Chemistry, like life, evolves through a maze of chances and choices. 2,4-Difluoro-3-methoxybenzoic acid, a name that only chemists could instantly recall, marks a point in the development of halogenated aromatic carboxylic acids. Decades back, as the demand for selective fluorinated compounds increased in pharmaceutical research, scientists started focusing on compounds that could tweak biological activity without overhauling the foundational chemistry. The arrival of compounds like this one didn’t exactly make newspaper headlines, yet inside chemical catalogs and research labs, each new derivative stood as a testament to curiosity and the urge to build better medicines, agrochemicals, and advanced materials. In my own research days, combing through early literature, I remember stumbling across bench notes full of analogs, each with slightly different substitutions, because the journey was always about mapping structure to function, not a race to publish.
Picture a white or off-white powder, often slightly pungent if handled outside the fume hood, showing up in small bottles labelled only for research use. Few outside the lab would appreciate how each aromatic ring, altered with a couple of fluorines and a methoxy group, controls the reactivity, solubility, and even safety profile of the compound. In medicinal chemistry circles, products like this one fit into bigger strategies — fine-tuning molecules until the desired biological response matches up with the theory. The commercial world doesn’t glamorize such intermediates, but anyone chasing a reliable fluorinated building block remembers the days before these compounds were a click away.
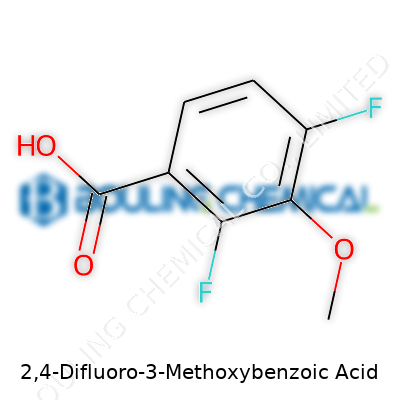
Many would see just a chemical formula, C8H6F2O3, but it actually gives away a lot about the molecule. The two fluorines bring noticeable electronegativity, creating a much less reactive aromatic ring than many benzoic acids, especially one with electron-donating methoxy tossed into the mix. Such stability stands out during synthesis—where some analogs fall apart under heat or strong base, this one holds the line. Handling this compound, the weight feels no different from others like it, but in a reaction flask, small differences in melting point or boiling point can mean fewer headaches in purification. We all appreciate when an acid stays an acid and doesn’t fitfully transform the minute the protocol gets tough.
Specifications reflect how the work gets done, not just what a label says. Purity, water content, trace metals — these details separate smooth syntheses from those marred by side reactions. Ask anyone who’s worked a late night shift trying to isolate a product and they’ll tell you that knowing the limits is better than chasing a phantom impurity. Still, reliance on technical sheets should never substitute for vigilance with analytical tools, especially HPLC or NMR. Labels tell a story, but it’s on the chemist to test, verify, and only trust results confirmed in their own glassware.
Most of the time, synthesis starts with the corresponding methoxy-benzoic acid, then introduces the two fluorines through selective halogenation—a tricky dance when every extra electron can shift the outcome. Routes vary: classic methods rely on directed ortho metalation or electrophilic substitution, and often call for specialized reagents nobody wants to spill on the bench. An efficient route means less waste, fewer purification steps, and a batch that meets the demands of downstream chemistry. Once, I spent a week chasing a robust route that yielded more tar than product, a reminder that reaction planning matters as much as the glassware streaked with fingerprints.
Once the bottle’s open, chemists eye possibilities for alteration. The carboxylic acid group lends itself to forming esters, amides, or using the acid in coupling reactions that build more complex scaffolds. The methoxy function tempts demethylation for those looking to prod the electronic nature of the ring. Fluorines—often prized for their impact on metabolic stability in drug design—mostly resist substitution, preserving the unique flavor of the starting material. Real innovation rarely shouts; it grows from relentless attempts to coax small changes until they yield newer, better outcomes, often deep into the evening when the safety lights flicker overhead.
In the world of chemical research, names shift as fast as company ownership and catalog numbers. “2,4-Difluoro-3-methoxybenzoic acid” sits snug in IUPAC territory, yet old-timers still prefer shorthand like DFMB acid or numbers and letters jotted in margin notes. Ask for synonyms and you might get trade names or local abbreviations—an alphabet soup signaling either comfort or confusion in crowded lab spaces. No matter the moniker, clarity always takes precedence, especially with international teams or shared projects relying on tight communication.
Safety is less about paperwork and more about habit. For anyone who’s breathed a little too deeply near spilled organic acid, vigilance sharpens quickly. Chemical safety data for compounds like this one push for gloves, goggles, and proper ventilation. Even small exposure risks, like mild irritation or long-term inhalation problems, call for respect. Spilled powder doesn’t clean itself up, and one careless move means a mess for others. Years of standing in front of a fume hood, I came to appreciate that operational standards are a community act. The best-run labs share information, enforce boundaries, and look out for the distracted newcomer eyeing an unfamiliar reagent.
In the hands of the right researcher, 2,4-difluoro-3-methoxybenzoic acid moves from curiosity to utility. Medicinal chemistry teams test derivatives for enzyme inhibition and biological signaling effects. Material scientists explore modifications for new polymers or as starting points in surface treatments where fluorine’s durability matters. Anyone pursuing crop protection chemistry hunts for new pathways around regulatory bans, often starting from established aromatic acids. My own experience working alongside colleagues in agrochemical research proved how foundational the right precursor could be; product improvement often comes from subtle molecular change rather than headline-grabbing new invention.
Innovation comes in stubborn increments. Modern research on this compound isn’t just about what the molecule does, but how easily it can fit into high-throughput screening or automated synthesis. Computational tools push chemists to rethink old synthetic routes, driving up yields while trimming cost and waste. Fluorinated building blocks remain relevant as big pharma pushes for drugs that resist metabolic breakdown. Where profits and patient safety unite, funding keeps flowing toward enhanced derivatives and greener, more selective methods—a trend I’ve watched grow from a niche pursuit to standard expectation in well-run labs.
Any discussion worth having about synthetic compounds faces the toxicity question head-on. For plenty of halogenated aromatic acids, acute exposure doesn’t raise red flags, but long-term effects ought never get lost in the shuffle of deadlines and pressure. Chronic exposure, environmental breakdown products, and the uncharted territory of cumulative dosing in living systems demand transparency. Looking back on some early studies, the culture around toxicity data has come a long way: from cagey one-paragraph summaries to open-access datasets and third-party verification. Community trust depends on evidence, careful classification, and immediate action when surprises turn up on the test bench.
There’s always another challenge down the line. For every compound that made a mark, dozens slipped quietly into the chemical archives—a story only researchers hunting through old journals will ever find. The future of 2,4-difluoro-3-methoxybenzoic acid circles around sustainability, safer manufacturing, and smarter functionalization. As automation creeps into every corner of synthesis, precision and reproducibility will matter even more. The demand for fluorinated platforms stays stubbornly high in pharmaceuticals and electronics, and many will soon ask for greener methods and more legible life-cycle data. I’ve seen trends in green chemistry take seemingly obscure compounds and move them to center stage. So long as the questions keep coming, people working in labs, classrooms, and startups will keep reshaping molecules like this one in response to a changing world.
Chemicals like 2,4-Difluoro-3-Methoxybenzoic Acid rarely turn up in mainstream headlines, but in the world of science and manufacturing, their importance runs deep. I’ve seen how such specialty compounds, maybe with a name only a chemist could love, act as the silent backbone in key processes. The value starts in the laboratory, where 2,4-Difluoro-3-Methoxybenzoic Acid becomes one of those tools that researchers lean on when they’re building or tweaking molecules, especially in the pharmaceutical and agrochemical fields.
Drug discovery stands or falls on reliable building blocks. With its unique combination of fluorine atoms and a methoxy group, this compound plays a major part in shaping molecules with the right activity and safety. Research groups favor it as a synthetic intermediate—meaning it helps them string together more complex molecules, often as they chase new treatments for hard-to-treat diseases.Fluorinated chemicals get chosen a lot because they help drugs last longer in the body and improve how the drug behaves—the so-called pharmacokinetics. This acid, in particular, opens doors to new derivatives that don’t show up naturally. It finds itself in the foundation layer as scientists look to optimize drug candidates, making a big difference in what finally ends up in a pill or vial.
Farming chemistry relies just as much on clever molecules. Agrochemical developers work in a constant contest against resistant pests and persistent plant diseases. Engineers behind new herbicides and fungicides aren’t just searching for the next “big compound” but are improving existing agents. Here, too, 2,4-Difluoro-3-Methoxybenzoic Acid enters as a synthetic stepping stone. With changing weather and stricter environmental policies, companies have to deliver solutions that tread lighter but protect yields. By adding fluorine or methoxy groups through compounds like this, chemists tweak toxicity and field-life, so treatments stick around just long enough to work, then break down more safely.
Easy as it is to praise the technical achievements, chemists and companies face real questions about safety and sustainability. Handling fluorinated materials brings concern—both for those making it and for environmental impact down the line. In labs I’ve worked in, extra care goes into collection and disposal protocols. Waste treatment becomes essential, and government regulations around fluorinated chemicals grow tighter each year.Demand for transparent supply chains in both pharma and agriculture puts pressure on producers. To step up, companies should invest in greener synthesis routes and better process controls. Green chemistry—making products with fewer solvents, less waste, and more renewable sources—should sit high on every chemist’s to-do list.
For all the innovation that 2,4-Difluoro-3-Methoxybenzoic Acid supports, we can’t skip the hard parts—staying honest about chemical exposures, working on cleaner production, and tracking where molecules end up when their job’s finished. Teams pushing into new drug or crop treatments have to balance cost, effect, and downstream impact every step of the way. That’s where hard-earned lab experience, up-to-date fact-checking, and careful planning matter. In my years on research teams, genuine breakthroughs always followed when people paired these compounds with clear goals for safety and stewardship.
Science classrooms teach basic molecular diagrams, but the real heart of chemistry beats in the details that make each compound unique. Take 2,4-Difluoro-3-Methoxybenzoic Acid. It isn’t just another tongue-twister among thousands of chemical names; its blueprint reveals a lot about the way modern chemistry intersects with medicine, agriculture, and technology.
This compound carries a benzene ring, which forms the core. “2,4-difluoro” means two fluorine atoms replace hydrogen atoms at the 2 and 4 positions on the ring. There’s a methoxy group — an oxygen attached to a methyl — at the 3 position. At the end sits a carboxylic acid group attached directly to the ring. Put this together and you get a molecular formula of C8H6F2O3.
Mapping these features onto paper with the classic hexagon makes things more concrete, but understanding grows sharper when you pause to consider what these groups bring. Fluorine carries enormous weight in chemistry: its tiny, electronegative nature changes a molecule’s reactivity, stability, and biological activity. Adding methoxy groups can tune solubility or block certain reactions. The carboxylic acid part opens the door to salt formation, solubilization, or further derivatization.
I’ve seen chemists spend months tweaking substitutions like these, hoping for a compound with better pharmaceutical action, less toxicity, or greater persistence in a pesticide. Even a single fluoro group swapped for a hydroxyl or a hydrogen can spell the difference between a breakthrough and a dead end. In fact, over half of new drugs approved in the last few years contain at least one fluorine atom. That’s not by accident: careful alterations to a benzoic acid core give researchers precise control over how a substance enters, survives, or leaves the human body.
These details don’t stay locked in the lab. They filter into products we use every day, from headache pills to crop protection. Poorly considered changes can make molecules too toxic or environmentally unfriendly. I’ve watched regulatory hurdles trip up projects when structural tweaks made a harmless molecule into something risky.
Chemists now rely on deep knowledge — both learned and passed down — to weigh benefits against possible downsides. Environmental persistence, bioaccumulation, off-target effects: these concerns push for smarter, greener chemistry. That means formulating substances that break down safely or do their job with less collateral impact, all starting from that basic aromatic ring.
If the goal is safer medicine or cleaner farming, solutions must begin at the molecular level. Technologies such as artificial intelligence speed up the hunt for promising new variations. Chemists who mix curiosity with responsibility help society sidestep old mistakes like persistent organic pollutants or unexpected drug side effects. In my experience, this isn’t just hype — it’s an outlook rooted in the stubborn realities of molecule-by-molecule discovery.
So when someone brings up 2,4-Difluoro-3-Methoxybenzoic Acid, that collection of fluorines, oxygen, and carbons isn’t just trivia — it’s the starting line for innovations that shape the world outside the laboratory walls.
Anyone who’s worked in a chemistry lab knows every substance has quirks, and some, like 2,4-Difluoro-3-Methoxybenzoic Acid, deserve respect for how easily routine practices can slip into oversight. I recall my first research assistant stint—labels faded, caps loosened, refrigerators jammed with mystery vials. The lesson stuck: chemistry rewards attention and safe storage habits, not shortcuts.
This compound doesn’t land in the “immediately dangerous to life” category, but it brings the same risks many benzoic acid derivatives carry. Solids like this can last years if air and moisture keep their distance. In real experience, keeping bottles sealed tight and marked with the date does more to keep purity than any fancy storage system ever could.
Temperature tells a big part of the story. I’ve seen well-meaning students pop organics straight into the freezer, thinking cold cures all, then wonder why condensation forms under the cap the minute it’s taken out for use. For 2,4-Difluoro-3-Methoxybenzoic Acid, room temperature works unless direct sunlight heats up a workspace. Fluctuating temperatures invite degradation, so a shelf away from windows and out of traffic gives better control. Think of it like keeping coffee fresh—stick it in a cupboard, not on a sunny windowsill.
I once watched a chemical supplier’s safety officer hold up a stained paper towel and talk about how water vapor finds any opening. Even stable powders suck in moisture over time, and that can start trouble for both experiments and disposal costs. Taking small steps—dessicants in the container, dry hands on the lid, fast returns to the jar—prevents clumping and preserves reactivity. I’ve lost too many sample batches to simple humidity to ever skip these precautions.
Avoid mixing benzoic acids with strong oxidizers and acids in the same cabinet. Cross-contamination happens fast. Even a few crystals in the wrong spot bring headaches during spill cleanups or, worse, stray reactions. Labels, secondary containment—these aren’t just lab rules, they’re practical tools that pay dividends in safety and cost.
Regulations ask us to store hazardous chemicals with clear labels and an inventory system. From my time training new lab techs, I saw how easy it is for small missteps—missing a date, skipping an entry—to grow into real hazards. Regular audits, prompt disposal of old material, and access to up-to-date safety data sheets build a safer culture, not just compliance. It’s easy to treat storage as an afterthought, but the costs—experiment failure, waste, or even personal injury—show up fast in this field.
Good stewardship of chemicals isn’t rocket science, but it does demand respect and habits that grow with experience. It’s worth it, not just for science’s sake, but for the people behind the beakers. That’s a lesson that sticks long after you’ve moved on from the lab bench.
There’s something immediate about chemistry when a pungent solvent or an odd-smelling acid creeps out of a fume hood. Years spent working in lab spaces tell me that familiarity can convince people to cut corners. With 2,4-Difluoro-3-Methoxybenzoic Acid, the clear, white powder doesn’t shout danger, but the risks sit just beneath the surface. You won’t see smoke or sparks, but respiratory irritation and skin sensitivity aren’t rare. It’s the daily routine—measuring, weighing, transferring—that can make someone forget just how quickly a mistake turns into an accident.
The most basic step starts at the container. Reading and understanding hazard labels isn’t about compliance. It’s about memory—almost everyone in research or industry has seen gloves eaten through by an unexpected spill. For this compound, inhalation promises trouble. Use it in a well-ventilated spot, ideally inside a fume hood. Control means turning that sash down to eye level, not propping it up for convenience. Don’t fool yourself into feeling safe in a closed room with a fine powder.
Gloves should never be afterthoughts. Nitrile types stand up well to aromatic acids like this one, lasting longer than latex in a pinch. Change them often and watch for pinholes—a few seconds checking beats hours spent treating a rash. Eye protection means tight-fitting goggles, not slim glasses. I learned more than once how a splash slips under regular specs. If you think you look silly, just ask anyone who’s washed their eyes for fifteen minutes at the eyewash station.
Forgetting about ventilation gets people in trouble fast. Even small samples disperse dust. A gentle breeze from a fume hood does more than exhaust fumes; it keeps those invisible particles away from your nose and mouth. Wearing an N95 mask gives an extra shield, especially when weighing the powder. Don’t trust your sense of smell to warn you; many chemicals announce their presence only after causing harm.
Lunch at your bench looks efficient, but it’s a dangerous routine. Cross-contamination from chemicals—especially with fluorinated aromatics—raises real risk. Wash hands with soap, not just water, after handling. This step matters most before grabbing a snack or answering your phone. The habit sticks with you, and it is worth repeating for anyone new to a lab.
Small spills happen. Sweep up powder with damp towels, not dry ones, to keep dust from scattering. Dispose of all contaminated materials in the proper waste bin—do not toss them into regular trash. For anything bigger, call in the trained spill response crew. Trying to solve a big spill solo can spiral out of control. Don’t cut costs by ignoring proper disposal—waste facilities exist for a reason, and shortcuts pile up risks for everyone in the lab.
Safety culture in the lab boils down to habits: reading labels, using barriers like gloves and goggles, maintaining good airflow, and respecting the line between food and chemistry. These steps feel routine until someone skips one and shows up in the ER. It’s worth treating every new bottle as your first. This attitude not only keeps your skin unharmed and your lungs clear, it builds a workplace where sharing best practices comes naturally.
A Certificate of Analysis, or CoA, comes up often for anyone in research or chemical procurement. For a compound like 2,4-Difluoro-3-Methoxybenzoic Acid, its presence signals more than bureaucracy. Over years of working in labs and project teams, I learned that the CoA tells a real story—purity, method of analysis, batch records, actual figures, not just bold claims. It’s the difference between running a successful set of reactions and spending weeks troubleshooting mysterious failures.
Chemists who lean too much on luck risk setbacks. A small impurity in an intermediate like 2,4-Difluoro-3-Methoxybenzoic Acid can change catalyst performance, hide spectral signals, or skew yields. In pharmaceuticals, the stakes run even higher—trace byproducts can bring regulatory rejection or endanger patients.
It surprises new researchers how much time solid documentation actually saves. Instead of checking and rechecking mysteries with hush-hush suppliers, it’s faster to source from a distributor or manufacturer who shares a complete CoA. I remember one project where my team spent weeks unraveling why an expected compound didn't appear after a coupling step. Only after chasing down the supplier did we see the CoA—an odd impurity just below the “acceptance criteria.” It made us rethink how our mating of starting materials could mislead unless we actually checked those upstream purity levels every time.
A CoA for 2,4-Difluoro-3-Methoxybenzoic Acid, if offered, typically lists real assay data, impurity profile, melting point, and the methods used for characterization—NMR, IR, even mass spectrometry when available. This paper trail builds confidence. The best labs I worked in made it a standard step to tick off criteria before any new chemical hit the weigh balance. Only then did the science move forward, rather than backward into troubleshooting.
No magic exists in a CoA alone, though. Not every supplier issues these documents by default, especially for chemicals used mostly as research intermediates or at small scales. Sometimes it takes an extra request, but responsible providers understand that transparency helps everyone. In my experience, local suppliers hesitated more often, citing cost or batch size, while larger distributors had online systems for instant retrieval. This digital access cuts down waiting and reduces miscommunication.
Some worry that CoAs create legal risk or cost, but the real cost lands on researchers if they chase after contaminants down the line. Academic labs, short on funds, sometimes roll the dice. Honestly, it would be easier if the research community and industry demanded batch documentation as standard practice, even for less common intermediates. Professional societies and journals push this more now, making clear that solid sourcing and supporting data are part of reproducibility.
Making the CoA standard for compounds like 2,4-Difluoro-3-Methoxybenzoic Acid puts power in the hands of the chemist. Sourcing only from providers who publish these details openly—even at slightly higher price—usually pays off when projects stay on track and unexpected reruns disappear. Scientists, purchasing agents, and anyone handling chemicals can shift the market by demanding transparency. That’s how the cycle of quality gets stronger, for this compound and everything else on the bench.