Curiosity tends to drive chemistry forward, and 2,4-diaminobutyric acid serves as a prime example. Researchers started recognizing its potential after diving into the intricate world of non-proteinogenic amino acids in the early twentieth century. These compounds caught attention for their presence in rare peptides and their utility in building blocks for more complex molecules. It might sound far removed from daily life, but the history connects with everything from antibiotics to structural biology. As proteomics advanced, so did the need for novel amino acids, and more chemists tried to synthesize and characterize this compound. By the late 1900s, with advances in solid-phase peptide synthesis, interest in 2,4-diaminobutyric acid picked up, linking ancient curiosity with modern biochemical demand.
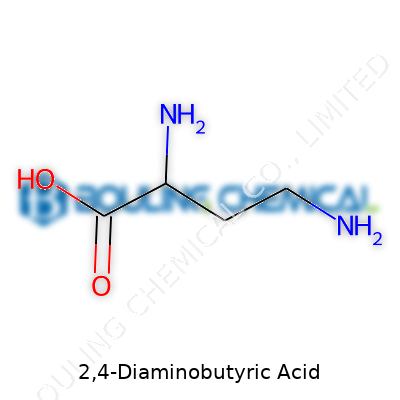
2,4-diaminobutyric acid takes its place among unusual amino acids, containing two amino groups and a carboxylic acid on a four-carbon backbone. In common terms, the molecule packs a punch because those extra amino sites open up new chemistry and novel biological activity. You’ll also spot it on labels as DAB or 2,4-DABA, and sometimes even with systematic nomenclature like 2,4-diaminobutanoic acid. The story of this molecule isn’t just about its structure—it’s also about applications stretching from pharmaceuticals to niche biochemical research.
This amino acid shows up as either a white crystalline powder or a compact solid, bearing the slight water solubility reminiscent of other short-chain amino acids. It melts at a temperature typically above 200°C, which speaks to its stability. In lab experience, this sort of thermal stability makes handling more forgiving—there’s less risk of decomposition during mild heating steps, which is handy for peptide coupling reactions. The molecule carries both alpha and gamma amino groups, which gives it a bit more flexibility in creating derivatives. Its pKa values reflect the basicity from both amines, influencing how it behaves in solution and how researchers construct peptides containing it.
Standards for materials like 2,4-diaminobutyric acid determine suitability for precise applications. Purity levels often reach above 98% for research purposes, with detailed certificates of analysis disclosing trace impurities or residual solvents. Chemical companies usually sell both the L- and D-enantiomers, as stereochemistry can dictate biological activity. Labels highlight physical properties, storage recommendations—think dry and cool to ward off decomposition by moisture or light—and sometimes, batch-to-batch variability. Any lab using this ingredient in medicinal chemistry or biochemistry takes such technical data seriously, as contamination can spoil experiments or skew analytical results.
Chemists usually synthesize 2,4-diaminobutyric acid from well-established organic pathways, starting with aspartic acid or other four-carbon precursors. Reductive amination and controlled hydrogenation often come up, all relying on careful protection and deprotection of the amino groups to keep reactions from turning messy. In my own bench work, the purification step takes real patience, since close relatives of this molecule notoriously co-elute during chromatography. Techniques like ion-exchange and crystallization do the heavy lifting. Labs that specialize in synthetic amino acids have been refining these protocols over decades, gradually improving yields and minimizing waste.
This compound’s real strength shines through in its chemical versatility. With two distinct amine groups, one can serve as a handle for coupling to other chemicals, while the remaining amine keeps the door open for further modification. This feature supports not just the construction of cyclic peptides but also branching architectures that aren’t possible with standard amino acids like lysine. Research teams often exploit this reactivity to design novel antibiotics, cross-linking agents, or enzyme inhibitors. The molecule also tolerates most standard protecting groups, which makes it a favorite in solid-phase peptide synthesis workflows. In practice, the number of functionalizations goes far beyond what’s listed in catalog entries—chances are, if you can envision a linker-based application in macrocycles or synthetic biology, it’s worth trying 2,4-diaminobutyric acid as a starting point.
Names like DAB, (±)-2,4-diaminobutyric acid, and even some trade codes surface across chemical catalogs. Such aliases usually stem from stereochemistry, but sometimes they just reflect different suppliers’ conventions. Some researchers document the pure enantiomers as L-2,4-diaminobutyric acid or D-2,4-diaminobutyric acid, and both see use in studies exploring structure-activity relationships. Before placing orders, double-checking the synonym and CAS number helps avoid mix-ups, especially since confusion with 2,3-diaminobutyric acid or strictly proteinogenic counterparts happens more often than it should.
Chemists treat even small molecules with respect, and 2,4-diaminobutyric acid falls into this category. Proper handling comes down to using gloves and working inside a fume hood, as dust or contact can lead to skin and respiratory irritation. Safety data sheets flag potential hazards, mostly centered on basic toxicity and environmental concerns linked to improper disposal. Labs train students and staff to avoid ingestion or inhalation and to store the powder away from acids, bases, and oxidizers. I’ve seen accidents arise from hasty weighing or careless spills, sending everyone scrambling for material safety sheets—so, safe habits save both nerves and reagents.
Those outside chemistry circles often overlook the breadth of 2,4-diaminobutyric acid applications. Its main claim to fame lands in antibiotic biosynthesis, especially as a building block in naturally occurring peptides with potent biological activity. Some research points to roles in enzyme inhibition, ion channel modulation, and structural studies of proteins with non-natural amino acids. Pharmaceutical scientists also lean on it to create analogues with altered charge states or new pharmacological properties. Peptide chemists value the molecule for branching structures or as a precursor for cross-linkers, and those studying bacterial resistance find its derivatives useful in dissecting mechanisms of action. Agricultural researchers, too, sometimes probe its potential in developing crop-protectant molecules, although hurdles remain before large-scale adoption.
Research involving 2,4-diaminobutyric acid moves fast, as both academic and industry sectors search for molecules that improve human health and agricultural yield. Synthetic biology now borrows heavily from this tiny amino acid, exploring novel biosynthetic pathways and protein engineering. Since it’s not coded for by DNA, insertion methods like solid-phase synthesis and tRNA engineering come into play, expanding the chemical universe beyond standard biology. In my own readings, structure-activity studies relying on this molecule offer hope for new generations of therapeutics, especially as antibiotic resistance turns into a global crisis. Funding agencies have started to notice, channeling support toward projects that create new peptide libraries or probe the immune system through molecular design.
No new molecule escapes scrutiny on the toxicity front. 2,4-diaminobutyric acid shows some cytotoxicity at higher concentrations, and particular concern arises in testing with mammalian cell lines. Researchers learned that its incorporation into foodstuffs or unapproved pharmaceuticals demands real caution. Regulatory bodies such as the FDA and ECHA expect thorough toxicological profiles before any application moves out of the research sphere. Chronic exposure studies remain limited but point to the need for further investigation, particularly given its use in the synthesis of antimicrobial peptides. Anecdotally, labs report headaches and skin irritation if exposure isn't controlled, echoing the need for sound ventilation and minimal direct handling.
Scientific progress never coasts, especially with molecules that offer unique scaffolds and reactivity like 2,4-diaminobutyric acid. Its track record in peptide discovery and therapeutic design suggests it’ll stay on the radar for synthetic chemists and biotechnologists. Advances in metabolic engineering and directed evolution promise more efficient ways to incorporate this amino acid into peptides and proteins, while tightening environmental and safety standards push for greener synthesis. As concerns about antibiotic resistance and precision medicine grow louder, demand for tools like rare amino acids grows with them. The journey ahead challenges researchers to harness its promise while keeping an eye on safety, sustainability, and real-world utility.
Some compounds in science stick around for decades without causing much excitement. 2,4-Diaminobutyric acid falls into that category—one of those overlooked molecules that’s been quietly sitting in textbooks, only catching a bit of attention in the labs of biochemists. Lately, though, it’s popped up in enough headlines to spark curiosity. What’s all the fuss, and why should we care about this four-carbon amino acid most people have never heard of?
For folks without a degree in chemistry, amino acids make up the building blocks of proteins. Most of us are familiar with the basic twenty. 2,4-Diaminobutyric acid isn’t one of them. It’s a nonproteinogenic amino acid, which just means you don’t find it in the main structure of human proteins. That doesn’t mean it serves no purpose—quite the opposite. Microbes, plants, and sometimes pharmaceuticals have found creative uses for it.
Researchers at universities and pharmaceutical companies study this molecule because it shows up in certain antibiotics. Take the example of polyamines, a group of molecules with a reputation for turning bacteria’s insides into confetti. 2,4-Diaminobutyric acid plays a role in that—it fits like a tiny wrench in bacterial machinery. Some antibiotics, such as those used for Gram-negative bacterial infections, owe part of their chemical punch to this amino acid.
Colistin, one of the last-resort antibiotics to fight drug-resistant bugs, contains something called DAB (the shorthand for 2,4-Diaminobutyric acid). Ask a microbiologist about colistin, and you’ll get a tight-lipped answer about its importance in hospitals, especially when nothing else works. When scientists tinker with DAB in a lab, they’re aiming to design new antibiotics with fewer side effects than the old stuff. This tinkering may lead to drugs that save lives without wrecking kidneys or nerves, a problem that’s dogged older treatments.
There’s more. Some scientists use this molecule to probe enzymes and metabolic pathways. By adding DAB where another amino acid would normally go, researchers can see what happens when a system gets thrown off balance. It helps pinpoint which parts of a protein are absolutely crucial, or which metabolic routes might be hijacked by disease—knowledge that sometimes leads to breakthroughs in cancer research or metabolic disorders.
The reach of 2,4-Diaminobutyric acid doesn’t stop in medicine. Some studies in the last few years use it to inhibit viral replication. Chemists synthesize endless variants based on its structure to see what’ll trip up a virus’s life cycle. And in a totally different sphere, some fermentation processes have put it to use, especially in shaping the microbial cocktails that produce enzymes for the food and beverage industry.
If there’s one lesson from my years around research labs, it’s this: molecules that seem small or obscure often steer big discoveries. That said, nobody should conflate lab results with medical miracles. Not every breakthrough molecule finds its way to a pharmacy shelf, and not every use carries the same risks or benefits. The rise of antibiotic resistance shows how tools like DAB play a delicate role in public health. We need strict stewardship and regulation before new derivatives hit mainstream use.
2,4-Diaminobutyric acid won’t show up in supermarket tabloids. Anyone concerned about the future of antibiotics, enzyme research, or even large-scale fermentation should keep an eye out for names like this, quietly doing the work behind the scenes of headlines.
Food and chemical names rarely ring any bells for most people. Often, we trust that if an ingredient has made its way onto a label, someone, somewhere, checked it for safety. Hearing about 2,4-Diaminobutyric Acid, you might imagine something out of a laboratory rather than your kitchen pantry. The real question people ask is whether it belongs anywhere near dinner plates or supplements.
Looking at the science, 2,4-Diaminobutyric Acid isn’t a common household name, but it pops up in certain contexts. It's related to other basic amino acids that act as protein building blocks. Unlike those, it doesn’t appear in large quantities in the usual foods. Scientists have dived into its chemical properties, as it shows up during fermentation or might get used in specialized industrial processes. Still, that doesn’t automatically translate to “safe.”
Regulatory agencies like the Food and Drug Administration and the European Food Safety Authority take a strict view on new food ingredients. If safety data haven’t been gathered from studies on how the body handles it, the substance generally lands on the “needs more research” shelf. Coming across this specific compound in peer-reviewed food safety studies, there isn’t much evidence that anyone has cleared it for routine human consumption.
From experience in food science communication, even substances that look similar on paper can act very differently in the body. Amino acids are essential, yes, but small changes to their structure lead to unexpected effects. An extra atom here or there throws off metabolism. Toxicology takes these tweaks seriously; just because something resembles a harmless nutrient doesn’t grant it a free pass.
Reports have surfaced about 2,4-Diaminobutyric Acid causing concern after being detected in contaminated food sources or mistaken for better-known amino acids. In animal models, high doses led to neurological symptoms. No routine dietary use and no solid, long-term studies in people means health risks have not been ruled out.
Shoppers deserve open, honest labeling — not just familiar-sounding or “generally recognized as safe” claims. Consumers wrestle with more chemical names than ever before, and clarity on labels makes a huge difference. If a food product or supplement contains something unusual, manufacturers should spell out what exactly it is, how it’s been tested, and why it belongs in the product.
Government watchdogs set standards for ingredient review. The process can move slowly, but the burden of proof belongs on those introducing a new chemical into products people eat or drink. Until real, peer-reviewed studies confirm safety, skepticism is the wise path. Jumping on wellness trends with little-known ingredients risks making people test subjects for side effects.
Insisting on clear, third-party tested safety assessments pushes the industry in the right direction. Public funding for basic research also matters, so the gap between what’s technically possible and what’s genuinely safe doesn’t keep growing. Training for nutrition professionals on how to interpret “chemical-sounding” food labels helps keep both experts and the public on the same page.
Trust builds on transparency, evidence before marketing, and honest conversations about health risks. Until the safety profile for 2,4-Diaminobutyric Acid is crystal clear, cautious curiosity works better than blind faith.
Life boils down to the small stuff. Take 2,4-diaminobutyric acid—one of those molecules that doesn’t spark big headlines but quietly earns its place in biochemistry. This amino acid, not as storied as lysine or alanine, carries a simple carbon backbone twisted with two amine groups. The chemical structure: NH2-CH2-CH(NH2)-COOH. You get a four-carbon chain, with an amine on the second and fourth carbons, plus the classic amino acid carboxyl at the end.
I still remember holding a physical model in the lab, the colored balls and sticks showing those two amino groups sticking out. Students crowded around, a little bemused that such minor rearrangement of atoms could change the way a molecule interacts with the body. With 2,4-diaminobutyric acid, the extra amine broadens its chemical potential—nature’s small changes opening doors for new interactions or synthetic experimentation.
You don’t see 2,4-diaminobutyric acid popping up everywhere in nature. That slight quirk in its carbon skeleton keeps it rare in living systems, but researchers pay attention. The two amines along the backbone let this molecule form strong hydrogen bonds and stack up as a handy building block in synthetic peptides or as a raw material for certain antibiotics. Chemists recognize that swapping the position of those amino groups means the difference between a mild metabolite or a potent pharmaceutical precursor. The most infamous analog is 2,4-diaminobutyric acid’s role in the antibiotic viomycin—one of those molecules that keeps tuberculosis in check.
From a structural perspective, the molecule steps up acidity and basicity. Two amines mean more potential sites for protonation. In many labs, folks exploit that property for attaching chemical tags or building new molecules that can slip past bacterial defenses. I’ve watched colleagues run careful chromatography and mass spectrometry, hoping to catch these minor chemicals before they break down. The unique chemical handshake of 2,4-diaminobutyric acid underpins that search.
There’s another upside: creativity. With its simple four-carbon chain, this amino acid stands as a foundation to build on—engineers prod at its structure, thinking up new pharmaceuticals. Drug designers think about swapping functional groups on the backbone, twisting the molecule to block antibiotic resistance or channel its reactivity.
Researchers spend real hours figuring out how to harness these niche amino acids. Manipulating 2,4-diaminobutyric acid’s scaffolding can lead to more robust drugs or quick ways to diagnose rare diseases. The way the amine groups play on the carbon chain changes the fate of a molecule in blood or tissue. For students and scientists, tracing the path of each atom helps forecast chemical behavior, and with that, new therapies can be built from the ground up.
Studying less-common amino acids trains us to think beyond the well-paved roads. The story of 2,4-diaminobutyric acid isn't about an unusual chemical—it’s about what happens when you ask how tiny changes to a backbone unlock new chemistry. More minds digging into these structures could reveal novel ways to defeat antibiotic resistance, develop new diagnostic tools, or understand rare metabolic conditions. Good science comes from this mix of curiosity and precision, tracing how each molecule fits into life’s larger patterns.
Some chemicals command more caution than others. 2,4-Diaminobutyric acid is one of those compounds that, if left unmanaged, could introduce real headaches for labs and manufacturers. This small molecule often crops up in research settings or specialty chemical projects, but poor storage often invites trouble ranging from product spoilage to safety risks. Anyone who has worked with sensitive reagents knows stories—powder clumping, decomposition, odd smells that point to chemical breakdown. Skipping proper storage rarely ends well.
Chemists and lab managers know the value of a pure, stable chemical stock. 2,4-Diaminobutyric acid, with its amine groups and reactive nature, doesn’t thrive out in the open. Air, especially moisture, quickly leads to degradation. I’ve seen more than one batch of specialty reagent turn yellow or tacky after just a few days in a warm, humid room. If the goal involves reliable data or consistent formulation, exposing sensitive materials never just affects the current experiment—it compounds into next week’s problems too.
Regulatory guidelines and scientific reviews back up this experience. Standard chemistry resource books note that organic acids often drop in purity if they pick up water or react with carbon dioxide, which isn’t uncommon in labs where airflow and temperature aren’t controlled tightly. With 2,4-Diaminobutyric acid, contamination ruins everything from analytical accuracy to downstream synthesis. Not only does this delay work—it costs money, a lot more than a decent desiccator or fridge ever would.
Smart storage means controlling air, light, and heat. Researchers almost always recommend keeping 2,4-Diaminobutyric acid in tightly sealed containers, away from moisture and direct sunlight. Most chemical suppliers use amber bottles for a reason: light exposure sparks unpredictable changes. Storing the compound in a cool, dry cabinet or, for longer-term projects, in a refrigerator, preserves its character. If the material sits with other chemicals, cross-contamination sometimes happens, so separate containers and clean scooping tools cut down on risk.
Labeling sounds basic but can save real trouble down the line. Every researcher knows the embarrassment and potential danger of grabbing an unlabeled or ambiguously-marked bottle. Dating each batch right at receipt and after first opening gives a trail to follow if anything begins to look or smell wrong.
Taking these steps often feels routine, but experience says it’s everything. My time in academic and industrial labs always circles back to the same truth: reliable results start with materials in predictable condition. Bad storage invites wild results and costly do-overs. Good practice not only keeps people safe, but also shields expensive research time from being wasted by surprises in the bottle.
So, anyone handling 2,4-Diaminobutyric acid should treat storage as serious as the science itself. Double-check those caps, use dedicated containers, and keep it cool and dry. Little changes make the difference between smooth work and scrambling to fix preventable problems.
Some say chemistry is full of details that don’t make a difference. They haven’t worked in a real lab. I’ve spent years working on synthesis benches and troubleshooting biological experiments. Tiny changes in purity turn what should be a straightforward process into hours lost and budget down the drain. When you see “2,4-Diaminobutyric Acid” on your bench, the first question isn’t if it looks right, but if it’s pure enough. There’s a reason — it only takes a little contamination to throw off sensitive research or mess up downstream chemistry.
Most researchers who reach for 2,4-Diaminobutyric Acid do so for peptide synthesis, metabolic studies, or as a building block for more complex molecules. In these contexts, impurities aren’t just a minor inconvenience. A batch with only moderate purity can introduce unknown variables into reaction outcomes or cell culture results. During my graduate work, I learned the hard way that even traces of similar amino compounds can block a reaction or confuse biological assays. False positives, mis-labeled peaks in chromatography — headaches that could’ve been avoided by opting for the purest sample available.
Suppliers often claim purities above 98 percent for lab-ready 2,4-Diaminobutyric Acid. Most analytical standards push above that, closer to 99 percent or even higher. The last percent isn't just numbers. That small gap hides everything from inorganic salts to leftovers from synthesis. These aren’t just invisible—they can push a reaction out of balance or introduce misleading signals when running tests.
I’ve seen teams burn weeks chasing odd results, only to realize it stemmed from a slightly impure batch. In molecular biology or pharmaceutical research, accuracy defines progress. With biomedical patents and peer review at stake, even one mysterious impurity can turn an easy publication into a pile of troubleshooting reports.
It’s not just about pride — it’s about money, trust, and science. Funding agencies and journal reviewers increasingly want to see hard data on reagent purity. Miss that bar and you’ll repeat experiments, lose confidence, or worse, throw out an entire batch of data. That’s not an academic problem; that's a real-world setback.
I’ve learned to never take a supplier’s purity at face value. Always request documentation or batch-specific certificates of analysis. Use techniques like HPLC or NMR to check samples, especially for critical experiments. If your research depends on it, ask for ultra-pure grades—even if it costs a bit more. The extra spent upfront usually pays off in reproducible results and fewer headaches down the road.
Some labs now include routine purity checks as part of their quality control process. Others pool funds to buy higher-grade precursors and share them across research teams. Simple steps, but they save effort, money, and scientific reputation. Pure reagents make reliable science, and in my experience, that’s a shortcut worth taking every time.