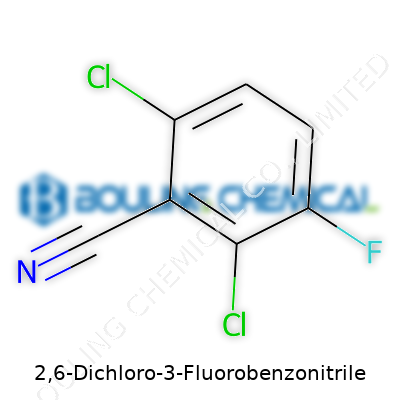
Back in the late twentieth century, chemists started focusing on new halogenated aromatic compounds, drawn in by the promise of improved reactivity and enhanced selectivity in technical applications. Among these chemicals, 2,6-Dichloro-3-Fluorobenzonitrile began gaining traction. Researchers, both academic and industrial, valued it for its potential as a versatile intermediate in the synthesis of complex molecules. As the pharmaceutical, agrochemical, and specialty chemical industries grew, so did the demand for more specialized aromatic nitriles. This compound’s unique structure, with two chlorine atoms and one fluorine flanking the benzonitrile core, set it apart as a building block that could insert reactivity just where chemists wanted it. Patents from the 1980s and 1990s started referencing this compound in formulations and synthesis routes for crop protection agents, bringing it slowly but surely into the mainstream.
What makes 2,6-Dichloro-3-Fluorobenzonitrile valuable extends beyond its three halogens or aromatic character; the chemical serves as a go-to intermediate for a handful of challenging syntheses. By nature, molecules with both electron-withdrawing nitrile groups and halogen substitutions often bring greater reactivity, making them well suited for further substitution or coupling reactions. For chemists accustomed to troubleshooting complex syntheses, having such a compound available means fewer steps to a finished product and greater control over the transformations along the way. The benefits show up not only behind laboratory benches but also in scaled-up production, where reproducibility and selectivity spell the difference between commercial success and expensive gridlock.
With its dense aromatic ring and multiple halogen atoms, 2,6-Dichloro-3-Fluorobenzonitrile stands out as a pale solid with a noticeable, but not overpowering, chemical odor. Anyone who has handled chloro-fluoro aromatics will recognize that faint scent, impossible to completely mask. The melting point tends to fall in the upper range for substituted benzonitriles, lending it a degree of thermal stability critical for downstream synthesis. Low water solubility, a direct result of the compound’s strong aromatic backbone and halogen substituents, often means less dispersal during processing—a mixed blessing for waste treatment but helpful during purification. Basic organic solvents like dichloromethane, acetonitrile, and even toluene can bring it into solution, easing operations in synthetic labs and chemical plants alike.
Accuracy in labeling matters. For a compound with a CAS number and established nomenclature, which includes synonyms such as "3-Fluoro-2,6-dichlorobenzonitrile," chemical purity takes center stage in every specification document or shipment certificate. Purity matters in research, since stray impurities—often carried through from halogenation or cyanation steps—can derail sensitive reactions in pharmaceuticals or crop protection products. Specifications typically lay out allowable limits for moisture, heavy metals, and residual solvents. I’ve noticed that some suppliers over-promise on crystalline appearance or solubility, but in the end, practical testing in the intended reaction trumps any glossy technical bulletin. Real-world results—how the compound behaves in the flask or reactor—shape its reputation far more than a sanitized certainty in documentation.
Synthesizing 2,6-Dichloro-3-Fluorobenzonitrile demands precision timing and a solid grasp of aromatic chemistry. Most production routes start from a suitably substituted benzonitrile or benzene ring, and stepwise halogenation using chlorine and fluorine sources under controlled conditions. The order of introduction—whether halogenation comes before or after cyanation—tends to influence yield and selectivity. In practice, metal-catalyzed cross-coupling methods, including Suzuki or Sandmeyer reactions, give access to specific substitution patterns. Laboratory experience shows that controlling temperature and stoichiometry allows for fewer by-products, higher yields, and easier workup. That small detail—temperature control—often separates a successful synthesis from a failed batch, particularly with reactive fluorinating agents that can trigger side reactions or decomposition.
What sets this molecule apart is its openness to further chemical modification. The two chlorines make attractive leaving groups for nucleophilic aromatic substitution, enabling derivatization by adding amines, thiols, or alkoxides, depending on the intended use. In pharmaceutical settings, I’ve seen it serve as a scaffold for making complex heterocyclic rings, sometimes requiring careful reaction monitoring to avoid over-substitution or degradation. The benzonitrile group frequently participates in cycloaddition and functionalization, opening new chemical space for medicinal chemistry. In agricultural chemistry, tweaks to the substitution pattern can dramatically alter bioactivity, an effect that underscores just how carefully downstream modifications need to be planned.
Naming conventions for this compound vary, and this has caused more than a little confusion during inter-lab communication or supplier orders. Chemists use names like 2,6-Dichloro-3-Fluorobenzonitrile, 3-Fluoro-2,6-Dichlorobenzonitrile, and sometimes abbreviated codes in research papers. Each synonym refers to the same core chemical entity, yet ambiguity in labeling can translate into costly mistakes, especially when regulatory filings, product registrations, or customs documentation get involved. These nomenclature quirks remind me of the time our lab accidentally received a related isomer, setting our project back a week and providing a humbling lesson in the importance of chemical communication.
Halogenated aromatics bring a set of safety considerations that anyone who works in chemical production or laboratories knows well. I’ve seen 2,6-Dichloro-3-Fluorobenzonitrile handled with nitrile gloves, safety goggles, and strict fume hood protocols. Skin contact and inhalation should be minimized, not just because of toxicity concerns, but also due to the persistent nature of halogenated compounds in biological systems. It’s not just about individual risk; safe handling practices keep chemicals out of wastewater and the environment, which becomes increasingly important as regulatory scrutiny ramps up. Today’s plants and labs implement SOPs that go far beyond the minimum, including closed transfer systems, well-maintained ventilation, and spill kits at the ready.
In my experience, 2,6-Dichloro-3-Fluorobenzonitrile sits squarely at the intersection of the agricultural chemical and pharmaceutical industries. It’s a favored intermediate in the synthesis of herbicides and fungicides, delivering selectivity that helps reduce off-target effects and environmental burden. For pharmaceuticals, its chloro and fluoro substitutions provide a bridge to unique biological activities, since specific halogen placements can dramatically enhance target binding or metabolic stability. Material science applications exist, mostly as specialty intermediates for electronic chemicals, where stability and tailored reactivity are prized. At every step, the presence of nitrile and halogen groups means companies can reach further into newly patentable chemical space.
The search for next-generation agrochemicals and pharmaceuticals keeps demand for new synthetic routes alive. Chemists everywhere seek to optimize yields, improve atom economy, and reduce environmental impact. The trend now points to greener halogenation and cyanation methods. Replacing old-school chlorinating and fluorinating agents with milder, more selective reagents reflects a commitment to worker safety and sustainability. Collaborative industry-academia projects have accelerated improvements in catalyst design and process intensification. It’s encouraging to see how fast innovation cycles have become, fueled in no small part by open analytical data and high-throughput synthesis. Once-slow iterative optimization now feeds directly into scalable processes. From my time in the lab, I’ve learned that having an experienced process chemist on the team pays off, catching scale-up pitfalls before they become expensive failures.
Rigorous safety assessment defines the onward journey of this compound from factory to field. Toxicological studies, both acute and chronic, focus on the impact of both parent compound and any plausible degradation products. The presence of multiple halogen atoms triggers specific concerns about environmental persistence and potential endocrine disruption. Safety data often drives formulation adjustments, containment strategies, and waste management improvements. Newer regulations in the EU, North America, and parts of Asia have raised the bar for acceptable emissions and workplace exposures. Ongoing research examines bioaccumulation tendencies, aquatic toxicity, and genotoxicity, sometimes stalling product launches as new data unfolds. Anecdotally, I’ve seen project managers spend weeks chasing down fresh toxicity data in response to a change in regulatory landscape. Honest conversations about uncertainties and safety gaps are taking root, driving more responsible development pipelines.
Looking ahead, 2,6-Dichloro-3-Fluorobenzonitrile stands poised to benefit from ongoing demand for smarter molecules in crop science and pharmaceuticals. Progress in catalysis, especially in the selective introduction of halogens and cyanide groups, could bring not just new products but cleaner, lower-waste streams. Regulatory trends point to tougher scrutiny and higher transparency expectations for complex intermediates, suggesting that robust safety and environmental data will become a non-negotiable feature of commercial success. The market for designer building blocks shows no sign of slowing, and as digital tools like retrosynthetic planning and reaction prediction engines become standard, the value of flexible, modifiable molecules only grows. Greater public scrutiny of chemical footprints will keep companies invested in safer, greener chemistry, transforming both the perception and practical reality of specialty chemical manufacturing. My own experience has shown that meaningful progress comes from listening to both frontline staff and regulatory feedback, and never losing sight of chemistry’s impact far beyond the laboratory door.
Some people find organic chemistry daunting, full of long words and complicated diagrams. I get it. Yet the structure of something like 2,6-dichloro-3-fluorobenzonitrile isn’t just another puzzle for the chemistry crowd. It sits at the crossroads of what makes modern chemical technology fascinating — you get to see how a few tweaks on a simple benzene ring open doors for all sorts of useful compounds.
At its core, this molecule starts with a benzene ring, familiar to anyone who’s stared at a high school chemistry textbook. Add two chlorine atoms on the second and sixth positions. Pop a fluorine atom at the third spot. Attach a nitrile group (-CN) to make it a benzonitrile. That’s it on paper, but its meaning stretches a lot wider.
Chlorine and fluorine don’t just show up here for show. They shape how this compound reacts — and what it can do for us. In industry, molecules like this get picked apart and rebuilt into crop protection products, pharmaceuticals, or specialty polymers. These aren’t random choices. That fluorine atom, in particular, gets plenty of spotlight. It can make the resulting chemicals more stable or change how enzymes, pests, or bacteria interact with the molecule. Add chlorines at those two spots, and you start controlling both physical properties and reactivity pretty specifically.
That’s where experience kicks in. I recall working in a research lab where the introduction of a single chlorine atom meant starting over after months of failed crystal formation. Chemical synthesis doesn’t just involve mixing things together and hoping for the best. It demands precision, and every atom counts. Changing positions — swapping a chlorine from two to four — can mean the difference between a compound that melts at room temperature or one that refuses to dissolve in anything but hot acid.
Knowledge about structure shapes regulation, safety, and even public trust. Compounds with chlorines and fluorines throw up questions about persistence in the environment. Chlorine atoms on the benzene ring, for example, affect how soil microbes break the chemical down. The nitrile group has its own story, driving reactivity that can lead to other products — sometimes useful, sometimes harmful. Taking shortcuts or skipping a thorough study of structure has led to real-world consequences before. Cases of chemical pollution and poorly understood toxicity tricks echo through news stories and environmental reports.
Chemists and regulators face a challenge: balance innovation with responsibility. In our current context, accurate structural information flows directly into risk assessment models, guides product design, and leads to more sustainable chemistry. That means demanding public databases, promoting research openness, and training the next generation to respect both the enormous power and the possible fallout of manipulating molecules.
Looking at 2,6-dichloro-3-fluorobenzonitrile shows a tiny snapshot of the wider dance between science, society, and the environment. Chemical structure isn’t just for textbooks. It’s a real-world driver for progress, for risk, and for new possibility. When the structure is clear, both science and policy have a fighting chance to keep pace with what we invent next — and what we later find in the world around us.
You don’t see folks talking about 2,6-Dichloro-3-Fluorobenzonitrile at family dinners. Still, it’s one of those compounds that pops up more than you’d think, especially when you start asking around in labs or chemical plants. I’ve learned over the years, sometimes the chemicals that rarely make the headlines form the backbone of industries others depend on every day.
Most mention this compound in the same breath as crop protection. Think about how many times a year fields get sprayed to keep weeds from invading our wheat or corn. 2,6-Dichloro-3-Fluorobenzonitrile acts as an important starting point in making special herbicides. It’s the sort of building block you don’t see, but without it, a whole portion of agricultural production wouldn’t stand a chance against tough weeds.
The process isn’t just a matter of mixing things together. It takes precision: careful addition, right conditions, and experienced hands. Chemical engineers work with this compound because it helps give rise to molecules that target weeds or pests without killing off entire crops. This selectivity boosts yields and reduces the pressure on food prices, something everyone feels in their supermarket bill.
Medicine production keeps looking for faster, cleaner ways to put together molecules for antibiotics, cancer drugs, or neurological treatments. While the limelight tends to stay on newer molecules, the industry leans on older, reliable compounds to form hard-to-synthesize pieces: 2,6-Dichloro-3-Fluorobenzonitrile is one of these. Chemists use it for assembling unique rings and frameworks found in modern drugs.
Getting these substances ready for mass use means following strict procedures. It takes trained staff and sound management to stop cross-contamination and limit waste. Recycling leftover materials and using efficient reactors make these processes safer for both workers and the environment. Good manufacturing practices follow from watchdog groups, academic research, and lessons learned when things go wrong.
Every industry has a footprint. Making and handling compounds like 2,6-Dichloro-3-Fluorobenzonitrile can bring risk — both to workers and to communities near chemical plants. In my experience, regular safety drills and testing equipment keep accidents in check. Information sharing between companies helps, too. Mistakes in handling or disposal have led to polluted water or unhealthy air in the past. The lesson: don’t cut corners.
Better tracking systems, fast spill response, and updated training stop most accidents before they spread. Old storage tanks no longer cut it; operators now invest in better containment and sensors to catch leaks. Besides, there’s a push to build smaller, modular reactors, so a problem in one unit doesn’t become a plant-wide emergency. Real progress comes from practical experience, not just rulebooks.
2,6-Dichloro-3-Fluorobenzonitrile keeps proving its worth as a chemical starter. Whether the next breakthrough lands in farming, medicine, or new materials, it’s clear that safety and environmental care need to match the pace of innovation. For my part, I hope future generations see fewer accidents and more smart chemistry. Progress isn’t always about something new — sometimes it’s about using what we have, but using it better.
Every year, mishaps in laboratories, warehouses, and even university classrooms trace back to someone underestimating the hazards sitting inside neatly labeled bottles. On my first afternoon volunteering in a small research lab, a misstep with a much less stubborn chemical put the whole place on lockdown while a veteran calmly mopped up an unexpected spill. The lesson landed fast: all chemicals come with a personality, and 2,6-dichloro-3-fluorobenzonitrile isn’t shy about showing its more irritating side if ignored.
2,6-dichloro-3-fluorobenzonitrile serves as a raw material in pharmaceutical and agrochemical stuff. Its molecular makeup brings together chlorine, fluorine, and nitrile groups, and that combination means it packs a punch in the irritation department. Direct contact with skin or eyes spells trouble. Vapors can catch you off guard, biting at the nose and throat. There’s nothing macho or smart about testing your luck—once this stuff gets under the skin, gloves and goggles start feeling like a small price to pay.
The need for barriers isn’t about regulations trying to make life difficult—it’s about protecting people. Thousands of chemical injuries every year involve simple steps skipped: not using gloves, skipping eye protection, or pouring without a fume hood. In my own experience, the people who dodge PPE tend to gamble until luck runs out.
Anyone handling a chlorinated nitrile compound needs quality gloves—nitrile or neoprene beats the usual latex. Chemical splash goggles leave far less room for regret than ordinary glasses. Good ventilation isn’t a bonus—it’s a requirement. Lab hoods exist for a reason and keeping up with their maintenance means less guessing about air quality. It’s too easy to convince yourself the air “doesn’t smell too strong,” but irritation often sneaks up quietly, and long-term effects can add up.
It’s tempting to treat chemical waste as an afterthought, especially when you’re finishing a long day. But proper storage in tightly-sealed, labeled containers makes a difference. Mixing incompatible chemicals sets the stage for leaks, fires, or toxic fumes. Disposing of waste in line with local rules isn’t just about ticking a box—it builds a safety net for the entire crew.
Too many organizations stick laminated safety checklists on the wall and call it a day. A better path involves hands-on training and regular refreshers, led by someone who’s seen what complacency can cost. The best labs inspire respect for both the science and the risks, and that culture bleeds out into every decision. Too often, I’ve seen newcomers start off responsible, then cut corners after they get comfortable. Good leaders in science make safety habits stick by calling out shortcuts and supporting open conversations about near misses.
Staying curious about chemical hazards isn’t just about passing an audit or avoiding fines. It builds trust among team members and keeps the entire operation running without unwelcome surprises. Once you see just one accident that hardens into a long-term health problem, the daily rituals of safe handling stop feeling burdensome and start looking like plain old common sense.
There’s a certain kind of trust that goes into every bottle, drum, or sack of industrial chemicals. You count on what’s inside to be what’s promised on the label, especially when handling tricky compounds like 2,6-Dichloro-3-Fluorobenzonitrile. This isn’t something most folks run across in daily life, but in the worlds of pharmaceuticals, crop protection, and specialty chemicals, it plays a quiet but vital role. The purity of this compound says a lot about the dependability of what comes next down the line—be it a crop-safe pesticide, a research-grade pharmaceutical intermediate, or just a batch of something that keeps the wheels of industry turning.
Anyone who’s handled chemicals in a lab understands the frustration of an experiment derailed by impurities. Even a tiny fraction of contamination can throw off results, waste money, or worse, create unsafe conditions. Common practice for aromatic nitriles of this class involves aiming for a purity of at least 98%. Top-grade batches push higher—with 99% or above often demanded for more sensitive applications. That sort of number isn’t just paperwork. It’s a stamp of reliability that means production teams and researchers alike can focus on building something useful, not troubleshooting mystery outcomes.
Hitting that level isn’t simply a matter of running a factory a certain way. Careful control during synthesis, meticulous purification steps, and strict packaging routines all play a part. Companies use chromatography and spectroscopy to check for the tiniest blips of leftover starting material or side products. A single impurity might block a critical reaction or gum up machinery during scale-up. Even unnoticed, those leftover fragments can spell regulatory headaches or diminished yields, so most buyers want a clear certificate proving their lot runs at the expected quality.
The way I see it, the whole notion of purity in chemicals mirrors a larger point about attention to detail. Take a look at the regulations around pesticide ingredients: if a supplier ships a batch that misses the specs by half a percent, years of safety trials and entire product registrations sink into uncertainty. In the pharma world, that same half percent can bring delays, extra batch testing, or a total recall.
Much of my own experience with chemicals came from early mornings in university labs. The professors made a big deal out of labeling and double-checking reagents. We learned the hard way that chasing “just-good-enough” purity meant watching reactions fizzle or failing every quality control check. On an industrial scale, that lesson sticks. Laboratories rely on those tight specifications so the chain of trust doesn’t break somewhere unseen.
There's a big push these days for better transparency and more rigorous quality controls. Third-party audits, traceable lot numbers, and detailed impurity profiles are more common. Labs and purchasing managers don’t just want assurance that the bulk is pure—they’re asking for breakdowns of what, exactly, the impurities might be. This demand for detail comes from experience; surprises in the bottle often mean expensive delays outside the lab.
Tools have gotten better. Advanced chromatography and mass spectrometry help manufacturers not only hit high targets for purity, but also spot trace contaminants that flew under the radar a decade ago. With supply chains under scrutiny worldwide, customers expect more than just a purity number. They want evidence and integrity every step of the way.
Good chemistry doesn’t shy away from the hard numbers. The best shops meet the standard—and prove it—because everything that follows, from medical breakthroughs to safer farms, starts with getting the little things right.
Chemicals like 2,6-Dichloro-3-Fluorobenzonitrile rarely make headlines, but anyone working in a lab or industrial environment recognizes how something as simple as careless storage can lead to problems fast. With a background in organic synthesis, I’ve gotten a front-row seat to both good and bad habits. Once, a friend left a bottle of this compound in a regular cabinet near the sunlight. A couple of weeks later, the label turned brown and the cap showed serious degradation. That’s when the importance of smart storage really sank in.
This compound comes with its own set of challenges. It’s not just the strong smell or the potential for skin and respiratory irritation. It breaks down faster when hit by heat or light and, like many halogenated organics, sits right on the edge of reactivity. Wrong storage can turn valuable chemicals into hazardous waste. Over the years, I’ve seen chemicals in high school storerooms forming odd crystals or leaky bottles in chilly supply closets. These mistakes aren’t just inconvenient—they’re dangerous.
Storing 2,6-Dichloro-3-Fluorobenzonitrile calls for a steady, low-humidity environment, away from strong light or sources of heat. Fluctuating temperatures lead to condensation, which risks clumping up powders or making liquids separate. In university labs, we always set aside a lockable, ventilated chemical storage cabinet—no exceptions. As the years passed, I learned to triple-check that bottles are tightly sealed and labels remain legible. After one scare with an illegible label, I adopted the habit of using fade-proof markers.
Some folks think any shelf will do, but mixing this compound with incompatible substances, especially strong acids or oxidizers, is just asking for trouble. The reality is, segregating chemicals is not about bureaucracy or red tape. I’ve watched chemical reactions go sideways simply because a spill happened between two bottles that shouldn’t have sat shoulder to shoulder. The separation of incompatible materials is an easy, powerful way to avoid headaches or worse.
Even with the best precautions, accidents can sneak in. In my early years, I saw a careless attempt to open a sticky cap turn into a dose of liquid on someone’s gloves. Good lab design includes ready access to eyewash stations, spill kits, and ventilation—small investments that have paid off more than once. Quick response only works if you don’t have to fumble through unrelated clutter to find the tools you need.
It’s easy to get complacent. Some believe if nothing bad has happened yet, nothing ever will. The reality doesn’t match that optimism. Making solid guidelines for chemical storage and making them stick by regular oversight has worked in labs I’ve managed. Sometimes it's about running through quick monthly checks. Sometimes it's about staff training sessions to refresh everyone’s memory on best practices. People working with chemicals take on real-world risks—tackling those risks head-on doesn’t just protect products or projects, but lives and livelihoods as well.