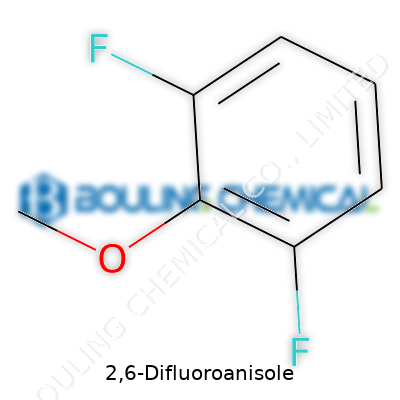
The roots of 2,6-Difluoroanisole go back to a time when the field of fluorinated aromatics was just beginning to expand. Chemists in the mid-20th century started to realize that adding fluorine atoms to aromatic molecules like anisole could unlock a host of new behaviors—both in the lab and in industrial practice. Even now, labs are still refining the processes for making and using this compound, learning from the trial-and-error spirit that defined much of early medicinal and polymer chemistry. Looking at scientific literature, it’s clear that fluorinated anisoles didn’t enter the scene on the back of one breakthrough. Their rise came from steady progress—improved synthetic routes, better understanding of reactivity, and recognition of valuable uses, especially in pharmaceuticals and materials science.
2,6-Difluoroanisole stands out among its peers because of its dual nature: the anisole core offers reactivity that appeals to organic chemists, while the fluorine atoms at distinct positions influence both physical and electronic properties. Unlike its parent compound, anisole, this difluorinated version lends itself to modifications that open doors in agrochemicals and high-tech manufacturing. To some, it may seem like just another laboratory reagent; to others, it's a building block that helps shape molecules central to disease research or next-generation materials. I’ve seen firsthand how a subtle change like swapping hydrogen for fluorine can lead to unexpected benefits—longer shelf life of pharmaceuticals, heightened resistance to degradation, or simply unique ways for molecules to interact with living systems.
Fluorine’s reputation for stubbornness comes through in 2,6-Difluoroanisole. The compound typically appears as a colorless liquid and gives off the faintly sweet, medicinal aroma one expects from many small fluorinated aromatics. Its melting and boiling points differ from non-fluorinated anisoles, marking out its chemical personality. Fluorination changes not just the molecule’s resistance to breakdown, but also its solubility and volatility. At the bench, handling this solvent-like liquid calls for gloves and care, but it mixes well with organic solvents and stands up to conditions that degrade simpler aromatics. The electron-withdrawing nature of the fluorine atoms also affects chemical reactivity, steering which types of reactions work best.
In the world of fine chemicals, clear labeling is a must—purity levels, batch details, and hazard tags all show up on containers of 2,6-Difluoroanisole. Labs and factories rely on these details to keep research consistent and safe. A purity over 98 percent usually satisfies research needs, while industrial users sometimes request tighter specs to avoid contamination in downstream synthesis. Shipping this compound demands compliance with both local and international rules. Labels and safety sheets aren’t optional—they form a key step in responsible chemical handling.
Researchers have developed several routes to prepare 2,6-Difluoroanisole, most drawing from halogen exchange or direct fluorination. Older methods exposed precursors to hazardous reagents, but newer techniques make use of milder conditions and purer starting materials. What matters most for chemists is often yield and the presence of side products; extra fluorination can cause unwanted isomers or overreaction, so recipe tweaks based on lab experience can make the difference between success and failure. It’s a process that combines deep knowledge of aromatic chemistry with creative problem-solving, especially when scaling up for production.
The two fluorine atoms in 2,6-Difluoroanisole set the stage for a range of downstream chemistry. They pull electron density from the aromatic ring, changing how the molecule interacts with electrophiles and nucleophiles. As a result, typical reactions like Friedel-Crafts acylation shift in rate and selectivity on this scaffold. I’ve worked with chemists who prize this compound for the way its electron-poor ring behaves—it allows access to patterns of substitution or functionalization that are hard to achieve with unsubstituted anisole. Many drug researchers start with this molecule when they want to introduce fluorine into active pharmaceutical ingredients, hoping to improve how long a medicine stays effective in the body or reduce breakdown by enzymes.
On a shelf or in a catalog, 2,6-Difluoroanisole rarely wears just one name. Chemists may call it 2,6-difluoromethoxybenzene, o,o'-difluoroanisole, or refer to it by a system designation used in a research project. Sometimes the synonyms matter—searching by a specific name can make the difference between finding a crucial safety study or missing it entirely. This tangle of names traces back to overlapping naming conventions in organic chemistry, but over time, databases and digital libraries have made it easier to track compounds, whatever label they wear.
Every compound with active fluorine deserves respect. Though 2,6-Difluoroanisole doesn’t announce its hazards with obvious fumes or dyes, its toxicity and flammability mean operators must follow clear protocols. Personal protective equipment stands as the frontline defense—nitrile gloves, splash goggles, and fume hoods keep accidental exposure at bay. Facilities run regular safety training, review handling procedures, and update storage conditions based on the latest risk assessments. Companies push for compliance with local labor and chemical regulations not only to avoid fines but because no research or manufacturing schedule is worth a health emergency. Laboratories keep spill kits on hand, and staff measure out quantities with precision to avoid incident and waste.
The uses of 2,6-Difluoroanisole read like a guide to modern applied chemistry. Medicinal chemists use this compound as a springboard for molecules that resist metabolic breakdown and show new biological effects. Material scientists build polymer components out of this core—putting its resistance to weathering and heat to good use in coatings and specialty plastics. Agrochemical researchers use its structure to design crop protection agents that break down at a controlled pace, aiming for improved safety for both plants and people. For those who build sensors and electronic materials, fluorinated aromatics offer durability and tailored electronic features. Each time research pushes into a new field—drug discovery, electronics, agriculture—2,6-Difluoroanisole finds fresh demand.
Academic and commercial R&D teams view 2,6-Difluoroanisole as a versatile probe for both fundamental and applied investigation. Scientists investigate its reactivity to develop more sustainable synthetic routes or cleaner catalytic processes. Organic chemists map how electron-withdrawing groups like fluorine alter reaction profiles, aiming for new classes of medicines or materials. Partnerships between universities and chemical companies, supported by grant programs, fuel these efforts. My own observations show how collaboration between synthetic and analytical chemists can speed up progress: one team lays out a synthetic challenge; another develops sensitive assays to detect trace impurities or environmental byproducts. Over time, as more is learned about this molecule’s quirks, it can take on even bigger roles in both lab-scale and industrial contexts.
Studies into the health and environmental impact of 2,6-Difluoroanisole carry weight, especially as regulators pay greater attention to persistent organofluorine compounds. Research teams run toxicity assays on both cell cultures and mammals, checking for acute effects and long-term risks. Data from related compounds show how the position and number of fluorines influence toxicity and persistence. Scientists look for potential endocrine disruption or bioaccumulation, always alert to signals that suggest either added danger or, conversely, a margin of safety for wider commercial use. These studies steer guidelines for allowable exposure, disposal standards, and even future synthesis routes that limit hazardous byproducts. Ongoing review and transparency remain central, as industries and the public both expect chemicals to carry a traceable safety profile from lab to market.
2,6-Difluoroanisole faces a future shaped by both opportunity and responsibility. On the one hand, demand will move with trends in medicine, advanced materials, and chemical manufacturing. As scientists ask tougher questions about environmental stewardship and regulatory compliance, the field adapts, looking for greener syntheses and tighter process controls. The rise of sustainable chemistry isn’t just a slogan—researchers design new versions of old reactions, striving to lower energy use and reduce hazardous waste at every step. Meanwhile, curiosity fuels new application areas: biologists and engineers turn to molecules like 2,6-Difluoroanisole for next-generation solutions, from more stable drug scaffolds to rugged materials for space exploration. As research uncovers new risks or avenues for improvement, adaptability and open information-sharing will keep 2,6-Difluoroanisole relevant and responsibly managed in the years ahead.
Chemistry weaves into so many aspects of daily life, yet most of us don't realize how often it shapes the things we encounter. The chemical formula of 2,6-Difluoroanisole is C7H6F2O. At first glance, it’s just a cluster of letters and numbers, but behind those symbols lies a story that touches everything from pharmaceuticals to pollution control.
In research labs, accuracy in chemical formulas sets the foundation for every project. Take C7H6F2O: this formula tells chemists exactly what goes into the molecule. One mistake in the formula, and the end compound could serve a totally different purpose, or worse, give off unexpected reactions. Reliable data about chemicals supports everything from safe handling to effective application in scientific discoveries.
This matters not just to people in white coats but to anyone who depends on modern medicine, clean water, or even safe household products. Researchers use 2,6-Difluoroanisole as a building block in synthesizing advanced materials and pharmaceutical intermediates. A clear understanding of its molecular structure helps make those discoveries reproducible and trustworthy.
Fluorinated compounds have become central in creating targeted therapies, pesticides, and specialty polymers. The two fluorine atoms on the anisole ring don't just tweak the molecule—they push it into new realms of usefulness. For instance, the presence of fluorine atoms can improve a compound’s metabolic stability and bioavailability, which have direct impacts on medicine development.
Environmental safety comes into play here, too. Fluorinated products show up in landfill and soil samples all over the world. Awareness of formulas and molecular structures like C7H6F2O fuels responsible innovation. As someone who talks with people across environmental research, I've noticed the same worry—poor monitoring and unregulated release of such compounds sometimes lead to persistence in the environment and long-term impacts on animals and humans.
The push for green chemistry and careful industrial design starts with knowing what each new synthetic compound brings to the table. Fact-driven approaches raise the credibility of research and protect public trust. In the chemistry community, mistakes have real consequences, whether from an error in documentation or a flawed structure. I’ve seen teams go back to fix product batches, rewrite safety procedures, or even shelve promising projects simply because a structural detail didn’t match up. It all comes back to knowing exactly what you’re working with.
Solutions grow out of layered knowledge and clear communication. Researchers can focus on full documentation, open sharing of chemical data, and broad collaboration across the academic and regulatory world. Any step that increases transparency—whether routine molecular checks or publishing open-access data—makes the process safer for everyone. For anyone not in the lab, supporting rigorous science means holding companies and public agencies to high standards for transparency and accountability in chemical use.
Understanding something as specific as the formula for 2,6-Difluoroanisole gets us closer to better, safer, and more responsible science. It’s a reminder that tiny details often carry the biggest weight.
Specialty chemicals drive more progress than most folks realize. Take 2,6-Difluoroanisole—at first glance, it doesn’t stir up much excitement, but this compound quietly shapes a broad range of products and research. I once walked through a fine chemicals facility and saw chemists almost babysitting reactors where this stuff goes into play—not just because it’s valuable, but because it serves as a clever starting point for other compounds.
Modern drug discovery leans heavily on reliable chemical building blocks, and 2,6-Difluoroanisole brings a unique edge. It acts as a key intermediate, especially in the hunt for new medicines. Chemists applaud its stable benzene ring, tweaked by both fluorine atoms and a methoxy group, which sneaks just the right balance into a target molecule. That chemical trickery matters—introducing fluorine can help a medicine last longer in the body, making it more effective and easier to dose for patients. You can trace the presence of this compound in a string of laboratory syntheses, especially for candidates targeting inflammation or cancer.
Crop protection needs bright chemistry. Out in research fields, safer pesticides and herbicides mean healthier yields without collateral damage. 2,6-Difluoroanisole plays a behind-the-scenes role here too. By acting as a precursor, it allows scientists to fine-tune products that handle pests efficiently, with fewer side effects for the environment. No one’s eager to dump old-school, broad-spectrum poisons when a precisely engineered molecule does the job better. My conversations with industry farmers and researchers show real hope in these “designer” agents, and many start with compounds like 2,6-Difluoroanisole.
Anyone tinkering with material science or performance chemicals may run into this compound. Specialty polymers, specialty coatings, and advanced dyes pull on the versatility of molecules with halogen atoms like fluorine. That trifecta—a benzene ring, two fluorines, and a methoxy group—unlocks properties you just can’t get from basic hydrocarbons. I remember one small lab that used 2,6-Difluoroanisole as a launchpad for tailoring UV-stable coatings for medical devices. With demand for medical plastic growing fast, these tweaks become a real competitive advantage.
Everything with industrial chemicals eventually circles back to safety and the planet. Experience has shown that while 2,6-Difluoroanisole holds value, it also demands careful handling. Its manufacturing and use need thoughtful control—limiting exposure and keeping by-products out of waterways. A push in recent years involves green chemistry, encouraging manufacturers to swap harsh reagents for milder ones or to use safer solvents. I’ve seen more production shops run their reactions at lower temperatures and pressures to minimize waste—a small thing, but the ripple effect for safety and sustainability matters.
Today’s progress with 2,6-Difluoroanisole says something simple: pay attention to the pieces that make up our medicines, chemicals, and even safer food. Stronger partnerships between chemists, environmental scientists, and industry leaders can turn promising molecules into real-world solutions, provided the details around handling and sustainability remain a priority. Chemical building blocks like this shape much more than just lab results; they touch lives in ways most of us never see.
Sometimes, folks look at chemicals and think about a world of tiny, invisible things that only researchers and white coats worry about. But knowing the molecular weight of a compound like 2,6-Difluoroanisole actually has a day-to-day impact across many fields—medicine, forensic science, and environmental testing to name a few. This number acts as a building block for how we dose medications, track pollution, or even detect illegal substances. It’s not just for chemists solving equations in a silent lab. It’s a piece of information that shapes the way products are made and regulations get written.
For 2,6-Difluoroanisole, the formula is C7H6F2O. Calculating molecular weight means adding up the atomic weights:
Add those up and you get 144.12 for carbon, 6.06 for hydrogen, 37.996 for fluorine, and 16.00 for oxygen. The total comes to about 204.18 g/mol.
In labs, it often comes down to weighing out the right mass for an experiment or a reaction. If someone is developing a medication or a pesticide, getting this number wrong throws off batches and creates safety risks. In my time working with graduate students, I’ve watched someone sweat it out, recalculating because they mixed up a few digits in the formula, setting research back by days. Every error ripples out into cost, safety, and reliability. That’s not something anyone claps about, but it keeps quality high and risk low.
The world cares more and more about trace chemicals in the air, water, and food. Regulatory agencies lean on molecular weight for dose calculations, safe exposure limits, and proper labeling. In my environmental testing work, hitting the mark with molecular weights helped my team connect the dots between what was in a sample and how much might actually matter for health. No one wants residue in their food or their kids’ playground, so this figure becomes a step in keeping things safe.
With the open access to databases online, students and professionals get the right data faster. Resources like the NIST Chemistry WebBook and PubChem help cross-check numbers in seconds. Before digital resources, getting molecular weights meant a trip to the library or the back of a textbook. Now, mistakes should drop. Investing in quality training and keeping up digital resources will go a long way toward helping newcomers stay accurate.
Teaching the “why” behind numbers makes the science less abstract. 2,6-Difluoroanisole’s weight might not show up in your dinner conversation, but it shapes products, rules, and studies in ways that touch everyone’s life. That little number keeps science moving forward, one equation at a time.
Give the name “2,6-Difluoroanisole” to most people and you’ll probably see blank stares. It sounds like something from a lab, and that’s because it is. Researchers and chemical manufacturers use this type of compound in plenty of science-based industries. You can spot derivatives of anisoles in pharmaceuticals, dyes, and even as intermediate steps in making other chemicals. Just because you’re not likely to run into it on grocery store shelves doesn’t mean its risks don’t ripple outward.
Whenever I dig into chemical safety—either for work or writing—my first step is to look for reliable studies. Peer-reviewed research, governmental toxicology reports, and established chemical safety databases offer the best guides. For 2,6-Difluoroanisole, there’s no mountain of public information, but a few things stand out. Fluorinated chemicals, as a group, don’t always behave the same way, but they can cause health effects depending on how much and how long someone gets exposed.
Take inhalation. In the short term, breathing in vapors of aromatic ethers can irritate nasal passages, throats, and lungs. I once worked in a lab where someone accidentally tipped a small vial of a related chemical, leading to a room full of coughs and quick evacuations. Gloves, goggles, and proper ventilation keep most technicians safe, but accidents happen. Skin contact leads to redness or dryness. In lab settings, these chemicals rarely end up running down your drain or seeping into neighborhoods. The risks jump if disposal steps get skipped or workers ignore safety basics.
Organic compounds with fluorine atoms draw concern, especially since some linger in soil and water. They don’t all stick around for decades like PFAS, but long-lasting pollution drives stricter scrutiny worldwide. People living near chemical plants often ask hard questions about toxic emissions. Studies suggest some anisole derivatives can harm aquatic life at certain concentrations, but specific research on 2,6-Difluoroanisole’s environmental fate is still growing.
When studies don’t give clear answers, it pays to look at similar compounds for clues. Regulators tend to act cautiously, even when direct human toxicity isn’t well-studied. If a chemical joins a production line without full public research, the companies handling it carry a heavy responsibility to prevent leaks, limit emissions, and share information with their communities. Transparency matters most for those living near factories or waste sites—and history shows what happens when it’s missing.
I’ve seen safety transform over the past two decades. What used to look normal in labs—open benches, wide-open fume hoods—has shifted to sealed systems, real-time sensors, and training refreshers at every level. The green chemistry movement marked a clear shift in thinking. Less hazardous reactions, less waste, more accountability. For specialists working with 2,6-Difluoroanisole, education stays front and center. Knowing the right gloves, masks, and handling steps means fewer surprises and healthier staff.
The bigger picture always comes back to informed decisions. Reliable sources, effective local oversight, accessible data—that’s what helps both workers and neighbors stay safe. Chemicals like 2,6-Difluoroanisole aren’t going away, but the way we handle them separates safe innovation from the next environmental headline.
People who work with specialty chemicals every day know the risks pile up, especially when overlooked details invite accidents. 2,6-Difluoroanisole isn’t a household name, but it shows up in labs, research spaces, and manufacturing settings tied to organic synthesis. Over the past years, I’ve seen situations where lax handling—even with chemicals less reactive than this one—has turned minor mishaps into hazards. These experiences keep reinforcing the same lesson: storing and handling compounds with clear respect matters more than the label on the bottle suggests.
2,6-Difluoroanisole flows as a colorless liquid, releasing a characteristic odor. This alone signals the need for a closed container—no one benefits from breathing what shouldn’t be in the air. Volatile organics do not play well in sunlight or with temperature swings. From my own experience in chemical storerooms, mixing incompatible substances can trigger unexpected reactions; sudden pressure build-up, leaks, or vapor release are all real possibilities. If there’s a fire hazard lurking, it often starts with storing flammable liquids near a heat source or direct sunlight. Simple practices, like shelving this compound well away from oxidizers, acids, or even loosely capped bottles, seem basic but protect labs from chemical chaos.
Flammable storage cabinets, ventilated and grounded, provide peace of mind. Labels matter, but vigilance matters more—regular checks for leaks, cracks, or corroded lids keep control where it belongs. Chemists and lab techs can spot trouble by relying on senses and training, and regular safety drills make correct responses automatic. After visiting labs where fumes went undetected for days, I learned just how easily an invisible risk grows if ignored.
Real-life lab spaces jostle with containers, glassware, and all sorts of distractions. Personal protective equipment—goggles, gloves, and lab coats—feels cumbersome at first. Over time, the habit sticks, especially once you catch a splash of solvent on bare skin or smell harsh vapors. In my earliest days at the bench, I watched an overconfident grad student tip a small flask, sending a few drops down the bench. They brushed it off, but the scent lingered, reminding everyone that lax handling costs more than a ruined shirt. Spraying down surfaces, ventilating the room, and double-checking every lid reinforced the lesson: careless spills poison air and ruin workdays.
Fume hoods aren’t just for highly toxic chemicals. Even moderate solvents like 2,6-Difluoroanisole vaporize at room temperature. Keeping work inside the hood makes mistakes containable and limits accidental exposure. Training drills on spill cleanup, eye-wash stations, and safe transfer techniques sound dry in theory but play out in real scenarios—seen it myself during late-night research runs.
Reliable chemical management systems build safety into the workspace. Barcode tracking weeds out expired or degraded bottles, cutting down on unexpected hazards. Routine education—like weekly safety briefings or refresher videos—does more than look good for compliance; it actually sinks in decades after leaving the classroom. Talking openly about near-misses or safety scares builds a culture where people watch each other’s backs, not just their own. Reporting a storage lapse or mishap doesn’t trigger blame, it prompts better support and systematic fixes.
It often takes a near-miss to shake people out of complacency, but with chemicals like 2,6-Difluoroanisole, waiting for a wake-up call courts unnecessary risk. Treating every bottle as a potential problem, not just a routine part of the job, keeps people and projects on track. My years in the lab taught me: smart preparation and defensive habits turn dangerous compounds into manageable tools, not ticking time bombs.