Looking at the rich story of benzoxazole, it’s clear how curiosity and persistence shaped modern chemistry. Chemists in the late nineteenth century, with limited tools, cracked the code of this heterocyclic compound. They ended up uncovering not just a single structure, but a roadmap leading to diverse materials that keep showing up in daily life and advanced industries alike. Early researchers realized benzoxazole’s backbone connected to a family of functional and stable molecules, encouraging countless spin-offs through the decades. Since then, universities and private labs have picked apart its core structure and demonstrated surprising versatility, building bridges between theory and practical applications. Today, every discussion about benzoxazole pays a silent tribute to those methodical pioneers.
Benzoxazole stands at the intersection of practicality and ingenuity. Its ring system holds together a fused benzene and oxazole, balancing aromaticity and electron distribution to produce something quite stable and reliable under a range of circumstances. Scientists from core chemistry, materials science, and pharmaceutical research depend on this structure. When you look at those organic light-emitting diode screens, specialty fibers, or even everyday items like UV stabilizers in plastics, you’re probably encountering some descendant or cousin of benzoxazole. For every one of these products, there’s an industry betting on its proven ability to deliver consistent performance batch after batch.
The physical properties of benzoxazole sometimes seem unremarkable—crystalline appearance, relatively high melting point, and modest solubility in water—but under the surface, these traits provide a springboard for technical uses. The six-plus-five-membered ring imparts rigidity and influences both chemical reactivity and resistance to breakdown at elevated temperatures. These same properties attract interest among polymer chemists and coatings developers, who need compounds to withstand harsh environmental or operational conditions without wearing out too quickly. The molecule’s electron density also affects how it interacts with light, making it a recurring candidate for optical brighteners and dyes.
Detail-oriented chemists avoid missing the specifics—structural notation, percent purity, and functional group checks. The structure in textbooks or labels—C7H5NO—is more than basic chemistry; it’s a benchmark for everyone from the research chemist running NMR scans to the compliance officer tracking inventory. Technical paperwork details storage, hazard classification, and purity so that workers know exactly what they’re dealing with. Anyone in the supply chain—not just in labs but in transport and manufacturing—must recognize the difference between technical grade and high-purity variants, since a minute contaminant can throw off sensitive processes such as those in pharmaceutical synthesis or OLED fabrication.
Getting benzoxazole in the bottle, ready for use, demands a combination of craftsmanship and engineering. Researchers originally relied on condensation reactions between o-aminophenol and formic acid or similar reagents, typically under heat. That textbook synthesis still provides a reliable baseline. Industrial-scale routes tweak temperature, solvent, or even catalysts, trimming costs while boosting yield and reducing byproducts. Chemists working on sustainable chemistry keep refining those protocols, switching out dangerous reagents with greener alternatives wherever possible. This not only addresses environmental concerns but can also shave time and cost, creating a virtuous cycle between safety and efficiency.
Reactivity is where benzoxazole’s potential really opens up. The nitrogen and oxygen atoms in its ring throw open the door to targeted chemical modifications. Medicinal chemists often tinker with side chains to tune biological activity, seeking out candidates for antimicrobial or anticancer therapies. Material scientists customize substituents—adding electron donors, tweaking ring positions—to manipulate properties like light absorption or chemical resistance. The range of reactions possible with this core is vast: electrophilic substitution, reductions, couplings, and even polymerization. Changing just a single atom or functional group can mean the difference between a bland intermediate and a product that drives a billion-dollar market.
Research literature and global trade don’t always speak the same language. Benzoxazole shows up under IUPAC, common names, registry numbers, and branded names, each reflecting a particular context. In academic settings, the strictest chemical naming prevails—while customs forms or shipping labels might lean on shorthand or catalog terms. Anyone tracing sourcing, regulatory paperwork, or patent filings learns quickly how a single compound gets cross-referenced in hundreds of ways, linking databases and global supply routes. This demands attention from anyone invested in process safety or regulatory reporting.
Anyone who’s spent years around chemicals knows there’s no substitute for robust safety standards. Benzoxazole isn’t flagged as high-risk compared to some chemicals, but proper PPE, ventilation, and handling practices stop accidents in their tracks. Industrial and academic settings provide clear protocols: storage away from oxidizers, segregation from incompatible materials, controlled disposal, and environment monitoring. Agencies like OSHA and REACH outline how to communicate risks, minimize exposure, and train personnel so that health and environmental impacts stay minimal. Responsible producers and users treat emissions controls, wastewater management, and fire suppression as non-negotiables—not bureaucratic hurdles.
Benzoxazole quietly underpins products you wouldn’t expect. In textiles, derivatives reinforce performance fibers, shaping bullet-resistant gear and aerospace composites. Dyes and optical brighteners brighten fabrics and paper, supporting everything from fashion to banking security paper. Electronics turn to specialized benzoxazole compounds that enhance durability and conductivity in displays, sensors, or protective coatings. Pharmaceuticals lean on the core ring for active molecules in diagnostics and treatment. Market growth in renewable energy, 3D printing, and microelectronics rests partly on how this backbone can be modified to address new requirements. You see the benefit every time innovation calls for something tough, lightweight, or long-lasting.
R&D labs keep benzoxazole in sharp focus. Universities and corporate labs develop next-generation polymers, test new biomedical applications, and engineer molecules that address resistance or durability bottlenecks. Recent research explores pushing photovoltaic efficiency, creating smart polymers for sensors, and fusing benzoxazole with other functional groups for molecular imaging. These efforts bring together computational chemists, process engineers, and environmental scientists who test how tweaks in the structure translate to large-scale utility. As industries call for sustainability, the push is on for synthesizing these compounds using renewable feeds or novel catalytic methods. The next major leap—whether in medicine, electronics, or green tech—may rest on how far researchers can stretch this versatile skeleton.
Workplace safety officers and public health researchers devote plenty of attention to toxicity, even for chemicals usually classified as low hazard. Current science suggests most benzoxazole derivatives carry modest toxicity, but chronic exposure, byproducts from large-scale operations, and unregulated disposal can pose localized risks. Some studies highlight irritation or impact on local aquatic life at higher concentrations. Continuous toxicological testing, feeding into updated SDS documents, keeps both workplaces and communities protected. Industry watchdogs and green chemists have kept the pressure on for transparent data, prompting improved monitoring. Sound policy and responsible waste management help keep these compounds from building up in the environment or food chain.
Benzoxazole’s journey is nowhere near done. Demand grows for compounds that match rising performance standards and environmental regulations. Research into greener synthesis will decide which producers capture changing markets as customers demand less toxic byproducts and renewable feedstocks. Polymer engineers look to new derivatives that can deliver electronic performance without relying on rare earths or hazardous additives. Medicine continues to probe benzoxazole’s rings for leads in everything from antibiotics to diagnostics. Beyond that, advanced manufacturing—from additive processes to smart textiles—hungers for adaptable, safe, and high-performance chemicals. The drive for sustainability, efficiency, and innovation will keep this old molecule in the modern spotlight, bridging old traditions with new frontiers.
Benzoxazole doesn’t come up much in conversation unless you spend time in chemistry labs or work in an industrial plant. I first ran across it back in college during a project on advanced materials. Not the sexiest molecule at first glance, but dig deeper, and it quickly shows why chemists value it in so many applications.
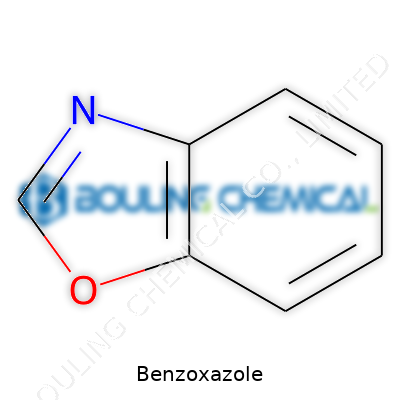
Picture a molecule: two rings, fused together—one nitrogen, one oxygen. Benzoxazole itself is simple but serves as the “blueprint” for making a range of valuable products. Its chemical stability stands out, making it a favored starting point for scientists and manufacturers craving reliability. Even if you’ve never laid hands on it, benzoxazole’s impact spreads throughout everyday life—everywhere from T-shirts to tires.
Ask anyone with experience in performance wear: strength matters. Benzoxazole derivatives form the backbone of polybenzoxazole (PBO) fibers—materials woven into fabrics for firefighter suits and bulletproof vests. These fibers offer heat resistance and mechanical strength no ordinary polymer can match. Having worn hiking gear made from advanced synthetic fibers, I’ve seen firsthand how important long-lasting, high-quality textiles are, especially when out in the field miles from any store.
Farmers have a different relationship with benzoxazole. Farmers don’t ask for the compound by name, but they rely on crop protection chemicals that contain benzoxazole derivatives. These molecules end up in fungicides, efficiently defending against blights and rots that threaten harvests. Agriculture depends on a steady food supply, and research supports the effectiveness of benzoxazole-based substances in halting the spread of crop diseases. The challenge lies in balancing protection and sustainability, so ongoing studies keep a close eye on residues and environmental persistence.
Step into research labs and you’ll find benzoxazole derivatives tested for new medicines. Compounds built on this scaffold show promise in treating cancer and bacterial infections. I used to volunteer at a health clinic, and access to new antibiotics or cancer therapies could mean hope for patients who had exhausted standard options. Discovery and testing move slowly, but benzoxazole-based drugs spark hope among chemists searching for new cures.
OLED displays rely on organic compounds to produce the vivid colors in screens on phones, TVs, and tablets. Scientists turn to benzoxazole derivatives for their stability and light-emission properties. Next time you swipe open your phone or catch a movie on a crystal-clear screen, think of the chemistry that quietly powers that experience—benzoxazole’s flexible structure makes bright, efficient displays possible.
Chemistry always brings tradeoffs. Benzoxazole and its family of compounds push industries forward, but also raise questions about worker safety, environmental impact, and responsible disposal. Regulators keep a close eye on exposure and waste, setting strict thresholds and cleanup requirements. Efforts to develop greener production methods deserve credit—less toxic byproducts, smarter waste management, and recycling strategies all count toward safer use of advanced materials in the long run.
My experience working with industrial chemicals taught me to respect both their potential and their risks. Benzoxazole does a lot of heavy lifting in textiles, agriculture, medicine, and electronics. By understanding how it works and staying alert to its effects, we can aim for progress that carries its weight without unnecessary cost to our health or environment.
Benzoxazole stands out as an important small molecule in both science and industry. At a glance, it’s a bicyclic compound, formed by the fusion of a benzene ring and an oxazole ring. That fusion matters because it sets up some unique chemistry. The arrangement of nitrogen and oxygen in the oxazole ring opens up reactivity you don’t get from plain benzene.
Anyone who’s worked in a lab knows the value of a reliable building block. Benzoxazole brings both stability and the ability to undergo selective reactions. It doesn’t break down easily under moderate heat or in air. This helps researchers trust that their modifications will stick, as long as things stay within reasonable reaction conditions. The aromatic system makes it less reactive than something with a lot of lone pairs or open carbonyl groups, but it still offers a good platform for adding other atoms if you provide the right push.
The way benzoxazole holds its atoms together allows it to resist mineral acids and weak bases. I’ve watched students try to force it into harsh reactions, only to realize it won’t budge without real effort. The position of nitrogen in the oxazole ring means you can do substitutions at the 2-position, whether you’re aiming for pharmaceuticals or dyes.
Solubility sometimes causes frustration for chemists. Benzoxazole dissolves well in organic solvents like acetone, chloroform, or ethanol, so it’s possible to try out reactions without the nightmare of insoluble muck. Water, on the other hand, doesn’t play well with it. This matters if you’re using it in formulations that stray outside the lab, since someone handling waste management or cleanup has to make sure it doesn’t hang around in groundwater or get away downstream.
Benzoxazole’s chemical stability contributes to its use in optical brighteners and high-performance plastics, giving consumer products a boost in everything from laundry detergents to cell phone screens. I remember chatting with a polymer chemist who praised benzoxazole’s rigidity; it keeps plastic chains tight, bumping up strength and endurance. In medicines, researchers prize its scaffold for designing molecules that bind to proteins with high precision. The electron-rich structure allows for custom modifications, which can help target specific enzymes or disease processes.
No commentary on a chemical’s importance can skip over its safety profile. While benzoxazole itself doesn’t blaze like gasoline or turn to poison in the air, any compound can become a problem if you dump enough of it where it shouldn’t go. There’s growing pressure on industries to understand environmental breakdown products. Some derivatives don’t break down quickly, so it’s smart to keep an eye on long-term buildup and ask hard questions about the wastewater coming from big plants that use it.
Think about the next generation of materials or medicines: benzoxazole stands as a tool to reach those goals. If you want to design new light-absorbing agents, or aim for drugs that slip through biological membranes, you’ll see this bicyclic structure pop up on reference sheets. Better waste management, green chemistry initiatives, and deep research into derivative toxicity can provide a safer future while keeping the benefits.
No molecule solves every problem alone, but benzoxazole’s chemistry opens real doors. Building a bridge between strong science and practical responsibility remains the best way to tap its promise while keeping risks in check.
Benzoxazole slips under the radar for a lot of people outside chemical manufacturing, but those who handle it at work or see it in product ingredient lists know it deserves a closer look. As someone who’s worked in industrial environments and keeps an eye on environmental safety debates, I’ve watched the conversation around chemicals like benzoxazole heat up. Claims about it being hazardous deserve unpacking with some candor.
Benzoxazole forms the backbone of many specialty chemicals, especially in dyes, optical brighteners, and polymers. Most people never encounter it deliberately, yet those in production, lab, or disposal sites can face regular exposure. That’s where the real questions start—does long-term or accidental contact with this stuff lead to trouble?
Research into benzoxazole itself lags far behind the attention given to more notorious compounds. Still, some studies point to impacts on aquatic organisms after runoff leaks into waterways. Other research shows repeated exposure may cause skin or eye irritation for workers. A handful of data suggests possible breakdown products can persist in soil and water, sometimes accumulating in creatures lower on the food chain.
Occupational guidelines focus on keeping dust and vapor out of the air, underlining the assumption that breathing or touching sizeable amounts isn’t risk-free. In my experience, companies with strict safety rules do much better at keeping incidents down to near-zero levels, but that relies on training and consistent oversight.
Right now, benzoxazole avoids the heavy scrutiny placed on substances like benzene or PCBs. The European Chemicals Agency and U.S. EPA haven’t lumped it in with high-priority hazards so far, but that doesn’t equate to giving it a clean bill of health. It just means decision-makers work with available information, and for benzoxazole, data stays thin. If fresh evidence shows it lingers in the environment or harms people over time, the rules may tighten with little advance warning.
Worker protection standards push for gloves, eyewear, and face protection around benzoxazole powders or liquids. For the public, the real concern involves what happens if this chemical leaches into water systems or local wildlife. Disposal routines offer some basic protections, but without frequent testing and strict management, sloppiness creeps in, especially at poorly policed sites.
Companies dealing with benzoxazole face important choices. Ramping up investments in closed-system technology, containment measures, and real-time monitoring can chop down exposure rates and environmental leaks. Product designers in textiles and plastics should vet safer alternatives or reformulate blends to cut potential risks. In my own projects, pushing for periodic reviews and third-party safety audits has unearthed unnecessary risks—fixing them takes time, but the peace of mind is worth it.
Education plays a huge role. When workers know what they’re dealing with and how to react to spills, fewer bad surprises occur. Sharing lessons learned across industries—rather than hoarding safer practices—gives everyone a better shot at staying healthy and keeping waterways uncontaminated.
People deserve to know what chemicals turn up in workplaces, communities, and consumer goods. While benzoxazole doesn’t make headlines yet, ignoring its possible hazards means risking long-term problems for short-term convenience. Scrutiny works best when scientists, companies, and the public share facts and review honest mistakes. For now, keeping a watchful eye on benzoxazole’s trail means healthier lives for both workers and neighbors.
Benzoxazole turns up in a range of chemical processes and products. Folks in labs, factories, and even smaller-scale settings work with it, sometimes without thinking through all the small details that make a big difference. I’ve seen firsthand how bad habits around chemical storage can creep in over time, even for people who know better on paper. Common sense and experience both say sloppy routines put not just health at risk but also research timelines, manufacturing consistency, and even company reputation.
Benzoxazole brings certain risks in its solid and vapor forms. My own background in chemical safety taught me the importance of recognizing those hazards realistically. Reports from regulatory and scientific literature agree—it can irritate eyes, skin, and lungs, and there’s always the possibility of reactions with incompatible materials. That’s why keeping it dry, cool, and well-separated from harsh acids or oxidizers isn’t overkill. It’s just following the logic supported by established science.
I remember working at a place where folks stacked bottles high on top of one another in a hot storeroom with no airflow. Over time, poor storage led to minor leaks, broken containers, and confusion about read labels. Practically, putting benzoxazole in a tightly sealed container—out of sunlight, away from direct heat, and on a well-marked shelf in a secure area—spares you a lot of headaches. Labeling seems basic until someone mistakes the powder for another similar-looking compound.
People sometimes think of temperature control as optional, but with benzoxazole, temperature swings can impact product quality and safety. Based on the literature I’ve read and my own experience, a stable temperature a bit cooler than room temperature keeps the chemical from breaking down or causing pressure buildup in containers. Throw in a spot with low humidity, and you keep clumping, contamination, and accidental reactions to a minimum.
Wearing the right gloves and protective eyewear is the simple move, with many folks in the industry agreeing a well-ventilated workspace reduces the risks of breathing in dust or vapors. Rail thin budgets sometimes tempt companies to skip these steps, but I’ve never seen cutting corners on personal protection save money in the long run. Careful handling means supervising who has access, using spill trays, and setting up stations with clear instructions on what to do if an accident happens.
Keeping spill cleanup materials nearby feels like over-caution when everything goes smoothly. The moment a spill occurs, that simple preparation pays off. Make sure everyone who handles benzoxazole knows where to find these materials and how to use them safely. Relying on clear, practiced procedures beats any expensive, last-minute scramble. Regular reminders, training sessions, and feedback loops build a culture where people don’t get careless as months go by.
Plenty of industries have a rulebook for chemical handling, but the real difference comes in how these rules play out day after day. From years around labs and tech spaces, I’ve watched good managers reward attention to detail and openness about mistakes. One slip-up shouldn’t lead to punishment, but rather an honest review to figure out the weak spot in the system.
In the end, storing and handling benzoxazole responsibly sends the message that health, safety, and quality still matter. It keeps reputations clean, workplaces safer, and anyone downstream of that chemical—from workers to end-users—a little more protected.
It’s easy to ignore the chemistry behind everyday items, but sometimes a material quietly changes how things work across entire industries. Benzoxazole, for instance, pops up in places you might not expect. I’ve been in manufacturing plants and labs — the kind where the smell of solvents clings to your clothes — and in those settings, benzoxazole stands out for its versatility.
Start with textiles. High-performance fibers owe a lot to benzoxazole derivatives. You can find fibers like PBO — short for poly(p-phenylene-2,6-benzoxazole) — used in bullet-resistant vests, firefighter suits, and high-strength ropes. PBO offers a high tensile strength and heat resistance, properties that go far beyond basic nylon or polyester. Consider the trust a firefighter places in their protective gear or a worker relying on a climbing harness. There’s pride in knowing a bit of chemical ingenuity keeps people safe where it counts.
The electronics sector has also found plenty to love in benzoxazole. Printed circuit boards and microelectronics rely on insulation that can survive high temperatures without breaking down. Benzoxazole-based materials keep gadgets running cooler and longer, even in cramped spaces under constant load. My experience troubleshooting failing boards taught me how upgrading to thermally stable polymers prevents costly, embarrassing recalls and downtime for manufacturers.
Pharmaceutical research benefits from benzoxazole as well, mostly because its chemical structure lends itself to creating molecules with potent biological effects. Medicinal chemists have discovered entire classes of benzoxazole-based compounds with anti-bacterial, anti-fungal, and anti-cancer possibilities. There’s a sense of excitement in the labs when a smart tweak to a molecule saves years of research or brings a drug to trial faster. The stakes here are as high as they come — measured in patient lives, not just profits or convenience.
On the pigment and dye side, benzoxazole structures help deliver bright, durable colors for optical brighteners in detergents, plastics, and papers. It’s easy to overlook how much people want a shirt to stay white or packaging to stay eye-catching. Without strong, stable brighteners, every wash would make clothes grayer and duller, and printing companies would waste mountains of ink trying to keep packaging lively.
Environmental and safety concerns keep getting more attention, with regulators asking tough questions about persistence and toxicity. The chemical world has too many stories about substances once hailed as breakthroughs later linked to pollution or health hazards. Producers looking to stay on the right side of regulators and end users can invest in green chemistry approaches — for instance, pursuing biodegradable options and safer synthesis methods. This usually means costlier research upfront, but brands stand to gain long-term trust and avoid the headaches that come with product bans or recalls.
For young scientists or entrepreneurs looking for a way in, benzoxazole offers a lesson. Even something as niche-sounding as a heterocyclic compound can unlock big value when used thoughtfully. It pays to look at what the market needs today and what the world will demand tomorrow — durability, safety, high performance, and less harm to the planet. There might not be headlines every time a chemical quietly improves our lives, but behind the scenes, innovations like these are worth every bit of attention they get.