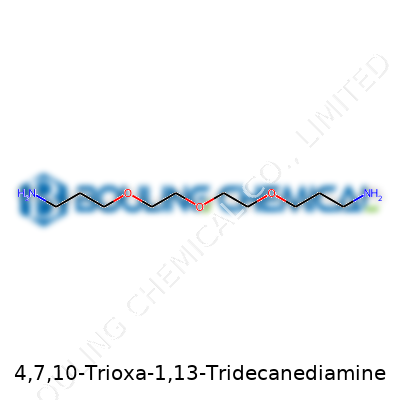
Years ago, the rise of synthetic chemistry led to a demand for compounds with both amine and ether functions. Researchers who once worked with simple diamines or polyethers soon realized that the merging of these two functionalities delivered new opportunities in materials science and formulation. 4,7,10-Trioxa-1,13-Tridecanediamine owes its existence to this push for versatile building blocks that move freely between hydrophilic and basic roles, offering manufacturers a broad canvas for innovation. My own experience watching colleagues solve tough polymer and surfactant challenges often led back to polyetherdiamines—tools that stitched together new networks, bridged gaps, and solved problems with subtlety that simple alkyl chains could never manage.
There is nothing flashy about the pale, viscous liquid that comes out of the sample bottle. Yet chemists who have used 4,7,10-Trioxa-1,13-Tridecanediamine soon learn that its three ether groups and terminal primary amines mean business. This molecule stands out as part of a family where chain flexibility, basicity, and water solubility cross paths. Mix up an aqueous solution and you see rapid blending, something that proves essential in adhesives and resins. The long, floppy structure lets the molecule maneuver where rigid diamines fail. In crosslinking, it often delivers surprising toughness.
Unlike volatile organics that fill the lab with fumes, this amine stays put in the flask. Its higher molecular weight and extended backbone keep vapor pressure down, minimizing exposure. The presence of oxygen atoms punctuating the chain makes the molecule more hydrophilic than plain alkyl diamines, which in turn means better compatibility with water-based formulations. The primary amines at both ends readily engage with epoxies, isocyanates, and acid chlorides, creating stable bonds and reliable performance. If you're looking for a greasy, stubborn liquid, you won’t get it. This compound rinses off with water and still holds its own in tough chemical settings.
Regulatory bodies look to accurate labeling, especially with specialty chemicals that find their way into chemical manufacturing. Labels indicate amine content, purity (often above 95%), and specifications for water content and color. These basic details forewarn about potential side products or contamination. In research settings, a chemist checks the batch certificate not out of bureaucracy but because trace impurities influence reactivity, shelf life, and downstream toxicity. The push toward sustainable chemistry also means scrutinizing synthesis routes and residuals, a trend driven by both regulators and cautious end-users.
Building this type of molecule isn’t a puzzle. The classic approach starts with polyether formation—joining ethylene oxide units with linking molecules to create the oxygen backbone. Terminal nitrile groups pop on, and selective hydrogenation follows. Now you have the desired diamine. What matters in the modern lab is process yield, control over side product formation, and whether catalysts will leave traces. Scaling up from a few grams in a round-bottom flask to drums in a pilot reactor always tests the ingenuity of process chemists. They push for minimal waste and efficient purification, often learning more from setbacks than successes.
The chemistry here swings between versatility and reliability. Those primary amines react swiftly with electrophiles. You can build polyurethanes or tough epoxy resins. Crosslinking doesn’t stall out, nor does it leave you with brittle results. More creative teams modify the molecule itself, adding alkyl groups or extending the chain to tune for hydrophobicity or molecular weight. Swap a few oxygens for other atoms, and properties shift—sometimes gradually, sometimes drastically. Over the years, I’ve seen groups create block copolymers from this core, bringing new performance to waterborne coatings. A thoughtful chemist respects not just the reactivity but the downstream effects—how small tweaks shape a final product.
The chemical industry rarely sticks to a single name, and 4,7,10-Trioxa-1,13-Tridecanediamine appears in vendor catalogs under several guises. Some call it polyetheramine D230, others prefer systematic or CAS terminology. For those ordering, tracking inventory, or reading literature, the tangle of synonyms can cause real confusion, especially across jurisdictions. In research, missing a reaction’s potential because of a naming slip-up is more than annoying—it puts time and resources at risk.
A molecule bearing amine groups, especially primary ones, always demands respect in handling. Cases of skin or eye irritation appear in safety reports, reminding lab workers to wear basic PPE. The same backbone that makes this amine effective translates to low volatility, so risks shift more toward spills or contact than inhalation. Spills wipe up with soap and water, but chronic exposure studies remain limited. Chemical suppliers supply guidance based on animal studies, but the real world rarely offers perfect data. For those operating reactors or mixing at scale, the focus lands on splash-proof gear, closed systems, and robust air handling. Experience shows that early adoption of rigorous hygiene pays off—no one wants to deal with unexplained rashes or headaches after hours in production.
Many see this diamine as a hidden workhorse. Epoxy formulation and curing become more predictable, giving rise to tougher adhesives and composite structures. In my direct experience, resin blends using this molecule resist cracking during thermal cycling or mechanical load. Polyurethanes built from this backbone show resilience in technological coatings or flexible foams. More progressive researchers look to surfactants or dispersing agents—areas where the ether groups anchor water solubility, while the amines offer stickiness for pigments or fillers. Medical applications remain rare, owing to regulatory caution, but the chemical’s behavior in biocompatible polymers earns it a place in academic journals. Rarely does a molecule find such a broad spread—construction, marine, electronic encapsulation, and paints all turn to polyetheramines for their subtle chemistry.
Innovation with this molecule traces to both public and private labs. Researchers dig for ways to recycle polymers that use polyetheramines, eyeing a future where circular manufacture beats landfill. Others dive into surface-active properties, developing new dispersants or emulsifiers from modified versions. Crosslink density, molecular weight tuning, and end group modification deliver not only superior materials but insight into structure-property relationships. In recent years, green chemistry has changed the research agenda, prodding teams to investigate biobased alternatives or safer process steps. Knowledge-sharing at conferences and in academic journals steadily scales up interest, sometimes revealing applications that industry never considered.
Nothing in synthetic chemistry slides by without toxicity scrutiny these days. Studies suggest 4,7,10-Trioxa-1,13-Tridecanediamine does not rank among the most hazardous diamines. It lacks the acutely reactive or carcinogenic traits found in aromatic amines. Yet researchers don’t relax: chronic low-dose exposure, allergenicity, and environmental persistence get checked in animal and cell culture assays. Waste handling and effluent release protocols point to the bigger picture—chemicals flow downstream, whether in diluted plant outflows or from spilled shipping drums. Regulatory authorities periodically recheck exposure limits, and producers lean into transparency, offering not just data sheets but ongoing research partnerships to fill knowledge gaps. As materials science keeps pushing new applications, toxicity research must not lag behind the market.
Chemistry built around 4,7,10-Trioxa-1,13-Tridecanediamine is hardly static. Future directions likely fold in smarter and more efficient preparation routes, delivering higher purity and lower energy use. Bio-based feedstocks attract attention as companies try to crack the code on renewable diamines with ether linkages. As consumer goods shift toward safer and greener profiles, demand increases for transparent additive traceability and risk assessment. Enhanced recycling routes, whether chemical or physical, appear on the R&D horizon—another move toward sustainable product lifecycles. As regulatory and consumer expectations rise, forward-thinking teams channel their expertise into both solving present material needs and designing molecules with safety and longevity in mind.
Walk into any facility where advanced materials are being developed, and you’ll find more than a few unpronounceable chemicals. 4,7,10-Trioxa-1,13-Tridecanediamine isn’t just another tough name to remember. This compound shows up where industries need more than just a simple amine. It’s a workhorse for chemists looking to build specialized polymers, grow coatings that stand up to tough environments, or create adhesives that last long after their first use. Most people don’t see this molecule on the shelves, but it still shows up in things we often take for granted.
For people who have spent time dealing with industrial applications—whether it’s in coatings, adhesives, or the manufacture of composite materials—the blending of functionality and stability is often the toughest challenge. 4,7,10-Trioxa-1,13-Tridecanediamine, with its ether linkages and amine groups, bridges gaps that simpler chemicals just cannot. On a technical level, those oxygen atoms improve solubility and flexibility, while the terminal amines help link things together during chemical reactions.
I’ve seen firsthand how companies introduce this compound into their epoxy hardeners and end up with coatings that fend off moisture and chemicals. Bridges, water tanks, pipelines—these structures demand coatings that don’t break down in a couple of years. Without high-performance polyetheramines, many of these protective layers would peel away much faster, leading to expensive repairs and even safety issues.
The wide scope of 4,7,10-Trioxa-1,13-Tridecanediamine goes beyond the lab. In adhesives, this compound lets manufacturers assemble electronics and vehicles with joints that keep their hold under stress and changing temperatures. A weak bond between parts might sound minor until that gadget stops working or, worse, someone faces danger on the road. In the field of composites—and I’m talking reinforced plastics for wind turbine blades and high-stress equipment—engineers need every bit of versatility from their raw materials. This compound supports that need, acting as a key ingredient that improves strength while allowing materials to flex instead of crack.
One area where 4,7,10-Trioxa-1,13-Tridecanediamine shines is in waterborne coatings. As environmental rules keep tightening, industries look for ways to reduce volatile organic compounds without sacrificing performance. Polyetheramines like this help replace older, more hazardous chemicals with options that give similar—or sometimes better—results. Less hazardous runoff and cleaner production floors benefit not just local communities but the global push for safer work environments.
No chemical comes without challenges. Handling and storage of amine-based compounds always need close attention. Employees need proper training and protective gear to avoid exposure issues. Companies also face questions about the end life of materials built with these ingredients. Recycling advanced composites or safely disposing of them becomes complicated, requiring ongoing innovation and regulation. More sustainable methods for making and reusing high-performance amines count as the logical next step. Green chemistry approaches, such as bio-based feedstocks or improved recycling techniques, could pave the way for a less wasteful future.
Through the decades, specialized amines have shaped safer roads, longer-lasting equipment, and more energy-efficient products. 4,7,10-Trioxa-1,13-Tridecanediamine stands out as one of those unsung contributors, behind the scenes but always making an impact. The more practical minds focus on responsible use and smarter sourcing, the more valuable these molecules become for everyone—scientists and consumers alike.
Chemical names often feel like a barrier, more like a puzzle than something that tells you how a compound works. But 4,7,10-Trioxa-1,13-Tridecanediamine isn’t just a string of syllables. Breaking it down, it might hit home for anyone who’s worked with polyetheramines or those deeply involved in materials science and industrial chemistry. The structure isn’t just an academic footnote — it has real consequences for how this molecule behaves, who uses it, and what comes next for the industries that depend on it.
This molecule has two amine groups, one at each end of a chain. Ether oxygens break up that chain at very precise places—carbon 4, 7, and 10, if you’re counting from one end. What does that actually mean? For starters, you get flexibility. As someone who’s spent hours looking for the right performance tweaks in epoxy formulations, I’ve seen what polyetheramines do to things like flexibility, water resistance, and ease of use. The backbone, spaced by ether oxygens, resists the rigidness you’d get from a plain alkyl chain. This lets the molecule act as a bridge, both physically and chemically, for a host of different reactions.
You won’t find this compound in a home supply store. Its value really shines in specialty manufacturing, coatings, and adhesives. I once worked with a team evaluating hardeners for epoxy floor coatings. Adding a polyether backbone (like the one in 4,7,10-Trioxa-1,13-Tridecanediamine) can change not just the cure time, but also the toughness and even resistance to chemical spills. That versatility comes from the molecular structure: long enough for flexibility, short enough to block phase separation, polar, yet not so polar that it ruins the material’s performance.
Downstream, companies push for greener chemistry and lower emissions. It’s no longer enough to just deliver on price or legacy performance. This compound’s structure makes it easier to fine-tune compatibility with water-based systems or to work at lower temperatures. Polyetheramines like this one have stepped up in creating less toxic, lower-VOC options in industrial formulations. The fact that you find these in everything from wind turbine blades to advanced coatings tells you they’re not just niche curiosities.
The other angle comes from safety. Any substance with amine groups raises eyebrows for those thinking about workplace exposure and environmental fate. Polyetheramines have improved profiles compared to smaller, more volatile diamines. Their size and structure limit volatility. For teams in charge of worker safety, that matters. It means less concern about airborne exposure during mixing and application. At the same time, responsible handling and disposal remain essential, a lesson learned by anyone who has ever run a materials shop or an R&D lab.
The journey of a molecule like 4,7,10-Trioxa-1,13-Tridecanediamine doesn’t stop with its chemical structure. It weaves into bigger stories—tougher composites, resins that last longer, and coatings that let infrastructure go further on the same budget. The drive to make materials smarter, safer, and more adaptable runs straight through the heart of molecular design. Chemists, engineers, manufacturers – everyone at the table knows the power hidden in the right scaffold of atoms. Structure isn’t just a name on a label; it’s the beginning of better solutions and, sometimes, disruptive change.
You don’t come across the name 4,7,10-Trioxa-1,13-Tridecanediamine in day-to-day conversation. This compound pops up in textbooks, chemical supply catalogs, and a handful of specialty manufacturing settings. For most people, that long name slips by pretty quickly. The issue isn’t the tongue-twister name, though. The real concern is what this chemical actually does to humans or the environment, and if handling it requires special care — beyond the latex gloves and goggles found in a high school lab.
For years, chemists and factory workers have known that most organic amines can be tricky. Many related substances irritate the skin, eyes, and lungs. After digging into regulatory filings and safety references, 4,7,10-Trioxa-1,13-Tridecanediamine doesn’t show up everywhere as a top hazard, but it brings similar risks to the table as other amine-based chemicals. That means possible skin and eye irritation and the chance of respiratory discomfort during exposure to high concentrations of vapors or dust.
I remember talking with a polymer chemist who worked long shifts with this family of materials. Gloves and face shields were a daily routine — not because they loved the look, but because accidental exposure brought on red skin and burning eyes. This isn’t just a matter of inconvenience; over time, repeated contact can sensitize people, leading to stronger allergic reactions. Industrial workers already know the drill: splash damage leads to stinging, and nobody wants to pass out from a lungful of strong amines.
Spills and leaks don’t just threaten people handling the chemical; they move through pipes and drains, winding up in streams and rivers. Some amines break down in water, but others linger, challenging water treatment facilities. I once worked alongside a team at a treatment plant — they dreaded industrial waste containing complex amines, since even small concentrations messed with aquatic life, threatening fish and insects that keep local ecosystems balanced.
Chemicals move through a web of rules, and agencies like OSHA, the EPA, and Europe’s ECHA look at facts from toxicity studies, accident data, and reports from the ground. According to the ECHA’s substance database, 4,7,10-Trioxa-1,13-Tridecanediamine appears on regulatory lists with warnings to keep it off the skin and out of the eyes, and to avoid breathing in the fumes. The EPA hasn’t flagged this specific molecule for the strictest controls, but its similar cousins have drawn attention for health and environmental reasons. That says plenty.
It’s easy to look at odd chemical names and feel distant from the risks, but these substances don’t ignore boundaries. Companies using this chemical need solid training for staff, enough safety equipment, and a plan for handling leaks or accidental contact. Storing it in sealed containers, labeling carefully, and keeping emergency wash stations at hand all make a difference. I’ve seen shops skip a step to save a few bucks, only for a single event to cost many times more in medical bills and legal headaches.
Reducing exposure wherever possible is smart policy, not only for health but also for keeping business smooth. It helps to push for greener substitutes where the chemistry allows. Plenty of science teams have looked at ways to redesign processes so these tough molecules aren’t required at all. Until then, everyone involved — from chemists to warehouse staff — needs real information, the right gear, and the freedom to speak up if something seems off.
4,7,10-Trioxa-1,13-Tridecanediamine is neither a household name nor the worst thing in the chemical world, but it deserves respect. Anyone working with or near this compound can’t afford to let their guard down just because it’s less infamous than more notorious toxins. That ordinary-looking bottle in the storeroom may not scream danger at first glance, but the smartest move is always caution, backed by clear-eyed facts and honest talk about risks.
Walking through any lab supply closet, I’ve seen the temptation to cut corners on storage. Odd-shaped bottles line up side-by-side, and labeling seems good enough. For specialty amines like 4,7,10-Trioxa-1,13-Tridecanediamine, those habits set the stage for costly mistakes. From firsthand experience, leaving proper storage an afterthought often leads to bigger headaches down the line—especially with reactive chemicals.
Many may overlook that trioxa tridecanediamine comes with a knack for soaking up water from the air. With hygroscopic chemicals like this, even short contact with humid air can lead to clumping or unexpected reactions, which then change physical properties and sometimes even their stability. Once, I noticed a bottle left open for a couple of hours and found the material inside had already started turning cloudy. This kind of oversight exposes staff and experiments to unpredictable risks.
Heat and light don’t play fair with amines. In a shared lab, a well-meaning colleague once left several reagents—including our trioxa diamine—on a sunny windowsill. After some days, unexplained smells started drifting from the bottle. We traced it back to low-level decomposition. By then, it was too late. The lesson became clear: storing this compound at room temperature away from direct sunlight and heat sources keeps its properties intact for longer periods.
Plastic may get the job done for some chemicals. For diamines, only high-density polyethylene or glass seem reliable. Thin plastics often show signs of softening, making leaks a real risk. I’ve seen the residue damage both shelving and cause enough odor to force unnecessary clean-up routines. Tightly capped containers matter just as much because the odor lingers, and open vapors are tough on sinuses.
It seems simple, but there’s a world of difference between a barely legible marker scribble and clear, robust labeling. During a routine audit, inspectors flagged half a dozen unlabeled bottles. Even seasoned chemists pause when they don’t know exactly what they are handling. Clear, permanent labels with hazard warnings keep people honest and safe.
Mixing incompatible chemicals just by keeping them side-by-side can cause reactions. Amines like trioxa tridecanediamine should never share storage space with acids or oxidizers. A shelf mix-up once led to a faint hissing noise and crusty residue—the sort of warning you don’t want to ignore in tight storage. Grouping similar chemicals and isolating reactive classes cuts the risk of unintended chemistry in storage.
I learned early on to treat every bottle as a spill risk. Having practical spill kits—absorbent pads, nitrile gloves, and ventilation—means the team acts quickly when something tips over. Just locking things away or relying on distant janitorial clean-ups isn’t enough. Practicing simple, repeatable responses to small spills builds confidence and keeps everyone on their toes.
Safe storage of 4,7,10-Trioxa-1,13-Tridecanediamine isn’t about following rules for the sake of compliance. It’s about respecting what these compounds can do when mishandled and valuing the safety of those sharing the workspace. Developing smart habits keeps everyone one step ahead—and keeps the day running without drama.
Too often, chemistry feels locked away in unreachable labs, far from regular folks. But that’s not the case. Chemicals touch our lives in ways we don’t always see. To keep all their names and forms straight, scientists use a unique system: the Chemical Abstracts Service (CAS) registry. Each compound gets a special number—no repeats. For 4,7,10-Trioxa-1,13-Tridecanediamine, the number is 4246-51-9. Behind those nine digits lies a lifeline for researchers and manufacturers who want clarity in a sea of chemical names.
CAS numbers help avoid confusion when dealing with chemicals that might have several nicknames or formulas. During my years working in laboratories and collaborating with folks in the coatings and adhesives field, I'd see the same material labeled five different ways, depending on which supplier or research paper someone read that morning. It takes only one mix-up to turn an ordinary experiment into a failed one. In a more industrial setting, mistakes with raw materials cost companies thousands or even shut down production if the wrong chemical gets delivered.
The CAS registry solves that headache. Instead of wrestling through pages of synonyms or vague chemical structures, anyone can punch in 4246-51-9 and get directly to what they need. Safety data, import documentation, regulatory records—these follow the CAS number, not just a fleeting product name. For universities, this consistency helps connect research groups across continents who might speak different languages but all respect the same identifier.
Let’s talk about the molecule itself. 4,7,10-Trioxa-1,13-Tridecanediamine isn’t as famous as many chemicals, but the class it belongs to—amines—plays a role in how paints, resins, and even pharmaceuticals wind up in their final form. From personal experience, working on polymer chemistry involves hours trying to nail down reagents that won’t throw off the results. The CAS number helped every step, removing guesswork and saving time. In safety protocols, eyes light up with relief when you quote the number, not just the name, since hazard databases always start with those digits.
Behind the scenes, regulatory compliance offices use these identifiers each day. Many countries require these numbers to be listed on customs documents for import and export. Sometimes, an unfamiliar supplier lists “Jeffamine ED-2001” as an ingredient, but a quick CAS lookup reveals it matches 4246-51-9—the puzzle piece slides into place, audit paperwork gets filled, and the shipment moves forward.
Real trust in science, medicine, and industry depends on clear information everyone can verify. CAS numbers look dull, but they keep dangerous mix-ups at bay. In my own projects, using the CAS number always settled debates and avoided mistakes that would have otherwise slipped through. If everyone used these identifiers more intentionally, both novice chemists and seasoned professionals could spend less time sorting out confusion and more time doing real work.
It pays to remember: details like a CAS number aren’t just for paperwork. They anchor honest discovery and keep products safer from the lab bench to store shelves. For 4,7,10-Trioxa-1,13-Tridecanediamine—CAS 4246-51-9—that single string of numbers does more than tag a chemical: it keeps research accurate and production responsible.