviewing changes and walking through their stores with a fresh eye. Over time, this reduced close calls and delivered real peace of mind.
Storing ethylene sulfate right isn’t just compliance theater; it’s about responsibility—to the environment, co-workers, and the bottom line. Learn from those who’ve lived through the tough lessons. It costs a lot less to get it right upfront than to pay for cleaning up a mess.
For anyone who’s spent time working in a lab, the label on a bottle means a lot more than just a name and a formula. Some chemicals barely raise an eyebrow, but ethylene sulfate always makes you pause. This compound shows up in some battery research, polymer chemistry, and specialized syntheses—not exactly the chemicals most people have under their kitchen sink. But its hazards demand respect from even experienced chemists.
Ethylene sulfate is a cyclic sulfate ester. Chemists know that rings like these can push unexpected reactions, making them useful but finicky to manage. With ethylene sulfate, the main worry comes from its reactivity. It takes very little to make this stuff break open and react with water or alcohols, and those reactions often release strong acids or even toxic gases. That means spills turn into genuine emergencies if the proper gear and containment aren’t on hand.
I remember one situation from grad school where a fume hood fan failed during routine work with a similar compound. Even brief exposure led to burning eyes, scratchy throats, and a scramble for fresh air. Ethylene sulfate isn’t famous for causing explosions, but its vapors and breakdown products can be dangerous if ventilation isn’t perfect. That experience really made me appreciate why working in a well-maintained chemistry department is no luxury—it’s the only way to learn and work safely.
Direct contact with ethylene sulfate can irritate the skin, eyes, and mucous membranes. This isn’t just about minor discomfort. Its chemical nature means that it can directly damage cells at the point of contact. If inhaled, its vapors can injure the lungs—sometimes with long-term consequences. There’s also the suspicion that some cyclic sulfates, when metabolized in the body, form highly reactive intermediates. While animal studies could shed more light, what’s known pushes anyone who handles it toward the highest level of lab caution.
There is some evidence that accidental ingestion leads to damage in the digestive tract. At research scale, nobody risks finding that out firsthand. The body doesn’t easily clear sulfate esters, and that slow metabolism can mean cumulative harm over time. This is one of those chemicals that you don’t want lingering on a glove, a benchtop, or any item that moves between lab and office.
People sometimes ask why we even work with compounds like this. The answer is almost always tied to discovery—better batteries, smarter plastics, more efficient reactions. Those benefits don’t eliminate the hazards though. Fume hoods, PPE, strict training, and proper storage all make the difference. Just wearing gloves doesn’t always cut it with reactive chemicals; splash-proof face shields, chemical-resistant aprons, and sometimes even air-supplied respirators define safe work habits.
Clear labeling and up-to-date safety data are essential. No one should assume a new assistant knows the drill without a thorough briefing. Regular safety audits help spot small leaks or improper disposal practices before they become big problems. Digital inventory control can keep outdated or deteriorating stocks from becoming hazards themselves. Lab managers who never let safety slide build cultures of trust that keep everyone out of the ER.
As people working with chemicals, we share the responsibility for safe practice. Ethylene sulfate isn’t special because of how it looks or smells, but because small mishaps can lead to outsized consequences. The incentive to push science forward stays strong, but it only makes sense to move carefully—with eyes open and risk controls tight—whenever a bottle labeled ‘ethylene sulfate’ comes off the shelf.