Decades ago, chemists searching for new building blocks to expand pharmaceutical and agrochemical libraries found something special in halogenated benzoic acids. 2,4-Dichloro-3-Cyano-5-Fluorobenzoic acid—mouthful of a name—emerged from this phase. Its roots trace back to targeted efforts that tried to combine electron-withdrawing groups on an aromatic ring, betting that the resulting molecule would display unique reactivity. By integrating chlorine, a cyanide, and fluorine onto the same benzene backbone, early innovators were attempting to engineer molecules ready to join reactions that other benzoates could not manage. The development reminds us of just how much scientific progress depends on old-fashioned curiosity.
If you hold a sample of this compound, you see a white to off-white powder, a typical appearance for many small organic acids. The fluorine and chlorine atoms pack the ring with electron-hungry force, shifting both its physical behavior and chemical attitude. The cyanide adds another layer. At room temperature, the solid seems unassuming, but these substitutions push the melting point higher or make it resist easy dissolution in plain water. Solubility opens up in polar aprotic solvents like DMSO or DMF. In the lab, this property frustrates people trying to run simple aqueous extractions, but helps for controlled, anhydrous reactions.
Putting together 2,4-dichloro-3-cyano-5-fluorobenzoic acid means orchestrating a dance between functional groups. A typical route starts with a fluorinated benzonitrile, followed by stepwise chlorination, each addition carefully monitored to avoid over-reacting and rearranging the desired orientation. Nitrile hydrolysis, if performed under strict conditions, brings in the acid group, wrapping up the synthesis with a strong, acidic work-up. Each step must be monitored closely to prevent formation of unwanted side-products, and purification often tests the patience and technical skill of any synthetic chemist. Chromatography sometimes gets tedious, but, to the practitioner, the sight of pure, crystalline product justifies the grind.
This benzoic acid shines as a versatile starting material. The electron-deficient ring takes well to nucleophilic aromatic substitution. Chemists often replace the fluorine or one of the chlorines with amines or thiols, spinning off libraries of analogs. These strategies breathe new life into routine research, as fluorinated aromatics have pushed the design of herbicides, fungicides, and anti-inflammatory drug scaffolds. Cross-coupling reactions, in particular, love this sort of electrophilic core. Suzuki or Buchwald–Hartwig couplings take advantage of the ring’s electronic quirks, making this molecule a sort of chemical crossroads for creative reaction design.
The official IUPAC label, 2,4-dichloro-3-cyano-5-fluorobenzoic acid, isn’t easy to weave into daily speech in the lab. Depending on the research group or catalog, you might find it as 3-cyano-2,4-dichloro-5-fluorobenzoic acid, or simply “2,4-DC-3CN-5F-BA” scrawled on sample bottles. These shorthands fill notebooks and inventory lists everywhere, a sign that chemists move faster than bureaucracy. Digital databases recognize all forms, translating between synonyms as scientists dig through literature for reaction precedents and safety concerns.
Any chemist will tell you respect matters more than routine with halogenated aromatics and nitrile-bearing compounds. Gloves and eye protection form the bare minimum because skin or inhalation exposure runs risk of irritation. Waste disposal never goes down the drain—halogenated organic acids demand proper collection and incineration. Strict labeling and separating the compound from strong oxidizers or reducing agents reflects lessons learned the hard way. Fume hoods provide the best workspace, limiting inhalation of dust or trace vapors. While decades of industrial accidents have built today’s safety protocols, staying up-to-date with evolving workplace guidelines remains crucial for everyone, not just the manager with a clipboard.
Outside the lab, people might expect a molecule like this to belong strictly to textbooks. Quite the opposite. It lives quietly in factories developing new herbicides targeting resistant weeds. Research teams use it as an intermediate to tweak drug-like molecules, thanks to the benzene ring’s tunable electronics. Fluorinated and halogenated molecules have carved out space in cancer drug research—small changes on the ring alter the biological properties in sometimes unpredictable ways. Analytical chemists use this compound as a calibration standard due to its defined purity and stability. Each application stands on hard data, with teams publishing results on everything from metabolic fate in soil to how structural tweaks affect toxicity.
R&D-heavy industries cherish building blocks that promise flexible chemistry. 2,4-Dichloro-3-cyano-5-fluorobenzoic acid has found itself at the crossroads of chemical biology, where demand for new molecular scaffolds never fades. Scientists reach for it when standard benzene derivatives don’t deliver the right electronic punch. Ongoing work tries to expand its modifications, improving yield and selectivity in functionalization techniques. High-throughput screening in major pharmaceutical companies often relies on such intermediates as they explore chemical diversity in their search for tomorrow’s medicines. Patents filed in the last ten years mention this compound in more and more combinatorial syntheses and process optimizations.
Toxicity assessment moves in lockstep with chemical progress. As a halogenated benzoic acid with a cyano group, this compound merits close scrutiny. Early animal studies indicated moderate to low acute toxicity, but attention now turns to chronic effects, environmental breakdown, and any potential for bioaccumulation. Soil studies show it behaves as a persistent organic pollutant under some conditions, troubling environmental health advocates. Yet, because its use mostly occurs in tightly controlled industrial or academic contexts, direct risk to the broader public stays low. That margin narrows if downstream use increases or if waste handling gets sloppy. Ongoing toxicological research focuses on metabolites, breakdown in plants and animals, and pathways for safer degradation—a story unfolding in real time.
This molecule typifies chemical innovation running up against 21st-century sustainability challenges. Expanding research on halogenated and cyanated aromatics reveals enormous capacity to generate novel drugs and crop protection agents. Yet, every jump in use brings sharper questions about disposal, persistence, and safe laboratory habits. As green chemistry principles reshape the way molecules get designed and synthesized, research already circles around less hazardous or more biodegradable analogs. Companies and universities, keen to score both scientific and environmental wins, push for advances that balance utility with responsibility. In future years, decisions about this compound won’t rest only on reactivity or market demand; they will weigh how each new process or discovery lines up with the broader obligation to protect workers, communities, and the world beyond the lab door.
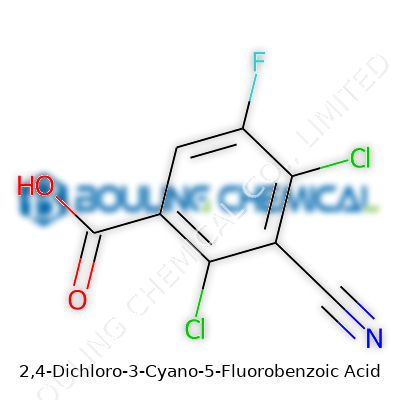
Chemistry classes bring up a lot of strange names, but few quite so specific as 2,4-Dichloro-3-Cyano-5-Fluorobenzoic Acid. The name itself tells a story. Buried inside is a benzene ring, or benzoic acid backbone, which a lot of folks know from pain relievers and food preservatives. Take that classic ring, bolt on two chlorine atoms at spots two and four, stick a cyano group at spot three, and plug a fluorine at five. Then, the carboxylic acid group claims the sixth position.
The chemical structure looks like a spoked wheel. The benzene ring forms the core, with its alternating double bonds. Chlorines, larger and more electronegative than most atoms you'd find on a benzene ring, hang off the ring at two positions. Chlorine doesn’t just add bulk; it changes the way the molecule interacts with both the air and any nearby living creatures. The cyano group, basically a carbon triple-bonded to a nitrogen, brings its own drama. People in the lab know it turns what could be a docile molecule into something sharper, both chemically and in terms of reactivity.
Fluorine, over at the fifth carbon, packs a strong punch for its size. Any chemist will tell you: swapping a hydrogen for a fluorine atom changes everything. Fluorine wants electrons so much it can shift how the molecule behaves around enzymes, bacteria, or even the soil. Money and research get spent finding ways to tune these side groups just right, to push weedkillers or medicines to do what we hope, but avoid unexpected side effects.
These details matter. Small changes to things like where the chlorine or fluorine sits can decide how well a compound decomposes, how quickly it gets cleared from a river, or how much builds up inside a berry bush. In my own soil science classes, folks pointed out how halogenated chemicals—and 2,4-Dichloro-3-Cyano-5-Fluorobenzoic Acid is no exception—hang around longer than something plain like acetic acid. Water won’t break those bonds easily. That’s good news for products that need to last, lousy news for farmers who don’t want chemical residue in runoff.
Benzoic acid derivatives fill university books on weed control and pest management. Some of these molecules work better when altered in just the right spots. Add a cyano group and the molecule often gets tougher, more persistent, but less likely to break down under sunlight. Add chlorine and you can make a weedkiller that strikes hard, but perhaps needs more care in handling and disposal.
Science keeps pushing for alternatives. Chemists and plant specialists spend long nights searching for new molecules that do the job, but don’t outstay their welcome in ground water. It’s an arms race: weeds evolve, pesticides adapt, and regulations chase after both. Designers of molecules like 2,4-Dichloro-3-Cyano-5-Fluorobenzoic Acid shoulder responsibility for studying not just its power, but its afterlife. I’ve heard more than one field researcher say they’d trade a bit of potency for a faster breakdown time, if it meant less lingering in the field.
Better monitoring, using non-halogenated alternatives, or developing targeted delivery systems offer hope. But no single tweak solves everything. The real lesson in studying the chemical structure of molecules like this one: small details shape big outcomes, for better or worse, in science and in the world beyond the beaker.
Walk into any agricultural research lab or a specialty agrochem plant, and odds are, you’ll stumble upon a lineup of bottles with long-winded names. One of them—2,4-Dichloro-3-Cyano-5-Fluorobenzoic Acid—might seem like a string of syllables only a chemist would love. But there’s more happening in that bottle than meets the eye. In my time working alongside agronomists and folks from chemical development, I’ve learned that the real impact comes not from the name, but from where this compound fits on the path between lab and food on your table.
Agriculture today isn’t about just seeds and water; it’s a high-stakes balancing act against weeds and pests that threaten harvests worldwide. This compound jumps into that scene as a building block for several modern crop protection products—especially selective herbicides. I’ve spoken to growers who rely on these formulations, and their feedback underscores one simple fact: without this kind of upstream chemistry, keeping fields productive gets a lot trickier. The underlying molecule often lands in the manufacturing routes for compounds designed to target broadleaf weeds, but spare crops like corn or sugarcane from collateral damage. That kind of selectivity and efficiency matters for both food security and farm economics.
Scientists rarely use raw 2,4-Dichloro-3-Cyano-5-Fluorobenzoic Acid directly on farms. Instead, its primary value comes as an intermediate—a stepping stone, if you like—to more complex molecules. Researchers, including friends of mine, spend days and nights fine-tuning how to tweak this benzoic acid core into newer, smarter molecules. These tweaks often boost performance against resistant weeds or bring down toxicity for non-target plants and animals. Each evolutionary step in chemistry can help farmers adapt to ever-shifting threats, instead of falling behind in a race that weeds often try to win.
Its place in the chemical supply chain draws a fair bit of debate among environmentalists, regulators, and researchers. As someone who’s attended a fair share of panel discussions, I’ve heard concerns about residues in soils and potential impacts on nearby ecosystems. While the finished herbicides derived from this intermediate go through regulatory scrutiny, the conversation doesn’t stop there. Everyone wants safer food, cleaner water, and thriving wildlife. That means greater transparency about how these chemicals are made, transported, and used matters. Universities and government bodies keep an eye on the latest research about environmental breakdown, guiding policy shifts as new data comes in.
Pathways leading from basic chemicals to finished farm products always offer room for improvement. Maybe biocatalysis can clear away some of the harsh steps in the current synthesis. Maybe digital tracking can make stockpiles less risky. In many companies, teams are chasing greener ways to make, use, and dispose of the ingredients behind our food supply. Sometimes, the best advances come from unexpected collaborations—chemists sitting down with environmental scientists, or industry leaders teaming up with watchdogs. Progress in agriculture often hinges on what happens far upstream in these chemical plants and research offices. For those of us invested in safer, more sustainable food, keeping tabs on boring-sounding acids like this one isn’t optional. It’s vital.
Walk into any warehouse or chemistry storeroom and the first thing that jumps out isn’t the ingredients themselves, but the numbers stamped next to “purity.” Lab managers watch those numbers like hawks. High purity isn’t just a box someone ticks for the sake of paperwork—it determines if a chemical meets research needs, keeps experiments reproducible, and stands up to industry audits or medical standards.
Over the years working in laboratories, nobody ever sighed in relief at finding ambiguity about purity. Instead, folks often ask, “How close to pure is this stuff, really?” Technical staff won’t compromise with vague answers—either it’s 97%, 99%, 99.9%, or a mark higher. Every decimal point can tip a balance in pharmaceuticals, electronics, or food production. Lower purity may mean extra contaminants and unplanned reactions. That leads directly to wasted money, junked batches, and sometimes even dangerous results.
The cost difference between grades proves eye-opening. Electronic-grade chemicals fetch a higher price because microscopic contamination ruins sensitive circuitry. In a pharmaceutical grade, a minor impurity could impact someone’s health. There’s no fudge room once a batch fails, and I’ve seen entire productions put on hold as people double-check paperwork for a decimal error. Rather than viewing these numbers as mere red tape, anyone making real-world decisions pays close attention because legal, financial, and safety risks hide there.
Packaging options deserve just as much respect. Small bottles—sometimes as tiny as 50 grams—pop up in research labs hustling through early-stage trials. Larger bulk bags serve factories pumping out metric tons of product. Once, my team received a shipment in a badly sealed bag; half the content was ruined. That simple packaging mistake cost us days of work. Vacuum-sealed containers, light-resistant bottles, and safety-sealed drums all offer solutions for different storage and transport headaches. Glass handles volatile solvents better, but some chemicals corrode glass and need thick plastic.
These choices ripple down the supply chain. Stable packaging with tough seals keeps moisture, dust, and sunlight at bay. That’s critical in places with humidity or temperature swings, which most warehouses face even with climate controls. Smaller units cut down on waste for small-batch runs. On the flip side, large drums reduce handling costs for scale-ups, and less packaging means less to dispose of, which appeals to the sustainability-minded.
So what’s the takeaway for anyone making a buying call? Look at your process from start to finish. Check every threshold that matters, from the purity required for safe product quality, to the stress and spill risk during transit, to the suitability for handling in your facility. Consider your most likely headaches—ingredient spoilage, contamination, wasted packaging, or cross-contamination between runs—and choose accordingly.
If your demands fit a modest purity, don’t overpay for the highest grade. In highly regulated sectors, never gamble on lower grades for short-term savings. For packaging, never underestimate freight conditions or humidity in your storage. Each step upstream and downstream can affect both safety and your bottom line. Regulatory bodies keep tightening the screws, and with good reason—patients, consumers, and entire companies rely on these hidden, supposedly minor details. A quality-focused mindset about purity and packaging options still trumps all the tech trends and buzzwords the market rolls out.
People who work with chemicals know stories about spilled beakers or strange new burns that show up after a long shift. I remember late nights early in my career, trying to clean up a mystery powder that just wouldn’t settle. Safe handling isn't about playing scared—it's about working smart and keeping everyone at the table safe.
Every compound comes with its own set of problems. Skip the safety briefing, and you might end up with more than a mess. Before cracking the seal on any new bottle, pull up a current safety data sheet. Not all hazards are obvious. Just because a substance looks like sugar or salt, doesn't mean it's as forgiving. Some powders react the instant they catch a whiff of moisture in the air or heat from a hand. Respect isn’t fear—it’s common sense built on real-life consequences.
In the field, nothing replaces gloves that fit, a sturdy lab coat, goggles you won’t keep yanking off, and proper shoes. No open-toed sandals ever, no matter how hot it gets outside. Nitrile gloves block a lot of trouble, but some chemicals eat right through them. Every worker brings home different experiences about what failed under pressure or after a week of use, so check your gear for holes, cracks, and weakness. Your hands and eyes remember mistakes a long time.
Stacking containers, labeling jars, keeping incompatible things apart—these habits make for quiet days, not headlines about accidents. Set up workspaces with splash shields if your compound splatters or vaporizes. Have absorbent kits, neutralizers, and clean water close by. People love to improvise in a pinch, but accidents reward the prepared, not the creative. Quick thinking helps, but built-in systems matter more.
Compounds that start fires or give off fumes don’t belong with snacks, coffee mugs, or personal stuff. Tight lids, real chemical storage cabinets, and regular audits prevent a lot of close calls. Check expiration dates—all chemicals age, some faster than others. Many disasters start from a dusty container somebody pushed to the back and forgot.
Never pour leftovers down the drain or toss them in the regular trash. Local rules for disposal keep toxins out of water and off city streets. Ask your supervisor or safety officer the best way to get rid of used-up stock. I've seen more fines and headaches from improper disposal than from any spill inside the lab.
Big organizations often insist on ongoing safety courses, and for good reason. Familiarity can breed carelessness, and rules change as science uncovers new hazards. Even in small teams, share stories about near-misses and updates from regulators. The best ideas come from workers speaking up about what works and what’s failed in the real world. A culture where questions and double-checks are encouraged is one where fewer people end up in the ER.
Nobody likes red tape or overbearing policies, but experience says simple routines—reading labels, keeping stations organized, using good gear, sharing what goes wrong—make homes and workplaces safer for everyone. Investing in real training, not just check-the-box videos, pays off over the long haul. Geography, lab size, and workplace culture matter, but safe handling starts with respect and personal choice. That builds habits people carry home, into the next job, and even onto the next generation of workers.
Most folks don’t think much about how a jar of white powder sits on a shelf in a lab. For chemists and researchers, stable storage isn’t just a boring requirement—it’s a layer of trust in the work that follows. With 2,4-Dichloro-3-Cyano-5-Fluorobenzoic Acid, a compound used in fields like agrochemicals and pharmaceutical research, the need for stability carries a much bigger punch. Unpredictable storage lets unknown variables creep into everything from cancer research to crop protection trials.
Heat rarely plays nice with organic molecules. Just a few degrees above room temperature starts knocking stability around—sometimes rapidly. I remember one summer, a colleague stored a sensitive benzoic acid derivative by a sunlit window for convenience. What followed was a string of failed runs and scrambled calculations. Later, we traced the problem to degraded stock. These chemicals do much better in tightly controlled environments—think cool, dry, and far from fluctuating heat sources like radiators or direct sunlight. Refrigeration helps, but only if the container stays tightly sealed and condensation risks are handled.
Water vapor causes trouble that’s easy to miss. Even a small leak or a container left open can let in enough humidity to affect purity. Hydrolysis reactions, changes in physical appearance, or subtle pH shifts happen before you realize it. A good friend of mine switched storage cabinets because his old metal one seemed to sweat every time it rained. Within weeks, materials he relied on for synthesis started giving inconsistent results. Chemical desiccants and a humidity monitor in secure cabinets make all the difference.
Bright lab lights or sunshine may not seem like threats, but photodegradation ruins delicate bonds in seconds. Amber glass vials, low-light storage areas, or simply tucking containers in opaque boxes go a long way toward protecting sensitive chemicals. Every time someone leaves a clear bottle out, there’s a gamble—the spectrum of light, especially UV, can quietly start breaking things down.
Clean, airtight storage containers guard against both environmental invasion and cross-contamination. I’ve seen much too much cost and time wasted on experiments ruined by a splash of something that snuck in during hurried weighing sessions. PTFE-lined caps, glass vials, and gloves help. Labels matter, too—date everything, track batch numbers, rotate stock, so you’re not relying on memory alone.
It’s easy to ignore the storage details when deadlines press and resources thin out. Down the road, lost time and unreliable data always catch up. A recent review in the Journal of Chemical Health & Safety pointed to over half of laboratory problems tracing back to basic storage lapses—oxidation, moistures, or careless mislabeling.
Solid protocols pay for themselves: store cool and dry, minimize light exposure, seal well, and never skip the label. This isn’t about rules for their own sake. Reliable science can’t grow from unstable ingredients.