Folks in the chemistry community have been paying attention to halogenated phenols for a good while. Once labs got the hang of introducing fluorine into organic compounds in the early 1900s, fluorophenols like 2,6-difluorophenol started showing up in the literature. Synthetic routes improved year over year, helped along by World War II and the pharmaceutical surge that followed. Interest grew as people saw that swapping hydrogen for fluorine at precise positions on a benzene ring could change not only the physical properties, but also how these molecules behaved in everything from solvents to drugs. For most of the last seventy years, this compound spent its time as a specialty chemical in research circles, rather than on the factory floor.
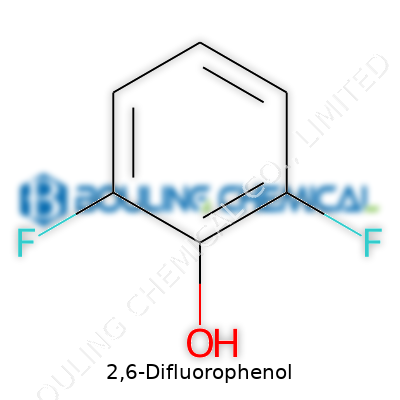
2,6-Difluorophenol stands out among halogenated phenols for its unique ring structure, which sports two fluorine atoms tucked onto the benzene ring at the 2 and 6 positions. That arrangement changes reactivity and gives it fresh life in research circles. Whether talking pharmaceuticals, advanced agricultural research, or the development of innovative organic materials, this compound has made itself useful in laboratories and development projects that care about how molecules perform under pressure or in new applications. Its key appeal: changing how the parent phenol interacts with other chemicals and living systems, thanks to the added fluorine atoms, each one a game-changer in the molecular world.
Compared to its simpler cousin phenol, adding two electronegative fluorine atoms chills things out. Volatility rises, making it easier to distill for purification. Its boiling point hovers lower than standard phenol. The substance looks like a colorless to faintly yellow liquid; sometimes, it shows up as a low-melting solid, especially if cold. The presence of two fluorines means lower basicity at the hydroxyl group; that affects how it dissolves in water or latches onto bases and metals. It doesn’t give off the familiar “hospital” scent regular phenol does. The real magic, though, is in how those fluorine atoms make the molecule less reactive to some unwanted side reactions, giving it a toughness and selectivity researchers often look for.
Ask any chemist about purity in research and the answer is: it matters. This compound almost always shows up at high purity grades, with suppliers screening for contaminants and closely managed water content. In my lab days, detailed labeling for organics included exact purity levels, batch data, and known trace impurities. Hazards are flagged plainly, as the phenol backbone calls for respect and due caution. Most containers include hazard pictograms, signal words, and handling instructions. Safety and proper labeling reflect a broader industry trend toward trust and transparency, a necessity with these kinds of active chemical agents.
By now, there’s more than one trick to making 2,6-difluorophenol, but most routes go through either direct fluorination of phenol or multi-step syntheses using protected intermediates. Electrophilic aromatic substitution plays a central role: starting with phenol itself or pre-fluorinated benzene and then guiding the fluorine atoms into place. Some routes make use of 2,6-difluoronitrobenzene, following reduction and hydrolysis steps until the phenolic group stands ready. Each method lines up pros and cons: safety, cost, scalability, and waste. Experienced chemists weigh these options, picking the safest route for their work’s scale and goals.
Adding fluorine atoms isn’t just a cosmetic change. They make the phenolic ring tougher against electrophilic attack, slowing down reactions that would race ahead on plain phenol. Still, the hydroxyl group remains reactive, letting you make ethers, esters, or dive into coupling reactions and polymerizations. The ring’s electron density drops, tuning down the kinds of chemical dances that happen in the lab. Medicinal chemists love these traits: they can slot this molecule into synthesis chains, building new structures with fine-tuned behaviors from the ground up. Sometimes, even tiny tweaks to these aromatic rings can mean the difference between a dud and a hit in new drug candidates.
You’ll find it listed as 2,6-difluorophenol, 2,6-DFP, or sometimes o,o-difluorophenol, though purists tend toward the standard IUPAC name for clarity. Keeping track of synonyms matters most for researchers digging through decades of technical papers or when shopping across suppliers who may use older trade names. Every label and technical bulletin in my experience tried to list the most common alternative names, keeping mix-ups at bay when you’re juggling dozens of similar molecules.
Working with 2,6-difluorophenol means treating it with the same respect as other low-molecular-weight phenols. Phenolic compounds can irritate skin and eyes, and inhaling vapors brings risks for respiratory systems. Gloves and eye protection form the backbone of PPE in any competent lab. Fume hoods pull their weight, keeping exposure to a minimum and avoiding unnecessary risk to labmates. Proper storage in cool, dry, shaded areas keeps it from decomposing or leaking odors. Safety data sheets help reinforce the right habits, keeping fellow researchers apprised of where dangers lie and how to respond if things get out of hand. Over time, regulatory frameworks like REACH in Europe or TSCA in the US shape how labs approach storage, labeling, and emergency planning. That structure builds a culture of shared responsibility and steady vigilance.
This molecule finds a home most often in the hands of innovators, whether developing new pharmaceuticals or probing materials science’s frontiers. Its modified ring opens doors for structure-activity relationship studies in drug discovery. Agricultural chemistry leans in as well, looking to move crop protection tools beyond legacy compounds, aiming for better performance and reduced toxicity. 2,6-difluorophenol can serve as a precursor for more complex building blocks, slotting into advanced organic syntheses. Academic labs use it to test computational models of aromaticity and reactivity, grounding theory in hands-on, bench-top results. It’s less suited for bulk commodity purposes; most of the impact comes through specialized, high-added-value R&D channels.
Research into phenolic compounds never lets up, thanks in part to how much is still unknown about their biological and physical roles. The unique set of fluorine atoms on this molecule draws keen interest from enzyme engineers, who use it to study metabolic pathways and trace how organisms process halogenated aromatics. Structure-based drug design projects revisit 2,6-difluorophenol when fine-tuning candidate molecules for stability, selectivity, or resistance to biological degradation. Analytical chemists lean on its distinctive spectra for calibration or novel detection methods. In recent years, a wave of papers explores greener, safer, and more scalable ways to synthesize this compound, reflecting broader pushes for cleaner chemistry throughout the field.
Concern for health and environment means no compound gets a free pass, no matter how useful. Toxicity testing on 2,6-difluorophenol flags the classic risks found in low-molecular-weight phenols, from skin and respiratory irritation to more serious nervous system impacts at high doses. Animal studies dig into how fluorinated rings affect metabolism and persistence compared to non-fluorinated relatives. While full environmental breakdown studies aren’t always available for every fluorinated compound, those that exist suggest these molecules resist biodegradation, raising flags for researchers looking to balance performance and ecological footprint. Most safety guidelines push for tight containment, low exposures, and robust emergency protocols, giving little room for complacency.
The outlook for 2,6-difluorophenol ties directly to trends in drug discovery, materials science, and sustainable chemistry. As fluorination strategies become more precise and cost-effective, customization gets easier, letting scientists tailor molecules for exactly the properties they want. New tools, like machine learning-guided reaction prediction, promise to reveal fresh applications and safer, greener preparation techniques. Increasing attention toward environmental persistence nudges researchers to reconsider both the use and fate of these fluorinated compounds. Demand may climb, especially as industries drive for new performance benchmarks that standard phenolics can’t reach. The challenge is the same as it’s always been: extract value, minimize risk, and answer society’s evolving needs with ever-better science.
As someone who has spent years following the ways in which chemistry helps shape the world around us, I see 2,6-Difluorophenol as one of those under-the-radar compounds with surprising reach. It’s not flashy, and you won’t spot it headlining news about green tech or medicine, but it quietly powers important work in labs and on production lines. Fluorinated phenols, especially this particular variant, have a way of showing up in places that matter.
The story often starts with medicine. Chemists lean on 2,6-Difluorophenol for building drug molecules that fight diseases or bring down inflammation. The ability to tweak a molecule using fluorine atoms changes how it behaves in the body. Here, two fluorine atoms set at specific points make the base molecule more resistant to breakdown and can help it slip past biological barriers. Drug designers constantly look for this kind of stability because it leads to better results for patients—and sometimes fewer pills. That’s real-life impact.
Published research backs this up. Studies from pharmaceutical journals show 2,6-Difluorophenol as a handy piece of the puzzle for non-steroidal anti-inflammatory drugs and central nervous system therapies. By plugging this building block into a synthesis pathway, researchers can spin out a new class of compounds to test against tough health problems.
Move outside the pharmacy and you’ll see this compound cropping up in crop protection work. Pesticide designers take advantage of phenols like 2,6-Difluorophenol to create molecules that can take on weeds or harmful insects, but not stick around forever and mess with the water. By weaving fluorine in this way, the final pesticide can deliver its job with more precision and fewer side effects for the environment. This careful chemical engineering helps keep harvests steady in a world that’s always bouncing between drought and flood.
Materials chemistry leans on 2,6-Difluorophenol for similar reasons. It serves as a starter for specialty polymers and resins that show extra durability or chemical resistance. In my years talking with researchers in this space, what comes up again and again is the demand for plastics and coatings that don’t break down under pressure or in harsh industrial cleaners. Swapping out a regular phenol for one with fluorine arms gives manufacturers just that edge for semiconductors, battery housings, or medical devices. You’ll find detailed references in scientific reviews covering how small tweaks in building blocks can lead to a big boost in performance down the road.
Alongside progress, concerns about environmental persistence and safety grow louder. Producing fluorinated compounds takes special care to avoid byproducts that linger in water and soil. I’ve talked with researchers who stress the need for closed-loop manufacturing and careful monitoring at every step. Solutions range from developing eco-friendly synthesis methods to recycling fluorinated intermediates. Many companies now invest in greener processes because regulatory pressure builds, but also because the science community—and everyday people—demand it. This is where transparent reporting and commitment to responsible chemistry make the most difference.
2,6-Difluorophenol probably won’t grab headlines, but it’s a tool that helps drive innovation across industries. The future steers toward safer practices and smarter applications. The next great treatment, the next food security breakthrough, or the next resilient material could have started with a smart piece of chemical engineering featuring this less-noticed hero. As long as there’s creativity in the lab and care in the factory, its story remains worth following.
Every chemical, whether used in a university lab or a manufacturing plant, needs a way to stand out. The CAS number for 2,6-Difluorophenol is 367-30-6. That number is more than just a label—it actually helps avoid confusion, even when names start to overlap and chemicals get mixed up. Folks who’ve worked with chemistry databases or tried to order materials for experiments can tell you, those digits can save a lot of time and even a few headaches.
Mix-ups in chemical identification aren't just a mild inconvenience. They can cause delays, create safety issues, and waste money. Back when I worked as a research assistant, I ran into trouble because the bottle on our shelf wore the wrong label. Only after cross-checking the CAS number did we realize the problem. The importance of getting digits right becomes clear fast in situations like that—especially since 2,6-Difluorophenol can be confused with similar compounds by name alone.
For manufacturers and end-users, the CAS number makes procurement a whole lot simpler. No one has time to verify synonyms or look up IUPAC names every single time there’s a need to reorder. Looking for 2,6-Difluorophenol? Plugging in 367-30-6 into a database or catalog narrows the options straight away. That accuracy matters more as regulations tighten and audits get tougher. A mistake at this step could mean the wrong substance in a product, fines, or recalls.
Global rules about shipping, handling, and storing chemicals rest on certainty. The CAS number serves as a common language. Agencies—from the EPA to the European Chemicals Agency—rely on this identifier when writing lists and creating tracking systems. International trade also depends on it, since different regions use a host of trade names or translations. If a customs official sees 367-30-6, there isn’t much room for ambiguity.
Even for emergency responders, knowing the precise CAS number makes it possible to track down safety profiles, find the right protective gear, and use proper neutralization agents. Safety Data Sheets all highlight this number for a reason. Lives, property, and the environment can depend on decisions made in a hurry, and that’s no time to flip through a dictionary.
Despite the clarity a CAS number brings, some users still struggle with outdated supplier catalogs or missing labels. This problem can slow down both lab and industrial work. A solution that’s picking up steam involves QR-coded labeling linked to real-time CAS number databases. I’ve seen labs that update their inventory with smartphones and a quick scan, which reduces mistakes and speeds up audits.
Everyone who handles chemicals benefits from cross-checking CAS numbers before purchasing, mixing, or disposal. Regulators could do more to push for digital systems requiring CAS input, not just names. Professional societies and trade associations have a role too, by offering resources to help people double-check what they’re actually using.
CAS number 367-30-6 doesn’t just identify 2,6-Difluorophenol; it adds certainty in a field that’s not always forgiving of mistakes. Whether you’re in the lab, on the shop floor, or running compliance for a chemical business, that identifier reduces risk and saves resources.
In any lab or industrial setting, small lapses with chemical storage can spell trouble. 2,6-Difluorophenol seems simple enough—just another halogenated phenol, clear and colorless, with a sharp odor. Yet, this chemical won’t forgive the kind of corner-cutting some folks try to justify. I’ve watched coworkers overlook basic guidelines, figuring “it’ll be fine for a few days.” That’s the wrong mindset.
A bottle of 2,6-Difluorophenol is more than just liquid in a jar. It’s corrosive—eats away at skin, eyes, and respiratory tissue in a way that stings, then lingers. If left open or kept in the wrong container, it emits vapors no lab bench needs to face. And moisture? It loves to sneak in, potentially triggering hydrolysis or wrecking sample reliability. From my own experience, nothing derails a project faster than a tainted reagent. Try explaining to your supervisor why the whole series of syntheses now sits in the waste drum.
One of the best lessons I ever learned was from a mentor who believed in clarity and repetition over fancy talk. He’d point out that for chemicals like 2,6-Difluorophenol, nothing beats a cool, dry, well-ventilated spot—away from sunlight, heat, and moisture. Even slight warmth tends to nudge volatility upwards, sending fumes across the workspace. Stashing it alongside incompatible reagents—like oxidizers or bases—can lead to fire or noxious byproducts. Locked cabinets, clear hazard labeling, and glass or fluorinated plastic containers go a long way toward keeping both people and products safe.
There’s a reason safety data sheets stress proper storage specifics. Compliance isn’t about bureaucracy for its own sake; it actually keeps people from harm. Some will roll their eyes, but workplace injuries and near-misses carry real costs. I recall a fire department inspection where a misplaced bottle had the potential to react with a leaky base. That could have triggered an evacuation and caused lasting damage. Local and international rules spell out container types, stacking height, secondary containment, and signage. Flouting these isn’t clever—it’s reckless.
Better practice begins with culture, not just checklists. Training should focus on the “why,” building respect for rules, not just reciting them. I’ve watched teams improve safety simply by double-checking inventory at the end of each shift and reviewing humidity or temperature readings as a habit, not a last-minute scramble. Technology helps, too—digital logging, real-time environmental monitoring, and alarms when things drift out of spec.
In my own work, I make sure every chemical—especially problem children like 2,6-Difluorophenol—has its own file, location code, and periodic review. If something leaks, evaporates too quickly, or seems undermined by the wrong storage, it gets flagged and replaced, no excuses. Ideas like staff empowerment, leadership by example, and crystal-clear reporting channels keep errors from snowballing.
A single incident can set back months of research or production, shatter trust, and bring painful cleanups—not to mention the personal cost of injured staff. Taking storage seriously—proper temperature, sealed containers, out of direct sun, and away from incompatible materials—shows respect for science and for the real people who walk through those lab doors each day.
Years spent discussing chemical safety with workers in research and manufacturing show one thing again and again: asking whether a chemical like 2,6-difluorophenol poses a hazard or is toxic isn't just a technical question. It’s one that calls for plain talk, honesty, and real care for health and safety. Many folks see a scientific name and skip to the next headline. Big mistake. Just because a chemical isn’t famous doesn’t mean it doesn’t matter.
Let’s Cut Through the Noise
2,6-Difluorophenol doesn’t draw crowds or headlines like some industrial chemicals. Yet, it's a building block in organic synthesis and turns up in pharmaceutical research. That means actual people use it—chemists, engineers, even students in a lab somewhere right now. For everyone handling it, the real-life risks matter.
Similar compounds in the phenol family don’t have a reputation for being gentle. They tend to irritate skin and eyes, sometimes causing burns. Inhalation brings its own set of problems: headaches, coughing, and throat discomfort. A few years ago, I managed cases where phenols caused occupational asthma in under-ventilated workshops. If 2,6-difluorophenol shares those traits—and given its chemical cousins, it probably does—hazard awareness isn’t optional.
Fluorine throws another wrench in the gears. Fluorinated compounds can be more reactive, toxic, or persistent in the environment compared to their non-fluorinated counterparts. While 2,6-difluorophenol isn’t a headline-grabbing supertoxin, it deserves respect. Acute exposures might cause immediate harm—red skin, watery eyes, coughing fits. Chronic, low-level exposures become harder to spot, but they still leave their mark. Too many workers ignore glove rules and rely on faulty fume hoods, thinking the risk is too small to notice until it's too late.
Find any peer-reviewed studies or regulatory documents on 2,6-difluorophenol, and the word “caution” comes up more than once. The US Environmental Protection Agency flags fluorinated phenols for further review, but hard data on this particular variant stays pretty sparse. That’s often the case with specialty chemicals—enough data to make you pause, not enough to fill in all the blanks.
Poison control records list phenolic compounds as common causes of accidental poisoning in laboratory and industrial settings. It doesn’t take a spill the size of a swimming pool—just a splash or a breath. Many phenols disrupt cell membranes and mess with proteins, which means even low doses can hurt over time. The liver and kidneys usually bear the brunt, and symptoms can linger for days.
Thinking through safety isn’t just about ticking off boxes or copying protocols from a binder. It’s about culture in every lab, every warehouse, every classroom where 2,6-difluorophenol plays a role. PPE—proper gloves, goggles, and sharp ventilation—makes a difference. The best chemical handling I’ve seen came from people who treated every unfamiliar compound with healthy skepticism and curiosity, not casual confidence.
Chemical manufacturers and managers must support access to fresh safety data and real training. Review those safety sheets and stop for questions. Ask for third-party reviews, especially in operations that use large quantities or high concentrations. If there’s uncertainty, sub in a safer alternative and reduce user exposure as much as possible. Insist on routine air monitoring and health checkups for repeat handlers—those are life-savers, not just paperwork.
The Chemical May Not Scare Headlines, But It Deserves Our Attention
2,6-Difluorophenol doesn’t have to be a household name to matter. For anyone using it, the stakes are real. Chemicals don’t forgive shortcuts or carelessness—history proves that. So, before reaching for that next bottle, ask the real questions about what’s in your hands, who’s around you, and whether you’ve done enough to work safely. Act now, stay safer later.
In the specialty chemical world, details make all the difference. Take 2,6-Difluorophenol as an example. Purity isn’t just a number in a lab report. For every chemist or researcher relying on this compound, purity shapes results, impacts downstream processes, and often determines the very safety of their team.
2,6-Difluorophenol usually comes with purity quoted at 98% or above. That extra couple of points can cost, but it guarantees fewer headaches during synthesis. Laboratories involved in pharmaceuticals or fine chemicals expect a certain level of quality. If those trace impurities sit above the advertised spec, whole batches risk contamination. That turns into lost time, wasted resources, and sometimes, endangered health.
Longtime users spot inconsistencies before a chromatograph even prints the numbers. Fluctuations from lot to lot give away shortcuts in quality control. The world has seen more pressure on supply chains over the past years, and with that comes a temptation to loosen standards. All too often, it isn’t until a reaction stalls, or an unexpected side product appears, that someone traces the problem back to a missed impurity, a solvent residue, or a forgotten stabilizer.
Many researchers draw on years of trial and error where minor slip-ups in chemical purity become apparent in the results. A late-stage pharmaceutical process can get trashed over a contaminant at 0.5%. Some customers insist on lot-specific certificates of analysis, and they want HPLC traces and even NMR data to reassure them there’s nothing unwanted lurking in their bottle. Analytical transparency earns loyalty far beyond the legal minimums. Nobody wants to repeat a month of benchwork for lack of a printout showing that their 2,6-Difluorophenol meets the bar.
Academic labs and startups often operate on shoestring budgets, so they can’t afford to buy twice. The more robust the purity assurance, the smaller the risk of failed experiments or reproducibility issues. These teams aren’t just interested in numbers; they want proof, context, and even background about the supplier’s track record. The pandemic showed that supply chains break down fast, so buyers turn to reliable documentation as a hedge against the unknown.
It always pays to look beneath the catalog listing. Buyers call for batch-specific reports, a direct line of communication with quality control, and even tours of some supplier facilities. Users should demand access to up-to-date analytical information and inspect storage and transportation protocols that might let impurities find their way in after the product leaves the factory. Some groups even run quality checks themselves, especially when scaling up work or delivering to regulatory authorities.
Feedback loops, with direct communication between supplier and researcher, let subtle problems get fixed fast. Industry groups are starting peer-review and reference sample programs, making it easier to spot the real outliers—good or bad. No certificate can offer real confidence without a system that lets people speak up and fix trouble fast. The result is fewer surprises and greater safety in the lab.
The real worth of 2,6-Difluorophenol rests on clear, unflinching analysis. Trust, not just purity, keeps labs running and results reproducible. That’s something every chemical supplier and buyer can agree on.