Many breakthroughs in synthetic organic chemistry come from tireless curiosity. 3,4,5,6-Tetrafluorophthalonitrile did not just appear overnight. Its roots follow back to mid-20th-century research exploring the fine-tuning of phthalonitrile derivatives. Early fluorination methods required determination and patience, especially since direct fluorination often yields unpredictable and sometimes explosive results. Over the years, chemists built their skillsets, learning to work safely with elemental fluorine and developing more refined nucleophilic methods. Each improvement enabled better access to these highly fluorinated compounds. Decades of academic work, coupled with the needs of the electronic and pigment industries, made the large-scale preparation of tetrafluorophthalonitrile practical and relatively affordable. Today, this compound sits on the shelf thanks to centuries of progress in both safety and process control, standing as proof that steady development outruns shortcuts.
3,4,5,6-Tetrafluorophthalonitrile grabs attention in research circles for one big reason—its structure unlocks new classes of materials. By swapping out four hydrogen atoms for fluorines on the phthalonitrile ring, chemists get a starting point for synthesizing phthalocyanines, valuable dyes, pigments, and advanced materials. Its straightforward structure belies a world of complexity in terms of reactivity and utility. Through the years, I have learned that such modifications never just tweak properties a little bit. Small structural changes, especially with something as powerful as fluorine substitution, can send stability, reactivity, and bulk material performance in completely different directions. Tetrafluorophthalonitrile gives researchers a tool to walk those paths, exploring strong electron-withdrawing effects and fine-tuning for specific applications.
From my experience working in fluorochemical synthesis, this compound appears as a white to off-white crystalline powder at room temperature. Don’t let its appearance fool you; the four fluorines make a big statement chemically, imparting remarkable thermal stability and resistance to acids and bases. Electronic effects play a serious role—by pulling electron density away from the aromatic core, fluorines stabilize the molecule and shift reactivity, especially in nucleophilic substitution reactions. This means extended shelf life, stable storage, and the kind of predictable behavior coveted in synthetic chemistry. If you have ever handled similar compounds, you know that low impurity levels matter, as side products can influence how well downstream reactions run. Manufacturers must take care with purification and drying because even small traces of water or unreacted materials can alter the final performance of this intermediate.
Chemists demand clarity about what they work with, and tetrafluorophthalonitrile comes with detailed labels: purity percentage, batch date, physical form, and specific hazard pictograms. Quality control is tight—you can’t afford off-spec materials here, especially in electronics or pigment synthesis. Regulatory compliance stretches beyond the bottle. Suppliers test for residual solvents, heavy metals, and moisture. One time in the lab, a supposedly minor deviation in melting point signaled a contamination issue that eventually traced back to an upstream solvent switch. That reinforced for me how essential it is to verify every part of the production and labeling process, starting with the right chemical structure and ending with rigorous documentation.
Most routes to 3,4,5,6-tetrafluorophthalonitrile use nucleophilic substitution. Starting with tetrafluorophthalic anhydride or the acid itself, you can activate carboxyl groups and convert them to nitrile units, all while keeping the four fluorines snugly in place. Choose the wrong solvent or let water creep into the system, and you risk hydrolysis or unwanted defluorination. Years spent around large reactors and Schlenk lines show that thermal management and clean reaction vessels are not optional in these syntheses—impurities, even in parts-per-million, can force entire batches into costly rework. Synthesis at scale also highlights the practicalities of waste management; fluorinated side products cannot just be dumped down the drain. Recycling and scrubbing systems run all the time, keeping the process both environmentally and economically sustainable.
This compound shows serious flexibility for further functionalization. Its activated aromatic ring, bristling with four fluorines, is primed for nucleophilic aromatic substitution where you can swap out a fluorine for a wide range of nucleophiles: amines, alkoxides, and even thiols. That opens a door to almost endless custom modifications, whether the goal is creating novel polymers, tuning optical properties, or tailoring charge transfer for electronics. In my career, seeing the jump from bench-scale substitutions to commercial volumes never fails to impress. Side reactions haunt these systems if chemists skip reaction optimization—side-chain byproducts, incomplete substitutions, and polymerization can pop up if handled carelessly. Still, for patient innovators, tetrafluorophthalonitrile gives a base molecule that can grow and adapt to whatever the latest scientific needs may be.
One thing that always trips up new researchers? The maze of chemical synonyms. 3,4,5,6-tetrafluorophthalonitrile also surfaces under names like tetrafluorophthalonitrile or TFPN, and I have even seen it listed as perfluorophthalonitrile, despite that being a little misleading. Careful recordkeeping matters—a quick typo or ambiguous order can land the wrong compound on your bench. Journals and catalogues increasingly stress the use of CAS numbers, and that helps keep procurement honest. No matter the name, the underlying chemistry drives its place in research and manufacturing.
Every researcher I know develops a healthy respect for fluorinated organics. The nitrile groups add their own layer of risk, especially if strong acids or bases are in play. Inhalation or skin exposure brings acute toxicity concerns; nitrile vapors can cause respiratory irritation, and there is always the background worry of cyanide release during thermal decomposition. Gloves, goggles, fume hoods—none of these count as optional. Facility managers must keep detailed records for storage and handling, and proper waste disposal is an ongoing challenge. Over the years, I have watched labs invest more heavily in regular safety training, updated chemical inventories, and swift incident response protocols. This isn’t just bureaucracy—it genuinely reduces near-misses and long-term health issues, especially when dealing with persistent organic pollutants. The industry’s move toward transparent safety metrics and fully disclosed safety data keeps everyone accountable, which is the right direction.
3,4,5,6-Tetrafluorophthalonitrile mainly shines as a building block for phthalocyanines. These molecules color our world—literally—showing up in pigments for everything from car coatings to banknotes. But the impact goes well beyond color. With tailored substituents, phthalocyanines serve as organic semiconductors, sensors, and even as materials in advanced solar cells. In my work with material scientists, the demand for sturdy, high-performance organic frameworks never dies down. The fluorination pattern here helps bump up electron mobility and push thermal limits, meaning devices or pigments last longer in tough environments. Electronics manufacturers prefer its purity for OLED and printed circuit board applications, where stability and batch-to-batch repeatability drive yields. It is a tool that finds new uses each time science throws down a new challenge.
Academic groups constantly circle back to tetrafluorophthalonitrile for new ideas. Recently, green chemistry initiatives have rewritten old playbooks, aiming for solvent-free or low-waste synthesis that still delivers high yields. In major chemistry journals, studies using this molecule read like cooking shows—step-by-step transformations yielding everything from new dyes to molecular sensors. Graduate students dive into structure-property relationships, publishing detailed work on how each new substituent impacts color, charge, or binding strength. Industry R&D teams chase better process economics, pilot-testing catalytic systems or exploring continuous flow reactors to trim costs and improve safety. At conferences, it’s clear discussions never circle around “if” this molecule will matter, but “how” it will fit next.
Much debate surrounds the health and environmental impact of fluorinated organics, and 3,4,5,6-tetrafluorophthalonitrile is no exception. Tetrafluorinated aromatics tend to resist biological breakdown. Toxicologists focus on monitoring its fate after accidental spills and checking for bioaccumulation, especially as industrial use grows. Studies look at acute toxicity endpoints, eye and skin irritation, and long-term inhalation effects. Most reports peg it as moderately hazardous—bad news if mishandled, manageable if strict controls are followed. New testing data from rodent studies, coupled with computer modeling, provide more peace of mind to researchers and regulators. As global regulations on perfluorinated compounds tighten, labs keep close tabs on not just human but ecosystem exposure, weighing every finding before greenlighting new applications.
Fluorine chemistry almost always points to rising demand. With the continued thirst for better organic materials in electronics, renewable energy, and smart coatings, 3,4,5,6-tetrafluorophthalonitrile remains a critical ingredient in innovation. Ongoing work on sustainable synthesis routes, whether by cutting hazardous waste or eliminating rare metal catalysts, aims to green the supply chain. Chemists eye opportunities for deeper recycling and reuse, trying to loop valuable intermediates back into manufacturing rather than letting them become single-use products. As research paves the way for new functions—biomedical imaging, advanced proton conductors, and flexible electronics—more companies will need access to this compound. I have seen how robust international collaboration hastens these advances, since no single lab or supplier owns every breakthrough. The molecule’s story is far from written, and the coming decades will surely add new chapters no one predicts today. In my view, the most exciting science always begins with thoughtfully prepared starting materials, and 3,4,5,6-tetrafluorophthalonitrile belongs high on that list.
3,4,5,6-Tetrafluorophthalonitrile might not sound like an everyday word, but in the chemistry world, it carries weight. Years spent in the laboratory taught me that every complex name in chemistry usually has a fascinating reason for being important. This compound belongs to a group called phthalonitriles, notable for their ability to build much larger and more complex structures. For years, the primary use for this compound has revolved around the creation of phthalocyanine dyes and pigments. Scientists and engineers depend on these dyes because of their stability, deep color, and resistance to fading, even under strong sunlight or harsh chemicals.
This molecule doesn’t just get thrown into dye vats. Its power comes from the four fluorine atoms locked onto its ring structure. That specific arrangement allows scientists to tweak the chemical's reactivity. Because of this, 3,4,5,6-Tetrafluorophthalonitrile acts as a cornerstone for crafting phthalocyanines that break the usual boundaries of what traditional organic dyes can achieve. Think of industrial coatings on bridges, roofs, or even cars—environments where most colors would break down after a few years. The dyes made from tetrafluorophthalonitrile keep their color and hold up to environmental punishment. As a chemist, I saw firsthand how coatings using these materials made a difference where longevity mattered.
The story doesn’t stop at dyes. As technology marched forward, research stretched the reach of this simple-looking nitrile. By carefully piecing it together with metals or carbon atoms, engineers could design materials for organic solar cells, sensors, and printed electronics. The heart of these products lies in their thin, flexible design—something that would be impossible without specialty molecules like tetrafluorophthalonitrile. Every time I peel a smart label or see solar panels blend into building facades, I remember how foundational molecules help make that leap from idea to product.
Every powerful chemical brings responsibilities. The presence of multiple fluorine atoms raises red flags if not handled the right way. Chemists have a duty to keep these substances inside controlled spaces, use rigorous containment, and prevent unintended release into waste streams. Attention to safety through robust protocols, careful waste disposal, and wearing protective gear isn't just smart science—it’s an ethical commitment to public health and the environment.
The future will keep pushing chemists to develop even safer and more efficient molecules. Research into alternatives, better purification, and recycling of spent raw materials can reduce risks and keep operations green. In the hands of thoughtful experts, compounds like 3,4,5,6-Tetrafluorophthalonitrile will keep building bridges between innovation and responsible science—delivering value to both people and the planet, not just the laboratory bench.
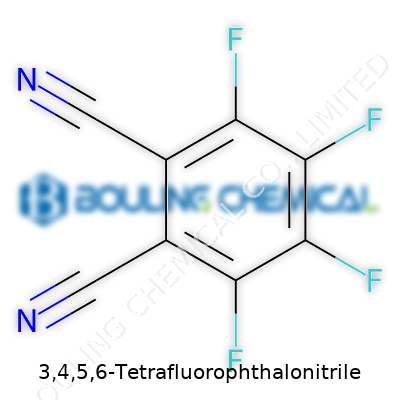
You don’t need to be a seasoned chemist to get a sense of how names like 3,4,5,6-tetrafluorophthalonitrile come about. The “phthalonitrile” part points toward a benzene ring holding two –CN (nitrile) groups on opposing ends. The “tetrafluoro” prefix means four fluorine atoms attach to the ring, specifically at positions 3, 4, 5, and 6. In real terms, the chemical structure starts with a benzene core, tosses out four hydrogens, and replaces those with fluorine atoms. Two cyano groups clip onto carbons across from each other, usually positions 1 and 2.
Getting your head around chemical structures isn’t just academic nitpicking. The placement of those nitrogen and fluorine atoms dictates how this molecule behaves. Fluorine, thanks to its small size and extreme electronegativity, transforms the ring, tightening up the electron cloud. As a result, the molecule resists chemical attack—a trait folks rely on in high-performance dye manufacturing and material science.
The nitrile groups aren’t just wallflowers here, either. Those triple-bonded nitrogens act like anchors for further chemistry, especially in producing phthalocyanine dyes. These dyes play a role in everything from solar technology to inks, and the performance bump you get by adding fluorine shows how tweaking structure leads to practical gains.
In the lab, building molecules with multiple fluorines is no picnic. I’ve seen first-hand how fluorinated aromatics almost shrug off reactions that work fine for plain cousins. That stubbornness comes from the electron-withdrawing effect fluorine piles onto the ring. Chemists have to adapt, switching to tougher conditions, or else use specialized catalysts. This isn’t just for fun—industry wants stable materials that handle heat or corrosive environments.
People might see chemicals as abstract drawings, but the story of tetrafluorophthalonitrile connects to daily products. The electronic properties it brings tweak the color, light absorption, and longevity of pigments. In solar panels and electronics, stability means fewer breakdowns and better efficiency. Consistent, reliable products flow from these molecular choices.
Not every improvement comes for free. Synthesizing highly fluorinated compounds often leads to handling stricter safety protocols, both in academic and industrial settings. Waste management becomes a bigger deal, as fluorinated byproducts sometimes linger in the environment. That teaches a clear lesson—future chemistry should pay just as much attention to greener methods as to performance.
Efforts in green chemistry already push for smarter processes. Some teams are turning to milder reagents, alternative solvents, or even bio-based starting materials for aromatic molecules. Fresh thinking around recycling and upcycling fluorinated materials opens more doors down the road. Industry gains, if balanced with environmental responsibility, lay out a better path forward.
The core structure of 3,4,5,6-tetrafluorophthalonitrile hints at a lot more than just atoms and bonds. It ties together progress in dyes, electronics, and sustainability. The lessons I’ve learned remind me that the tiniest changes at the molecular level ripple outward, guiding what we see in daily tech and how we manage its impact. For scientists, engineers, and anyone curious about materials, following the journey from structure to application reveals how chemistry matters—at every level.
3,4,5,6-Tetrafluorophthalonitrile isn’t exactly sitting on an average garage shelf. Labs use it for specialty synthesis—usually in making advanced chemicals like phthalocyanines. Handling this solid means taking real-life safety steps. My time in research labs taught me that few things spell trouble like ignoring basic storage rules for niche organic chemicals.
This compound packs both fluorinated and nitrile groups. One thing folks might overlook is its sensitivity to moisture or acids. Water in the air will speed up hydrolysis, turning your pristine chemical into something unusable. Before long, you’ve ruined your stash or made it risky to touch. I kept desiccants in every container when I worked with anything similar.
Glass bottles, tightly sealed, tend to be the best option. Polyethylene lets in more moisture than you think. Keeping the bottle in a desiccator draws out extra humidity. That isn’t just nice-to-have—that’s the line between an easy day in the lab and a wasted week of ordering more material.
Extreme cold is often a crutch when folks feel uncertain. Sticking everything in a freezer won’t fix careless habits. For 3,4,5,6-Tetrafluorophthalonitrile, room temperature storage works as long as the area stays dry and well-ventilated. No need to crowd the cold storage unless your workplace deals with extreme conditions or unusually high humidity. A research colleague once rushed to freeze everything “to be safe,” only to cause condensation in the jar later and lose the lot.
A dry, stable, and clean shelf away from sunlight gives you better results. Heat speeds up degradation—no one wants a mystery goo at the bottom of their bottle, especially if that bottle cost a fortune or came with a long lead time.
Most lab accidents happen because people take shortcuts or skip labeling. All it takes is someone grabbing the wrong jar, or not wearing gloves, due to faded labels or misplaced stock. My memory holds a moment where a bottle marked with a sloppy label led to three days of sorting out sample contamination. Neat, clear labeling matters at every step, for every jar. Add date received and date opened, so shelf life doesn’t slip past.
Double containment keeps odors trapped, and cuts down spill risk. This means placing the sealed bottle in a secondary container. The habit pays off in day-to-day peace of mind. I’ve seen a chemical store-room cleaner who thanked us for that practice, saying spills used to be the top safety issue before double-containment became the standard.
Policy updates on chemical storage connect directly to lab safety culture. Regular training with real examples—less “read the manual,” more “this saved me from a health emergency”—sticks with people longer. Safety equipment, like gloves and goggles, can go unused unless folks see their coworkers using them. Team leaders have to set the pace.
Chemical storage isn’t glamorous work. It’s all about habits honed by routine, backed up by the right containers, and kept alive by a shared sense of responsibility in the lab. From rookie bench scientist to old-timers, everyone benefits when this sense of practical care pushes out shortcuts and keeps compounds like 3,4,5,6-Tetrafluorophthalonitrile both safe and useful.
Working with chemicals always keeps me on my toes. 3,4,5,6-Tetrafluorophthalonitrile isn’t much different—except, maybe, a bit more sensitive compared to the usual suspects in the lab. This stuff packs both nitrile groups and a fluorinated aromatic ring, and those features mean you have to handle it with some guarded respect.
My introduction to tetrafluorophthalonitrile came in a workshop on specialty polymers. A chemist friend mentioned the combination of toxicity and chemical aggressiveness. The molecule’s structure lets it act as an irritant, and even low exposure can trigger health effects. Some folks react to even small amounts. The fluorine atoms, helpful in high-performance plastics, also lead to stubborn environmental persistence.
A study from the CDC spotlights nitrile compounds as respiratory hazards. Add in the known perils of aromatic fluorides, and the case for unwavering caution grows clearer. No one shrugs off a compound that can hit the lungs, skin, eyes, and maybe even the nervous system after extended exposure.
People sometimes roll their eyes at PPE reminders, but I haven’t met anyone who enjoyed a chemical burn or a trip to the doctor after a fume hood mishap. Splash goggles, gloves rated for chemical resistance—nitrile or even thicker—plus a sturdy lab coat form the minimum kit. Forget latex or bare hands. This chemical doesn’t mess around with skin contact.
Pure air matters just as much. Shortcuts with ventilation create problems fast. Even trace vapors should prompt a trip to the fume hood or a properly vented glovebox. Swallowing fumes won’t make you tougher—and the health cost can linger for weeks. In labs I’ve worked, a strict no-eating, no-drinking policy exists anywhere chemicals like this show up. One day-old coffee left near an active bench could become a contaminated hazard.
Some of the best advice I got on chemical safety came from a tired senior tech: “Clean as you go, or it’ll catch up to you.” This advice rings especially true for tetrafluorophthalonitrile. Spills and splashes need prompt cleanup with specific absorbent materials—not just a handful of paper towels. Skin exposure deserves a full rinse, no hesitation. The material doesn’t give second chances.
Waste management isn’t just a regulatory headache—it’s about not poisoning yourself or the ecosystem. Mark containers clearly. Mix with compatible solvents if needed and store waste away from heat or light. Colleagues depend on everyone’s attention to these details. No waste gets forgotten in a back corner.
No one learns chemical handling by reading a page of instructions. Lessons stick through careful mentorship, drills, and watching out for each other. I’ve watched smart folks become careless out of routine. An open-door approach—where anyone can call out unsafe moves—makes for fewer horror stories. Sharing what went wrong, as well as what worked, keeps the team sharp and humble.
Tetrafluorophthalonitrile won’t forgive laziness. The tools and systems needed to handle it safely aren’t a nuisance—they’re a matter of keeping your health, your job, and maybe the health of your family. Respect beats bravado, every single day.
3,4,5,6-Tetrafluorophthalonitrile is not a compound the public hears much about. This little molecule means a great deal to chemists who work in materials science, electronics, or fine chemical synthesis. Packs of the stuff show up in research labs and industrial facilities, sitting quietly like unsung heroes waiting for the right reaction. The catch with 3,4,5,6-Tetrafluorophthalonitrile has never really been about availability. The real story circles around purity, consistency, and why both play an outsized role in real-world applications.
I once worked in a lab where a single bad batch of a reagent derailed three months of work. Impurities turned up, unexplained results hit our notebooks, and our reaction yields dropped just enough to trigger a flood of troubleshooting. With 3,4,5,6-Tetrafluorophthalonitrile, most labs hunt for a minimum of 98% purity, with reputable vendors often advertising purities up to 99% or even 99.5%. Anything less than this, and you start to see unpredictable by-products, sluggish reaction rates, or worse—work that can’t get published.
A compound’s grade gets even more critical in fields like electronics or pharmaceuticals, where a contaminant at a fraction of a percent can wreck a batch. Labs holding projects together with shoestring budgets may settle for a lower grade during initial studies, but those chasing precision know that even trace levels of water, acids, or unknown aromatic compounds disrupt sensitive processes.
What you get from a bottle of 3,4,5,6-Tetrafluorophthalonitrile owes a lot to how it's produced and purified. Companies with rigorous quality control analyze every lot using NMR, HPLC, or GC-MS to guarantee what’s listed on the label. Without this, buyers are left guessing, and those guesses come with real financial and reputational risks. Some manufacturers cut corners, skipping additional re-crystallization steps or diligent solvent removal—choices that haunt the end user. Quality assurance isn’t a marketing bullet; it’s the foundation that stops research from spiraling in the wrong direction.
Researchers everywhere want better transparency from suppliers. Detailed certificates of analysis, lot-to-lot consistency reports, and honest impurity profiles win trust quickly. These simple documents build the backbone of EEAT (Experience, Expertise, Authoritativeness, Trustworthiness) in science publishing and product development. No lab wants to dig for answers about what’s lurking in the bottle. Investing in reliable analytics pays off by preventing failed projects, wasted time, or disastrous scaling-up surprises.
Some groups purify their own batches if spot checks reveal hidden contaminants. Recrystallizing or filtering under dry atmospheres knocks out a lot of trouble. Power users also pass on tips—sourcing the same brand and lot for repeat projects, testing a reference batch before big experiments, and log every hiccup, no matter how minor.
Most scientists don’t expect miracles. What they want is honesty about what’s in the bottle and the ability to chase answers if results aren’t adding up. This is where science gets personal: anyone who’s lost hours or days tracing nagging impurities understands that purity isn’t a luxury. For 3,4,5,6-Tetrafluorophthalonitrile, getting the grade right doesn’t just save money; it gives research the best fighting chance at discovery. Labs win. Data becomes reliable. Progress actually happens.