Chemists never stop reaching for new challenges, and the road that led to 4-Methoxymethyl-2,3,5,6-tetrafluorobenzyl alcohol demonstrates that thirst for the novel. In the late twentieth century, with organic synthesis entering a revolution, fluorinated compounds emerged as a priority in both academic and industrial research. It didn’t take long for scientists to latch onto the potential of multi-fluorinated phenyl derivatives. These molecules held promise in fields from pharmaceuticals to material science. As demands grew for precision, refinements, and novel functionalities in molecular design, various synthetic routes kept evolving—the goal was better selectivity, higher yield, and fewer hazardous byproducts. Early methods often relied on harsh conditions, making these fluorinated alcohols tricky to access in pure form. Over the decades, teamwork across the globe drove improvements in fluorination strategies, catalysts, and handling of toxic reagents, ultimately opening access to a suite of products including this tetrafluorinated alcohol.
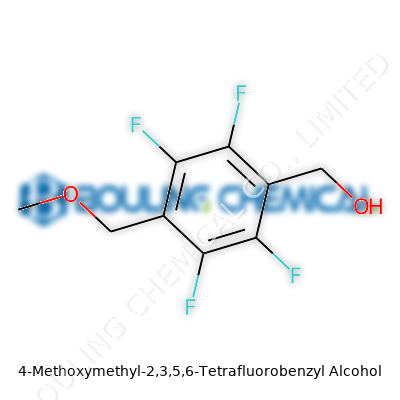
Now, the world sees 4-Methoxymethyl-2,3,5,6-tetrafluorobenzyl alcohol as more than just a niche chemical curiosity. This compound stands out due to its unique makeup: a benzyl alcohol core, four precisely-positioned fluorine atoms, and a methoxymethyl group. This combination sets it apart for use in drug design, specialty polymers, and advanced coatings. Fluorinated building blocks like this one often command a premium in chemical synthesis, not only for their chemical resilience but for their ability to deliver solubility tweaks, metabolic stability, or improved biological activity. Now running through experimental drug pipelines, crop protection research, and new material development, this alcohol finds itself at a juncture where innovation meets market need.
Anyone who has handled this compound in a lab will note its stability and unique traits. The high level of fluorination at precise positions on the benzene ring grants it strong electron-withdrawing effects, leading to lower reactivity compared to non-fluorinated analogues. Once you add the methoxymethyl group and the benzyl alcohol, you get a molecule that both resists some types of degradation and participates in a variety of coupling and derivatization reactions. Its boiling point sits higher than that of some simpler alcohols, and its solubility leans toward organic solvents. This is no accident—chemists purposefully design such molecules for processes that demand both organofluorine performance and ease of manipulation.
This isn’t something most people see on a store shelf, so standards matter. In any research or lab environment, precise labeling on bottles and in documentation preserves safety and accuracy. The compound must be delivered in amber glass to reduce light-induced degradation, and purity figures routinely approach or exceed 98%, given the difficulty in purging fluorinated contaminants. Typically, you find its molecular formula, batch number, and an up-to-date safety data sheet. Because it’s not regulated for direct consumer use, labeling follows conventions of the scientific community rather than retail code. In my experience, such standards reflect the pro-level rigor needed by anyone working on the frontier of new molecules.
Making this specific alcohol isn’t a simple one-step process. Chemists usually start from a suitable halogenated aromatic precursor, employ selective nucleophilic aromatic substitution or transition-metal catalyzed functionalization, then introduce the methoxymethyl group using well-tuned bases and solvents. The alcohol functionality might need protection and deprotection steps, depending on route and desired yield. Each stage demands care, as side products from incomplete fluorination or overreaction can spoil purity. It’s a good example of how deep knowledge of physical organic chemistry, coupled with practice at the bench, can elevate a synthesis from theoretical to practical. Most of the improvements in yield and selectivity come from persistent benchwork and shared learning across generations of chemists.
With four electron-withdrawing fluorines at ortho and para positions, this benzyl alcohol behaves differently compared with its non-fluorinated relatives. The alcohol group still reacts in standard esterification and etherification scenarios—but the electron-poor aromatic ring imparts extra stability to many otherwise-labile derivatives. Chemists appreciate this compound as a scaffold for building more complex molecules. For example, under the right catalytic conditions, it can serve as an anchor in Suzuki or Heck cross-couplings, with the additional benefit that the fluorines modulate local charge to affect reactivity. This kind of control helps medicinal chemists fine-tune binding to target enzymes, optimize pharmacokinetics, and create tailored properties in polymers or coatings, all from a relatively simple base.
Names can get confusing in fluorinated chemistry, so clarity is key. Chemists working in multinational teams sometimes rely on systematic designations or shorthand formulas. You may see this molecule written out as 4-MOM-2,3,5,6-tetrafluorobenzyl alcohol, or as a specific catalog number if referencing commercial suppliers. This helps avoid the pitfalls of similar-sounding compounds with vastly different properties. In practice, most teams stick to a preferred synonym for shorthand in research records, but always cross-check against the full IUPAC designation to prevent mishap.
Handling this alcohol, like most fluorinated organics, takes attention to detail and respect for chemical safety. Although not acutely toxic in trace amounts, organofluorines may pose long-term health risks if mishandled: skin contact, inhalation, or accidental ingestion can bring on irritation or systemic effects. Proper use of gloves, eye protection, fume hood ventilation, and secure storage all reduce unnecessary exposure. Disposal routines follow hazardous organic waste guidelines to avoid releasing fluorinated byproducts into water or soil. Over many years in labs, the teams I’ve worked with have kept accident rates low through a culture of accountability, routine safety drills, and active enforcement of safety protocols. Education and oversight do more to protect people and the environment than clever technology ever can.
The presence of four closely-arranged fluorines and a reactive alcohol opens the way to a broad array of applications. Medicinal chemistry often grabs the biggest headlines, where adding a tetrafluorinated motif can dramatically shift a molecule’s behavior in vivo. These tweaks, drawn from carefully-planned synthetic strategies, have changed the game for metabolic resistance and drug half-life. Outside pharma, crop science relies on tailored fluorinated scaffolds for robust, long-lasting plant-protection agents that stand up to harsh field conditions. Polymers designed for extreme chemical resistance often feature such fluorinated groups; the resultant materials withstand aggressive acids, bases, or solvents far better than earlier plastics. In coatings, this alcohol’s derivatives repel moisture, oil, and grime, which makes them ideal for surfaces that need to stay clean or reduce friction. As new regulatory scrutiny on older fluorinated products grows, chemists search for greener, safer alternatives—this compound’s unique structure offers a starting point for next-generation molecules less prone to environmental persistence.
In the research world, the story doesn’t stop with a single synthesis. Teams worldwide search for broader strategies to work with complex fluorinated scaffolds. Some recent breakthroughs stem from better catalysts that let chemists introduce or remove functional groups with less waste and at milder conditions. Fluorine chemistry rewards persistence, as unpredictable reactivity sometimes blocks progress or produces unexpected byproducts. Multinational consortia now pool information, sharing unsuccessful runs as well as successful ones to accelerate progress. In drug discovery, this alcohol forms a backbone for novel kinase inhibitors and enzyme-bound probes. Scientists in polymer science investigate how to blend these rigid, hydrophobic aromatics into flexible matrices for advanced insulation and packaging. Once-startling inventions like these now supply both short-term applied research and the deeper, slower projects that lay the groundwork for the next big leap in performance or sustainability.
Any responsible commentator must address environmental and health impacts. Some fluorinated organics make headlines for their persistence and bioaccumulation. For this tetrafluorinated benzyl alcohol, direct acute toxicity appears low based on available testing. Chronic exposure or metabolic degradation, though, raises longer-term questions. Research teams carry out ongoing studies into how it breaks down—does it linger as a fluorinated fragment, does it bioaccumulate, or does it degrade safely? The lessons of perfluorinated pollutants have left a legacy; companies and agencies now apply more caution, running tests for both animal and environmental safety before widespread adoption in products. Every new data point better informs future design and regulatory control, underscoring the need for continued vigilance, transparency, and responsibility.
The future of 4-methoxymethyl-2,3,5,6-tetrafluorobenzyl alcohol looks dynamic, shaped by shifting regulations, growing demand for sustainable chemistry, and the relentless pace of materials innovation. Researchers aim to design molecules that deliver high performance with lower environmental footprint. In pharmaceutical synthesis, the ability to tune reactivity and metabolic stability through targeted fluorination keeps interest alive in this and similar scaffolds. Material scientists, chasing lighter, tougher, and more durable plastics, seek new ways to use such motifs without creating persistent pollutants. Ongoing international dialogue over regulation, safety, and green chemistry keeps the field progressing, challenging chemists to push innovation that doesn’t merely solve today’s problems but anticipates tomorrow’s. My own experience in both industry and academia suggests that the chemists who pay equal attention to the real-world impacts and the elegance of their syntheses will drive the future of advanced fluorinated materials.
Anyone who has spent time reading about fine chemicals probably stumbled on names like 4-Methoxymethyl-2,3,5,6-tetrafluorobenzyl alcohol. Sure, the name’s a heavy lift, but the substance has earned its reputation in advanced labs. My time helping with a medicinal chemistry project showed me that even compounds with tongue-twisting names play quiet but crucial roles in current research and technology. Let’s talk about where this one fits in.
Drug discovery has always relied on the ability to swap functional groups around a core molecule, trying to boost activity or reduce side effects. Chemists know how precious a reliable benzyl alcohol derivative can be when developing new scaffolds or protecting sensitive portions of a molecule. This particular alcohol, with its four fluorine atoms and one methoxymethyl group, offers some real advantages. The fluorines change the way the molecule interacts with enzymes, often by increasing metabolic stability or changing how the compound fits in an active site. Fluorine changes a molecule’s whole personality—it can make it more resistant to breakdown or tweak how it gets absorbed by the body.
With this alcohol, medicinal chemists attach it to drug candidates as a short-term “mask” or protecting group, then remove it when that stage of synthesis wraps up. It helps keep reactive parts of their compounds safe until they’re needed later. From my own experience, some promising antiviral and oncology projects have used similar fluorinated benzyl groups to modify lead compounds and improve their pharmacokinetic profiles.
This alcohol doubles as a versatile intermediate. Academic labs see value in using it to build more complex structures for testing or for crafting unique ligands for catalytic reactions. The methoxymethyl part isn’t just window dressing; it acts as a handle for further modifications, giving chemists a way to make quick changes without having to redesign the entire route. Those fluorines, again, help steer the reactivity and make certain transformations possible that just don’t work with plain benzyl alcohol.
Anyone doing analytical work with fluorine-containing drugs knows the benefit of stable, unique fluorinated markers. Mass spectrometry and NMR both pick up fluorine signals easily, so this compound and its cousins become reference points or internal standards. At the early-stage pharmaceutical company I worked with, using benzyl alcohols like this one allowed for easy identification of products and helped in tracking reactions in complex mixtures—crucial for staying on schedule and keeping data honest during a patent race.
Despite all this, chemists still face hurdles. Fluorinated organics don’t play by the same rules, and production costs can rise sharply because multi-step synthesis and special handling become the norm. Environmental stewardship pushes people to look for ways to recycle fluorinated byproducts or find greener routes. In our group, we started re-examining solvent choices and making sure every gram of material counted, knowing costs weren’t just about dollars but also about minimizing hazardous waste.
Modern chemical research marches on the back of unsung workhorses like 4-Methoxymethyl-2,3,5,6-tetrafluorobenzyl alcohol. Teams pushing to solve disease or invent the next material pay close attention to properties like selective protection, ease of modification, and analytical clarity. For those of us behind the bench, keeping an open mind about new uses or more sustainable methods keeps science fresh — and keeps this compound central in advanced synthesis and innovation.
Talking with friends in the health and science world, I've learned something simple: purity isn't just a number on a label. Folks check for it in vitamins, cleaning products, even the foods they eat, hoping for safety and value. I get it. Who wants fillers or surprises when they think they're paying for the real thing?
Lab tests tell a bigger story than branding. Certified testing, using methods like chromatography and mass spectrometry, digs deep to show what’s really in that bottle or bag. Independent testing—with labs accredited for accuracy—can catch impurities, heavy metals, or sketchy residues that sneak past lax oversight or cut corners.
Consider supplements: in 2015, a New York investigation found that many top-selling herbal products didn’t even contain the plants listed on their labels. Instead: rice, houseplants, unknown substitutes. This matters a great deal. In medicine, contamination or mislabeling can harm. With food, allergies mean a trace can be dangerous. History gives us enough reasons to demand clear, honest purity information.
Asking about product purity goes beyond curiosity. If a chemical solution claims to be 99.5% pure, that sounds reassuring, but what makes up the leftover half-percent? That small slice could make all the difference for someone with sensitivities, or for a lab running precise experiments. In industrial settings, trace contaminants lead to flawed results or faulty equipment.
Numbers need context, too. Cosmetic and supplement brands like flashing big digits—99% or “pharmaceutical grade”—but regulations often lag behind marketing. The Food and Drug Administration, for example, doesn’t require all supplements to undergo batch-by-batch purity testing before hitting shelves. The result: trust shifts to third-party verification from organizations like USP or NSF, which keep watch where rules fall short.
Anyone weighing a purchase can start by reading third-party certifications listed on labels or company websites. That phrase—“independently verified”—is worth searching for, since it signals someone else checked the details. Researching lab reports, especially with products used daily or ingested, also gives peace of mind. More voices raising questions helps push companies to step up transparency.
For those of us choosing between brands, loyalty shifts fast. A company that lays out full ingredient lists, recent test results, and sources of raw material wins my business. Social media and review platforms let customers share experiences—flagging products that fall short or stand out. Grassroots pushback gets attention, leading to recalls, apologies, and sometimes better standards.
The technology for testing keeps getting cheaper and quicker, making it easier for smaller companies to ensure quality. Groups like AOAC International develop validation tools so accuracy doesn't depend on big budgets. Encouraging open data—where companies display recent batch results online—would help everyone make smarter decisions.
Asking about purity isn’t nitpicking; it’s protecting your health, investment, and peace of mind. By understanding what makes up that percentage number and demanding real answers, people can help foster a marketplace where honesty and science win. We all deserve to know what we’re putting in our bodies and homes, with no fine print hidden behind glossy packaging.
Bringing home a bottle of 4-Methoxymethyl-2,3,5,6-tetrafluorobenzyl alcohol doesn’t feel like picking up basic groceries. This isn’t a household cleaner or an over-the-counter medicine. Many folks who work in labs know the story—chemicals don’t forgive carelessness. A seasoned chemist can tell you there’s nothing routine about storing chemicals, especially those with a hefty name like this one. Something as simple as the storage setup can separate a normal day from a disaster.
Most organic chemicals last longer when kept in a dark, cool spot. Light introduces energy, and with enough of it, even stable molecules can break down. In my experience, a dark cabinet or amber bottle stretches the life of any sensitive liquid or powder. The cold slows reactions—think of it like keeping milk fresh in the fridge compared to leaving it out on the kitchen counter. Refrigerators dedicated to chemicals, not food, keep substances usable and safe for longer periods. I’ve seen experiments fail from degraded reagents, and few things frustrate research—both in university labs and in pharma—like lost time and wasted money from poorly stored chemicals.
Bottles of specialty alcohols like this frequently arrive with a manufacturer’s note about avoiding sources of ignition and keeping containers tightly closed. Those warnings might start to sound like white noise after the third or fourth bottle, but every lab fire or chemical exposure changes that tune fast. If someone in the workplace opens a chemical storage unit and breathes in unfamiliar fumes, it's clear something broke down or leaked. Relying on nose or guesswork risks real harm. That’s why chemical safety data sheets sit in binders or digital databases: they spell out what works and what doesn’t.
Fluorinated compounds may not fear water as much as sodium metal, but moisture creates unpredictable changes. Over time, humidity sneaks through unsealed lids or into less-than-airtight containers. Chemicals can break down, or react in ways nobody expected. I remember rainy seasons where small leaks in storage rooms ruined entire batches. Using desiccators and making sure caps fit tight becomes a habit once you’ve seen a year’s budget wash away on ruined stock.
Regulatory guidance spells out minimum standards, but real safety kicks in when workers and researchers talk openly about what could go wrong. I’ve worked with teams where experienced hands walk newcomers through the routine: keep incompatible chemicals apart, never store near acids or strong bases, and always label each bottle with the date opened. Mistakes shrink, spills get caught early, and injuries become rare. Routine checks on storage conditions make this approach stick. It never feels wasted—even on a busy day. Those small steps add up to a safer workplace.
So many discoveries in chemistry, life sciences, and engineering trace back to careful habits. Safe, stable chemical storage supports good results, strong teams, and well-run labs. Choosing the right storage protects more than bottles and budgets—it keeps people healthy and research reliable. It’s a simple truth: take care of your chemicals, and they’ll take care of your work.
Few topics create more confusion than the risks found in labs, warehouses, or even beneath kitchen sinks. Ask ten people if a substance is dangerous and you get ten answers, usually backed by confidence more than data. Plenty of factors come into play: some chemicals scream danger with a whiff, others do harm long before you notice a thing. Decisions hinge not only on labels, but also history—both personal and scientific—and the hard-earned lessons of trial and error.
A label can tell a story, but it rarely tells the whole story. Sodium hydroxide, used for cleaning drains, burns skin in seconds. Gasoline, present on every street corner, evaporates in the sun while filling the air with risk. Plenty of hazardous substances are everywhere, handled safely most of the time simply because workers wear gloves, ventilate rooms, and move with care. In my own experience, what separates a routine shift from disaster isn’t the chemical, but the habit of reading—and actually believing—what safety data sheets and old-timers say can happen.
People get used to things that are dangerous. Dull routines dull minds; I’ve seen coworkers handle ammonia as casually as dish soap, just because they’ve never had a close call. The trouble comes fast: a spill, a splash, a breath in the wrong direction. Some days, seeing hazards ignored leads to more worry than the substances themselves. It’s not rocket science—follow the basics, respect the unfamiliar, and pay attention to history. My own rule is never trust a shortcut with chemicals.
Public attention spikes after an accident, but fades quickly. Hazard awareness works best before trouble arrives. Training refreshers seem dull until someone forgets gaskets or mixes acids wrong. No fancy technology can replace common sense: store incompatible chemicals apart, label them clearly, and keep emergency gear within reach. People learn from mistakes, but much can be learned from the mistakes of others.
Rumors about chemicals run through every workplace. I’ve heard myths about substances being harmless, only to read later about their long-term risks in credible medical journals. Take benzene—a solvent many workers shrugged off decades ago, now recognized as a cause of leukemia. Hearing stories in break rooms can shed light on symptoms or accidents, but always check those stories against proper resources. The Environmental Protection Agency and Centers for Disease Control both offer reliable, research-backed guidance; it pays to use their materials as standard references.
Effective hazard awareness relies on habits built over time. Keep protective equipment ready and in good shape. Keep up with safety training. Develop a culture where asking questions about shelf lives and reaction risks earns respect, not ridicule. Encourage transparent reporting after incidents, no matter how small. Mistakes should start conversations, not cover-ups.
There’s no universal answer because context always shifts. What happens inside the controlled space of a university looks nothing like rush hour on a busy construction site. Relying on myths or luck won’t keep anyone safe for long. Familiar warnings don’t protect—preparation and ongoing skepticism do. The best workers I’ve known treat every new bottle, drum, or unlabeled container as a question worth asking, not a nuisance to ignore. The responsibility for handling hazardous chemicals starts and ends with each of us.
Ask any researcher or science teacher what gets kids interested in chemistry, and you’ll probably hear stories about color-changing reactions, odd smells, and a hint of danger from forgotten backroom experiments. Truth is, most of chemistry comes down to simple numbers and letters. That’s what you’ve got with a chemical formula and its molecular weight. It sounds dry, but these details drive real-world progress in ways you might not expect.
In my own work, I’ve watched entire projects stall because a team misidentified a compound’s formula. A single extra oxygen atom slipped into a sample meant a thousand-dollar batch didn’t work—and no one caught the mistake until the end. Formula errors lead to failed experiments, ruined supplies, and even safety hazards. Every molecule has its own rules. Water is H2O—everyone learns that early. Glucose, with its C6H12O6, fuels muscles and brains. Get even one number wrong, and either you’re making something else or you’re about to blow a fuse.
Molecular weight doesn’t just tell you how heavy a compound feels in a flask. It’s the key to figuring dosage, tracking chemical reactions, and lining up a recipe that works every single time. Heavy metals like mercury carry risks at even low concentrations. The same 1-gram dose delivers a very different punch depending on whether that gram is sodium chloride or lead nitrate. In pharmaceuticals and food production, accuracy saves lives. Just think about chemotherapy drugs—a slight miscalculation stemming from the wrong molecular weight can mean the difference between recovery and a toxic disaster.
Teachers drill formula memorization into students not because it’s fun, but because a foundation in details leads to better lab work. I’ve seen new chemists double-check formulas for hours, hunting down that obscure error left behind by someone too rushed to give a second glance. Trust is built through precision, and the industry keeps tight records because the smallest deviation might have legal, financial, or health consequences.
Anyone who’s spent real time in the lab will keep a few dog-eared reference books on hand. Trusted databases, like PubChem or ChemSpider, stay bookmarked in every browser window. Instead of copying formulas off the internet or scribbling them down from memory, most professionals cross-check with two or three sources before committing anything to a lab journal or a production floor. In big operations, teams sometimes hold short “formula reviews,” much like medical staff run “time-outs” before surgeries. These routines slow staff down, add checks, and stop errors before they lead to waste, or worse, injury.
Mistakes over formulas and molecular weight are easy to make but nearly impossible to fix after the fact. Every bottle in a cabinet, every powder in a bag—these have identities that demand respect. As someone who’s mixed, measured, and mopped up more than a few spills, I can vouch for the value of old-fashioned attention to detail. The numbers and letters might look small, but getting them right or wrong shapes the path every discovery takes.