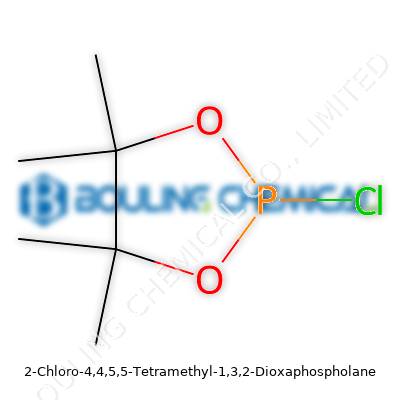
Chemistry has a way of dragging new compounds out of the laboratory and into the center of real-world progress. With 2-Chloro-4,4,5,5-Tetramethyl-1,3,2-Dioxaphospholane, you can see how incremental improvements stack up across decades. Back in the 1970s and 1980s, the search for phosphorus compounds that offer reactivity and better control in organic synthesis gained traction. As research moved away from traditional phosphorus sources—think red phosphorus, phosphorus trichloride—and toward clever ring systems, dioxaphospholane derivatives grabbed attention. This compound didn’t just pop into existence one morning; it carries the legacy of chemists tinkering with the dioxaphospholane ring to balance stability with utility, safety with excitement. The road to this molecule passed through a tangle of substitutions and ring-size experiments, picking up lessons from each detour. Today’s version serves as a marker of just how far phosphorus chemistry has traveled and reminds us that discovery is a cumulative effort, marked by the hands-on lessons of the bench and fume hood.
Look past the mouthful of a name—2-Chloro-4,4,5,5-Tetramethyl-1,3,2-Dioxaphospholane has earned respect among synthetic chemists. Its balanced structure, built on a five-membered ring with good crowding from methyl groups, finds uses ranging from reagent in analytical chemistry to a unique building block in organic synthesis. This isn’t just a silent player for one-off reactions, either. The compound’s profile draws on the best of phosphorus ring systems, showing up in nerve agent detection, advanced polymer modification, and specialty flame retardant research. By handling both nucleophilic substitution and controlled ring-opening, it stays adaptable when tools are needed that push reaction scope without derailing the safety train. For anyone who has wrestled with finicky phosphorus chemistry, finding a reagent that delivers both reactive punch and manageable handling can feel like a revelation.
If you’ve ever popped open a vial, you know this: 2-Chloro-4,4,5,5-Tetramethyl-1,3,2-Dioxaphospholane isn’t screaming for attention. Its clear to slightly yellowish liquid form doesn’t stand out on the shelf, but its subtle odor hints at a reactive phosphorus center. With a melting point below room temperature and boiling above 100°C, it sidesteps the volatility and hazards of many other chlorophosphorus reagents. It handles moisture with the usual wariness demanded by chlorinated phosphorus rings, hydrolyzing to release hydrochloric acid and forming the corresponding phosphonic acids. But there’s value in that controllable reactivity. The methyl groups shield the ring from runaway decomposition, and the phosphorus-chloride bond acts like a handle for targeted substitution. The density, refractive index, and viscosity all line up with a midweight organic liquid, so it pours and transfers like standard solvents—though gloves and eye protection stay non-negotiable.
Anyone who’s spent time working with regulated chemicals has run the labeling gauntlet, and this compound comes with the usual flags: a risk of skin and eye irritation, potential for inhalation hazard if handled without care. Product containers typically promise a purity upwards of 97%. Regulatory tags line up with other phosphorus-chloride reagents, with the usual language about keeping away from open flames, strong bases, and sources of moisture. That standard doesn’t trivialize the risks—it just signals a mature understanding. Trust in a product starts with transparency, so don’t cut corners: suppliers publish MSDS sheets that spell out storage in tightly closed containers under inert gas, disposal methods, and ventilation cautions. Every bottle you unscrew comes with a stack of trackable information, keeping both scientists and facilities out of regulatory crosshairs and building trust through full disclosure.
Synthesizing 2-Chloro-4,4,5,5-Tetramethyl-1,3,2-Dioxaphospholane rarely ranks as a DIY project for the amateur chemist—and that’s a good thing. Most commercial processes use the reaction of phosphorus trichloride with pinacol or tetramethylethylene glycol, managed under controlled cooling and dry, inert atmosphere. The heart of the method involves ring closure, with precise temperature and order-of-addition steps backing up any claims of reliability. HCl gas gets vented safely or trapped, sometimes in alkaline scrubbing setups. Filtration and distillation round up a product that’s both clean and true to form. All this effort ensures reproducibility and purity, and blends well with the growing push towards greener chemistry: minimizing waste and watching energy costs, not just chasing higher yields.
Chemists value a tool that adapts. One unique aspect of this dioxaphospholane is how it swaps out the ring-bound chloride in nucleophilic substitution reactions. Alcohols, amines, and thiols can all kick out that chlorine under gentle conditions, building new phosphonates and phosphoramidates—critical in organic synthesis targeting pharmaceutical or agrochemical groups. Open up the five-membered ring and fresh routes to phosphorus acids or esters emerge, which comes in handy for fine-tuning ligand design and catalyst development. Its balanced reactivity makes it popular in research labs pushing into new realms of phosphorus ligand chemistry, and the structure tolerates a fair bit of improvisation as chemists try new protecting groups or catalytic applications. Having worked with phosphorus-based reagents, the appeal lies in reliable, predictable transformations instead of surprises in yield or byproduct.
The chemical marketplace runs on both clarity and confusion when it comes to names. Some suppliers go with "2-Chloro-1,3,2-dioxaphospholane, tetramethyl- derivative" or just "chlorodioxaphospholane," lumping it together with similar ring compounds. Slight modifications in labeling—4,4,5,5-tetramethyl or TM-DXP—may appear depending on catalog or supplier geography. This tangle of synonyms reinforces why researchers lean on CAS numbers or standardized formulas, sidestepping translation errors that cost in time and, sometimes, safety. Familiarity with a compound’s aliases can help dodge ordering mistakes and streamline research progress; spending five minutes on label logic saves hours in the storeroom or the literature.
Handling phosphorus chlorides strips away any illusions about shortcutting lab basics. Even with the friendlier properties of 2-Chloro-4,4,5,5-Tetramethyl-1,3,2-Dioxaphospholane, experience has proven that full PPE, glove checks, and working in a certified fume hood never go out of style. Even brief skin or eye contact brings strong irritation, and its hydrolysis generates hydrochloric acid fumes. Labs that value long-term health set up procedures for spill management, fresh air flow, and regular safety audits. Emergency eyewash stations and robust waste management systems back up operational discipline. Over years in the lab, being careless—just once—has caused more headaches than all the careful prep combined. There’s rarely a second chance if you let routine slip with phosphorus reagents.
Application gives this compound more heft than its name suggests. In analytical chemistry, it transforms the landscape of phosphorus-31 NMR as a staple derivatization agent, helping quantify alcohols and other nucleophiles in complex mixtures. Organic chemists pull it into phosphonate ester synthesis, drawing on its ring system as both scaffold and leaving group. Material science researchers see potential in grafting onto advanced polymers, tuning flame retardancy or dielectric properties without losing processability. Its structure appeals in modifying biological probes, expanding the toolbox for enzyme tagging, or drug-delivery research. Regulatory scientists lean on its reproducible hydrolysis for testing safety in environmental labs. Years of development have dialed up the reliability factor, so researchers trust it when pushing analytical or synthetic boundaries that matter to the marketplace and to basic science.
Academic and industrial labs continue to dig into this compound’s reactivity and versatility. Structure-activity relationship studies stretch its applications in catalysis and ligand design, while new protocols in green chemistry seek to cut out hazardous byproducts in its synthesis. Analytical teams compare it against traditional phosphorus reagents, leveraging better sensitivity in NMR tagging and increased stability in storage. Cost and sustainability pressures ask for alternatives to older halogenated phosphorus chemistry, nudging progress toward derivatives based on renewable feedstocks or less toxic chlorinating agents. Workshops and publications push ahead on fine-tuning selectivity, especially for pharmaceutical intermediates and high-purity specialties. Each round of development feeds back lessons into undergraduate and graduate training, laying groundwork for safer techniques and more effective uses in tomorrow’s labs.
Toxicological profiles shape more than just safety checklists—they drive regulatory frameworks and translate benchwork into real-world use. This dioxaphospholane, though less notorious than some phosphorus cousins, still carries acute risks. Inhalation, skin absorption, and accidental ingestion each bring harsh effects: corrosive damage, respiratory irritation, and headaches line the short-term list. Chronic exposure studies suggest caution, as organophosphorus compounds share potential for cumulative toxicity. Environmental breakdown products, including organophosphonates and hydrochloric acid, push regulatory agencies to demand routine monitoring and solvent-management practices. Lab protocols and workplace exposure limits exist for good reason. Ignoring early warning signs creates a legacy of long-term health problems that stack up, both for individuals and for the full community of chemical workers.
Advances in chemical sustainability and automation promise new directions for compounds like 2-Chloro-4,4,5,5-Tetramethyl-1,3,2-Dioxaphospholane. As synthetic chemistry leans harder on greener, more efficient reactions, demand grows for phosphorus reagents that minimize waste and maximize control. Envision new synthesis routes based on catalytic, rather than stoichiometric, phosphorus chemistry. Digital workflows, AI reaction planning, and automated purification may untie bottlenecks in custom molecule production, making specialty dioxaphospholane derivatives accessible even to small labs. Money and regulatory pressure keep pushing for closed-loop manufacturing, better recycling, and sharper toxicity monitoring. If academic-industry partnerships stay strong, expect this family of compounds to help redefine what’s possible in pharmaceutical, material, and analytical chemistry—keeping one eye on safety, and the other on real innovation that raises the bar for every future chemist.
Walk through any lab working on polymers or cellulose, and you might run into a bottle labeled 2-Chloro-4,4,5,5-Tetramethyl-1,3,2-Dioxaphospholane. It's a mouthful, but behind that name sits a chemical tool that quietly supports fundamental research and industrial processes, mostly as a reagent for analyzing the building blocks of all kinds of materials.
In practical terms, this compound finds its most important action in the hands of chemists running phosphorus-31 nuclear magnetic resonance (31P NMR) studies. Most folks outside the lab may not know that NMR isn’t just for organic compounds; adding a phosphorus label opens the door for a whole new way of looking at things like hydroxyl groups in cellulose, lignin, and other polymers. By reacting with alcohol and acid groups, this reagent helps scientists "tag" them, so their chemical neighborhood becomes visible under NMR.
If you’ve ever worked with plant fibers or tried to modify wood, you probably understand why that tagging matters. Lignin and cellulose have tricky structures that dictate how strong a paper sheet feels or how eco-friendly a new plastic becomes. To figure out how a chemical treatment changes these materials, researchers run into the same roadblock: too many types of hydroxyls, all hiding in a maze. Using this dioxaphospholane reagent, they can measure specific sites, count how many free hydroxyls are present, and track changes during treatments. That leads directly to smarter recipes, less wasted effort, and better end-use performance.
For anyone tracking green chemistry, this sort of analysis isn’t just academic. Reducing waste or figuring out a bio-based alternative to petroleum products depends on understanding the basics. A seemingly small tool like 2-Chloro-4,4,5,5-tetramethyl-1,3,2-dioxaphospholane enables precise tracking of chemical modifications. Once researchers know which hydroxyl groups react and which don’t, they can optimize treatments or even cut out solvents and harsh conditions.
Of course, this approach only works with the right combination of practical skill and a reliable supply chain for specialty reagents. I’ve seen labs get stuck waiting for custom synthesis or swapping to less selective chemicals because supplies run short. It shows how interconnected everything is—new sustainable materials only succeed when tools like this are available and reliably produced.
Taming complex materials like lignocellulose counts on careful analysis, and that often means handling chemicals with hazards. Dioxaphospholanes like this need respect in terms of storage and safety, but their benefits far outweigh the hassle for labs prepared to use them wisely. Moving forward, improving access and working conditions can help more labs, especially in developing regions, benefit from this technology.
By leaning into thorough characterization and smart chemistry, future materials can edge closer toward sustainability. Every advance in reagents and analysis keeps the promise of greener processes within reach—and that all starts with substances like 2-Chloro-4,4,5,5-Tetramethyl-1,3,2-Dioxaphospholane enabling sharper insight where it matters most.
Chemical compounds don't all behave the same. Some are sensitive to light, some form clumps in the heat, and others break down if left where humidity gets high. In my own experience working with research teams, forgetting these details often leads to wasted time—and sometimes, ruined experiments. Even the smallest amount of moisture or change in temperature changes the outcome, leads to safety risks or can cost someone a lot of money.
Science can sound intimidating, but a lot comes down to simple steps and careful observation. Letting powders or liquids sit in the open air can cause contamination. Dust, mold spores, or even a splash of coffee can bring unwanted surprises. Storing compounds in sealed glass or specific plastic containers can do more than keep out dirt: it can save someone from explaining why half a lab’s monthly budget wound up as a useless mess.
At home, people treat strong-smelling cleaners or medicines with care—tucked away, out of sunlight, and away from curious hands. Labs and manufacturers take this further, with climate-controlled rooms, air filters, and regular checks on older batches. Skipping these basics doesn’t just risk spoilage; unpredictable changes in a compound can be dangerous, sometimes leading to fires or toxic leaks.
Some people think storing compounds just means a cool, dark place. That oversimplifies things. For example, something as common as hydrogen peroxide only stays reliable in the fridge. Leave it on a warm shelf, and it starts to gas off or become useless. Certain organic chemicals slowly turn brown under office lighting, making a case for amber-colored bottles or light-blocking cabinets.
Humidity also creates trouble. Desiccants, like little silica gel packs, buy some peace of mind for powders and salts that pick up water from the air. Over the years, I noticed powders that clump for no obvious reason often spent a day too long exposed during packaging or handling. Even a closed jar can’t help much if left in a damp, poorly ventilated closet.
Every time someone pulls a compound out of storage, that’s a chance for a mistake. Containers need labels showing dates and any warnings, not jotting them down to memory or on a random notepad. I learned the hard way early in my career that unlabeled jars meant not only wasted supplies but also a search for someone with a better memory—never a good way to build trust in a lab.
Many institutions now use inventory software to track what comes in and what goes out. If a staff member forgets to check this, expired or unstable compounds start to pile up. Timely audits catch problems before they disrupt workflows or, worse, cause incidents. Automation helps, but people still need to pay attention. Even in a high-tech facility, ignoring a cracked seal or swollen lid can spell trouble.
Simple habits—keeping things labeled, dry, and away from extra heat or light—go a long way. For bigger operations, investing in climate-controlled storage, staff training, and automated tracking can pay for itself by reducing waste and preventing safety issues. Across the board, storing compounds safely is less about fancy systems and more about respecting the real risks of cutting corners.
Pulling a reagent bottle off the shelf in a lab means trusting the label, trusting your memory from MSDS training, and trusting you haven’t forgotten what that long IUPAC name really represents. Seeing something like 2-Chloro-4,4,5,5-Tetramethyl-1,3,2-Dioxaphospholane immediately rings alarm bells for anyone who’s spent time in synthetic chemistry. Complex organophosphorus compounds never tend to be benign, and adding chlorine into the mix usually means trouble if it’s mishandled.
My time in research labs showed me people can get complacent around chemical hazards simply because nothing has gone wrong—yet. That’s part of why some overlook the dangers posed by tricky compounds like this dioxaphospholane derivative. Most rely on gloves, a fume hood, eye protection, and learned routines. That doesn’t erase the reality: chemicals with phosphorus-chlorine bonds react with water, humidity or skin to form hydrolysis products. Some of those products bring their own hazards, including corrosiveness and toxic vapors.
Scientists have documented, for similar reagents, how exposure can mean anything from skin burns to severe irritation of respiratory passages if the fumes build up outside the hood. If the slightest whiff emerges during transfer between bottles, you know to step back. I’ve watched solvents bubble up when traces of moisture meet these reagents—spattering solution everywhere, even eating holes in rubber gloves. Dry conditions, sealed containers, and a low threshold for what’s considered “enough ventilation” matter more with compounds like this.
Published material on 2-Chloro-4,4,5,5-Tetramethyl-1,3,2-Dioxaphospholane suggests a definite group of risks: corrosive nature, vapor pressure high enough to escape an open vessel, and the potential for generating noxious fumes when in contact with air or water. Organophosphorus compounds often pose threats to more than just your hands: reports over decades note toxic properties—especially after longer or repeated exposures. Both the National Institute for Occupational Safety and Health (NIOSH) and academic toxicologists stress extra protocols for even indirect contact.
Chemical burns heal, but lungs scar for life. Even a small spill, dealt with poorly, can affect people well outside the fume hood. Experience in crowded university labs taught me to read labels twice, slow down opening every single seal, and to never skip a respirator for these jobs. It’s tempting to skip full PPE—gloves alone don’t always block everything, and volatile acids formed during use can sneak around cheap goggles. Scientists have written about accidental exposure because someone underestimated the splash potential of concentrated phosphorus reagents.
Some workplaces take the hazards for granted, but training and preparedness make the real difference. Knowing end-to-end procedures, practicing a fast and thorough spill cleanup, and keeping current on the latest safety studies beats relying on luck. Old experiments using similar phospholane chemicals led some universities to keep calcium gluconate gel sitting within reach: a quick response tool if anyone gets their skin exposed. This sort of readiness converts lab theory into practical safety.
Workers should expect everyone handling this compound to go through proper chemical safety drills—mock spills, open container transfer training, and conversations about long-term exposure risks. Even in industry settings, stricter air quality monitoring and engineering controls can save doctors’ visits later. Chemical manufacturers have started pushing for more robust packaging, stricter labeling, and better shipment containment; still, the biggest difference comes from individual responsibility and constant culture of safety.
I’ve seen the outcome of skipped steps and rushed work: irritated skin, visits to the campus health office, notes to occupational safety. Companies and labs can do more to teach respect—never fear—for chemicals like 2-Chloro-4,4,5,5-Tetramethyl-1,3,2-Dioxaphospholane. Look beyond your own bench to see who else could be affected, and set the standard one safe bottle at a time.
Plenty of folks see chemical formulas as cryptic, but they pack a lot of power behind their strings of letters and digits. That little equation can influence how a product gets regulated, shipped, and even how safe it is to handle around the house or in a factory. If you work in science, healthcare, or manufacturing, knowing those details isn’t something to gloss over. Even parents glancing at ingredient lists might want insight into what goes into things sprayed in their garden or added to food.
Let’s talk about why formulas matter so much. Every compound gets its identity from how its atoms arrange themselves. Take table salt — two elements, sodium and chlorine, come together in a one-to-one ratio. That simple combo, NaCl, tells chemists exactly what’s inside and how it may react. If you swap in potassium, suddenly you’ve got a new story: different health effects, different behaviors in recipes or experiments. The tiny change in letters means dramatic change in reality.
The molecular weight packs almost as much punch as the formula. Some industries need to know molar mass because it changes everything from product labeling to trade regulations. Let’s say a manufacturing manager needs to blend raw materials. Precise weights stay crucial — anything off, and you could get a batch that either doesn’t work or even poses hazards on the line. Scientists don’t just guess; they use facts and clear data. History has taught enough harsh lessons on what happens without a clear grasp of the science.
Take acetaminophen, for example — that over-the-counter pain reliever found in homes all over. Its formula, C8H9NO2, signals specific chemical groups stuck together. The molecular weight, about 151.16 g/mol, matters for pharmacists weighing out dosages. In hospitals, exact amounts spell the difference between helpful and harmful. Those numbers go right into safety sheets and inform doctors and nurses in real time.
Sometimes, it’s tough to track down the exact chemical formula or weight, especially with trade secrets or proprietary blends. That’s where transparency should win out. Companies earn trust through clear labels and open communication about what’s inside their products. A little bit of sunlight here can help avoid another health scare. The public has a stake in that conversation.
That’s not just a matter for experts tucked in labs. Anyone who’s poured drain cleaner down a pipe or laundered clothes with new detergent finds themselves relying on that information — even if they don’t read the fine print. Better science education, more plain-English communication, and reminders that facts save trouble down the road, all help people make safer, more confident choices.
If someone requests the formula and molecular weight for a product, it’s not about being nosy or a stickler for details. They could be managing allergies, complying with environmental rules, or just watching out for kids and pets. Questions like these power progress, healthy households, and honest markets. Nobody benefits when information gets locked behind technical jargon or hidden by vague labels. The more those chemical facts get shared, the more control people have over their health, businesses, and environment.
Nobody wakes up ready for a spill on their watch. Still, it happens everywhere—from the bleach bottle at home to hazardous materials at a busy warehouse. These moments test how people respond, not just to the mess in front of them but to risks that linger far beyond a slippery floor. Years ago, I watched a shop floor grind to a halt because someone knocked over a drum of solvent and nobody wanted to admit they’d never read the safety data sheet. It could have been much worse. The real test isn’t in avoiding spills, it’s in how folks choose to deal with them.
The most important move doesn’t require special training: know what you’re working with. If a jug of vinegar crashes to the kitchen floor, grab paper towels and baking soda. If an acid container bursts in a lab, running for tap water and hoping for the best ends up risking burns, toxic fumes, and damage that can stretch far beyond a single shift. The facts say chemical incidents send thousands to the emergency room every year, costing millions and risking lifelong harm. Good management starts before the spill—by reading labels and knowing storage rules.
You can spot organizations that value safety long before the next incident. They invest real time in hands-on training, not just check-the-box videos. Employees practice small-scale clean-ups, use absorbent pads, keep spill kits in plain sight, and drill on what counts as ‘just mop it up’ versus ‘call for help right now.’ No one gets left guessing. For families and small businesses, this same logic applies: if it stings the nose, comes with special warnings, or could stain forever, keep a neutralizer and gloves handy. Don’t cut corners.
Too often, clean-up jobs get handed down the line, with newer or younger folks assuming the risk. I’ve seen pride get in the way—someone rushing in bare-handed to prove they can handle it, only to wind up at urgent care. Safety is a group project, and everybody wins when pride takes a back seat. Leaders gain trust by stepping up, not just in emergencies, but by making sure no one feels pressure to handle unknown substances alone.
Regulations only work if people believe in them. OSHA, EPA, and local health departments lay out strict guidelines for spill scenarios, setting penalties for violations and requiring logs of any exposure. Real improvement comes when businesses use these as a floor, not a ceiling. The right solution combines clear instructions, supplies ready at hand, and a culture that rewards speaking up. Having a documented plan protects more than legal interests—it keeps morale high and accidents rare.
It’s tempting to treat spills as isolated slip-ups, but experience shapes better habits. Every spill is a signal to upgrade training, replace missing gear, or rethink storage spots. Encourage reporting without shaming, keep instructions in plain English, and focus on health over speed. Spills will never become relics of the past, but smart preparation and a little humility keep the worst outcomes out of the headlines.