The story of 2,4,5-Trifluorobenzoic acid lies buried in the wider arc of organic chemistry and the relentless exploration of aromatic substitution. Fluorinated aromatics started making waves in laboratories during the mid-20th century, a time marked by the synthetic race for new materials and pharmaceuticals. Chemists recognized that once you slot fluorine atoms into a benzene ring, you don’t just tweak the molecule—you transform its identity. The methods that unlocked multi-fluorinated aromatics like 2,4,5-Trifluorobenzoic acid set new standards for both selectivity and durability. Amid Cold War efforts to build strong, unreactive materials and more precise drug scaffolds, research teams built the chemistry that let this compound step out of the shadows as more than just a niche intermediate.
2,4,5-Trifluorobenzoic acid sits in a crucial position among trifluorinated aromatics. Its framework opens avenues in agrochemicals, pharmaceuticals, and performance materials, and the arrangement of fluorines turns it into a launching point for dense molecular engineering. Real chemistry nerds find it a gift: it’s the sort of molecule that can act both as a target in reactivity studies and as a piece in broader synthetic puzzles. Over the years, it’s shown up in patent filings and research reports tied to herbicides, small-molecule drugs, and even liquid crystals, underscoring how a simple benzoic acid skeleton can punch above its weight once armed with a strategic set of fluorines.
This compound carries all the quirks of a carboxylic acid, but the three fluorines nod to a tendency for lower reactivity at usual positions. Its melting point and solubility highlight the balance between the polar acid group and the hydrophobic, fluoro-rich ring. Chemists who run columns with it learn fast that it won’t play by the same clean rules as its mono-fluorinated cousins; expect strong hydrogen bonding, tamed reactivity, and packing behavior in crystals that feels almost engineered. The electron-withdrawing effect of each fluorine does more than just drop the pKa; it also tightens up the ring, making some transformations distinctly sluggish. Compare that to unsubstituted benzoic acid, which sits wide open to attack—a world apart from the steric and electronic fortress put up by three fluorines at just the right places.
In laboratories and industry, purity standards for 2,4,5-Trifluorobenzoic acid don’t just float—they steer research and process safety. This compound shows sensitivity to handling conditions due to its tendency to form strong, tight crystals and the volatility possible with trifluorinated aromatics. Accurate identification comes from high-resolution spectroscopy—NMR and IR—since fluorine splitting patterns leave unmistakable fingerprints. True to modern chemical commerce, batch labeling needs to address batch number, synthesis data, and purity, built around keeping research reproducible and regulatory headaches at bay. I’ve seen teams switch suppliers after a shift in impurity profile or a UV trace that didn’t match; a reminder that specification isn’t paperwork—it’s the difference between an experiment working or stalling.
Traditional synthetic strategies revolve around the halogen dance—starting with the right fluoro-substituted precursors, then running controlled hydrolysis of corresponding trifluorobenzonitriles or esters. Some chemists push for catalytic fluorination, though the risk of over-fluorination chases many back to defined halogen exchange or selective substitution. Strong bases and robust temperature control become non-negotiable. No one who’s run a bench-scale prep soon forgets how a slight temperature overshoot can skew your mixture. For those scaling up, solvent choice becomes the battlefield between solubility, selectivity, and clean-up ease. Many advances in flow chemistry and continuous processing trace their roots to the challenges of purifying tricky aromatic acids like this.
As a trifluorinated aromatic acid, 2,4,5-Trifluorobenzoic acid stands out for its reluctant chemistry. Electrophilic aromatic substitution won’t get far with the ring so electron starved; the action shifts toward nucleophilic routes and metal-catalyzed cross-couplings. Transformations often aim to protect, reduce, or swap the acid group, or to displace fluorines under exacting conditions that won’t torch the whole molecule. Suzuki and Heck reactions can build larger structures from it, provided catalysts and ligands play along with all the steric baggage. Creative chemists find joy in tackling such an unforgiving platform, pushing palladium and copper reagents to their limits. You respect how a single acid can serve as a scaffold for a spectrum of high-value molecules, even if it asks for patience and technical smarts.
Those navigating literature or chemical catalogs will bump into a series of aliases—2,4,5-trifluorobenzoic acid, benzene-2,4,5-trifluoroic acid, or simply "TFBA" in shorthand. The systematic name spells out the substitution pattern, which proves critical for chemists shopping for exactly the derivative they want, since tiny differences in fluoro placement birth completely different reactivity profiles.
Handling fluorinated aromatics like this one draws a line between the casual benchworker and those who dig into safety protocols. Gloves and goggles feel obvious, but the real safety gear comes down to process—running everything in well-ventilated hoods, keeping containers tightly sealed, and respecting the volatility and possible acid fumes. Chronic exposure studies have called for caution, even though the acid itself isn’t as notorious as polyfluorinated perfluorooctanoic acid cousins. Disposal gets regulated, with spent solutions destined for specialized chemical waste channels. Technical teams savvy to green chemistry look for methods that ease cleanup and lower emissions, aligning bench practices with both workforce safety and environmental sanity.
The reach of 2,4,5-Trifluorobenzoic acid spreads across several sectors, each exploiting different features of its robust aromatic ring and tailored acidity. Crop science favors these molecules for building blocks in selective herbicides—here, the fluorines shield the core from premature metabolism, keeping actives around longer in tough field conditions. Pharmaceutical researchers, always chasing both stability and bioavailability, see trifluorinated acids as durable scaffolds, loading on side chains for activity tuning. Electronics and materials engineers borrow from that same playbook, leveraging the hydrophobic and electron-poor ring to create films, coatings, and polymers with both thermal and chemical inertness. I’ve seen startups try to thread it into OLED research, betting on unique optoelectronic properties, while others take it into battery additives as stability enhancers.
Research on trifluorinated aromatics rewards persistence and a bit of stubbornness. You see researchers devising clever new routes to lower activation energy, hunting for greener solvents, and trying to scale reactions economically—all with the hope of keeping cost and waste in check. The hunt for new drugs draws heavily on the stability of these acids, looking for analogs that skip unwanted metabolism or boost target binding through the right fluoro pattern. Green chemistry advocates press for new catalytic cycles and milder reagents, in part to cut reliance on hazardous halogen sources and slash downstream purification workloads. Fast-moving teams collaborate across continents, sharing NMR traces and yield data by email, all driven by the prospect of cracking a method that unlocks larger, better-behaved molecules from this base.
Every new round of applications demands answers about toxicity and environmental fate. While 2,4,5-Trifluorobenzoic acid doesn’t carry the shadow of long-chain perfluorinated chemicals, researchers put time into cell assays and environmental fate studies. Early work shows it resists microbial breakdown, hinting at persistence that warrants thoughtful lifecycle planning. Researchers keep a close eye on bioaccumulation, since even short-chain fluorinated aromatics can surprise with how they move through soil and water. Animal studies and cytotoxicity assays help spell out limits for both workplace exposure and release scenarios, feeding data to both industrial hygiene protocols and regulatory reviews. These safeguards keep progress from unraveling into unintended chemical footprints.
Looking forward, the trajectory of 2,4,5-Trifluorobenzoic acid holds promise and challenge in equal measure. For every upside—a new drug class, a tougher agricultural coating, an advanced polymer—there’s a call for more sustainable routes, cleaner synthesis, and sharper understandings of environmental fate. The ingenuity of chemists has always sculpted new roles for such specialty molecules. Funding shifts steadily toward routes with less waste and more renewable inputs. Industry partnerships are fueling pilot plants trialing enzymatic routes or flow reactors with tighter process controls. Consumers and regulators push hard for transparency about both benefits and risks. The molecule stands as a testament to how focused substitution on a benzoic acid ring can unlock utility across disciplines, provided science, safety, and stewardship move forward together.
2,4,5-Trifluorobenzoic acid hardly rolls off the tongue, and most people probably won’t cross paths with it in everyday life. In my experience, conversations about this compound only come up in the world of industrial chemistry. Yet, its importance shows up again and again, especially for those working with pharmaceuticals and advanced materials.
This compound, just by its structure, already hints at some real chemistry muscle. Trifluorination of an aromatic ring isn’t just about showing off – it’s about building molecules that do things regular benzoic acid can’t. The fluorine atoms make the molecule more resistant to breakdown. This stability means companies often use it as a precursor for substances that need to last – think active drug ingredients and polymer additives designed to stay put under pressure.
Pharmaceutical companies look at 2,4,5-trifluorobenzoic acid and see a toolbox. The trifluorinated motif is popular because fluorination can improve how a drug is absorbed or metabolized. Medicinal chemists will often swap in a trifluorobenzoic acid segment to adjust a molecule’s polarity or to slow down how quickly the body breaks a medicine down. I’ve watched research teams spend years refining the right substitution to eke out a better therapeutic effect, and compounds like this are part of that process. More stable molecules mean longer shelf life and, sometimes, medicine that works better for patients. One published study found certain anti-inflammatory agents increased their action in vivo after being tweaked with trifluorinated rings, tying a piece of research straight back to the utility of chemicals like this.
Organic synthesis labs don’t limit this acid to pharmaceuticals. Materials scientists borrow it for its electronic properties, using it to help design new specialty polymers. Electronic gadgets, solar panels, and sensors all benefit from parts that won’t corrode or fall apart after months of use. Trifluorobenzoic acid’s three strategically placed fluorines help engineers control properties like melting point, durability, and conductivity in the finished product. As someone who’s handled bench-scale reactions, I’ve appreciated how predictably this compound reacts, making it easier to control outcomes in a lab setting.
The conversation about any trifluorinated aromatics would be incomplete without mentioning environmental impact. Researchers are still sorting out what happens when these substances enter water or soil. I’ve noticed rising concern across conferences and publications about how persistent these molecules can be once released. Chemists are keeping a close eye on the so-called “forever chemicals” issue, and trifluorinated compounds often fall under that umbrella.
It falls to researchers, policymakers, and manufacturers to approach these substances responsibly. The chemical’s promising applications need balance against a responsibility to minimize spills and find greener ways to make and break down these molecules. Cleaner fluorination methods, stricter spill controls, and better recycling programs are already on researchers’ minds – and will shape how the industry handles trifluorinated chemicals in the future.
2,4,5-Trifluorobenzoic acid reminds us that innovation brings challenges and opportunities in equal measure. Whether shaping the next generation of medicines or toughening up solar panel materials, this compound occupies a crucial spot in the modern chemist’s arsenal. As the industry keeps evolving, responsible use and careful innovation will be what determine its legacy.
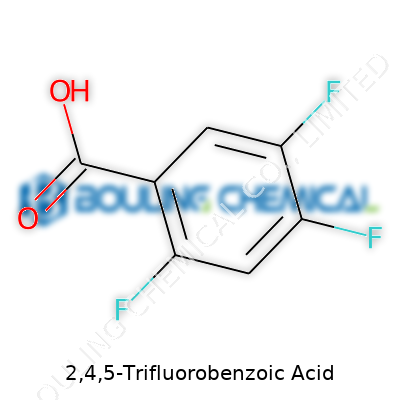
Small tweaks in chemistry can set the stage for big discoveries. Take 2,4,5-Trifluorobenzoic acid, for example—a molecule that looks humble at first glance. Its molecular formula, C7H3F3O2, hides a lot of potential. One benzene ring, three fluorines, a carboxylic acid group—each piece plays its part. If you know your high school chemistry, you might recall that fluorine atoms crank up the reactivity and can shape a molecule's behavior in both biology and industry.
Numbers matter. Count everything up—seven carbons (12.01 each), three hydrogens (1.01 each), three fluorines (around 19 each), and two oxygens (16 each). Punch that into a calculator, and you get a molar mass of about 176.09 g/mol. That’s the sort of number that researchers memorize and use without thinking. Walk through any organic chemistry lab and you’ll hear students call out these values as easily as they’d order coffee.
The three fluorines don’t just sit pretty on the ring. They shape the acid’s whole personality. Put fluorines on the benzene at 2, 4, and 5, and you launch the molecule into a new category. The acidity spikes. The electrochemical properties shift. Structure matters—a truism you see play out in pharmaceutical science and materials design. In my research days, testing related benzoic acids against various enzymes always showed just how different one tweak could make things. Toss on a fluorine or three, and suddenly you might have a hit for a new drug or pesticide.
Few people outside chemistry circles think about molecules like this, but they turn up in places that affect everyone. The pharmaceutical industry uses fluorinated compounds all the time—fluorines help drugs last longer in the body and bind more specifically to their targets. Agrochemical research draws similar lessons. Change the pattern of fluorination, and you can shift activity, reduce toxicity, and manage environmental impact. The trifluorinated variants often pop up in patent filings and research articles. This remains true for synthesis enthusiasts, where each substitution pattern offers a new puzzle.
Getting three fluorines on a benzene ring in just the right spots isn’t always straightforward. Labs turn to directed ortho metalation, clever use of protecting groups, or selective electrophilic fluorination. I remember the time our student group tried swapping halogens on a benzoic acid backbone—it took a few late nights and several adjustments to get clean conversion, but the results were worth it. The greener the method, the better. Academic labs now experiment with milder fluorinating agents to cut down on waste and risk. Chemistry as a discipline steps forward each year as more sustainable and cost-effective approaches emerge.
2,4,5-Trifluorobenzoic acid’s molecular formula and molar mass aren’t just facts to write on a chalkboard. They’re keys to unlocking behavior in the lab and in the world beyond it. Paying attention to small details, such as where a fluorine sits or how many get added, can shape outcomes in medicine, agriculture, and material science. Data and application travel together—always will. The new generation of chemists needs to keep asking how these tweaks can solve real-world problems and improve lives. For anyone curious about why these “little” differences matter, just look to the molecules shaping big discoveries every single day.
Any chemist who's spent time in a lab knows you can't cut corners when it comes to storage, especially with organic acids like 2,4,5-Trifluorobenzoic Acid. This compound pops up in research for its role in pharma development and as a stepping stone to more complex molecules. That makes the question of how you keep it not just technical, but absolutely practical.
In the lab, I’ve watched what happens when storage slips from the list of priorities. You come back to find crystals clumped, strange smells in the air, or compromised purity that throws off your data. With trifluorinated compounds, you’re dealing with high chemical stability, but you still have to respect the basics: moisture and light do them no favors, and so does excessive heat.
For a solid, dry, and crystalline powder like 2,4,5-Trifluorobenzoic Acid, the gold standard starts with airtight glass containers. Plastic doesn’t always cut it, since some organics interact with the surface or leach over time. Keeping the jar tightly sealed avoids contact with air and atmospheric moisture. Excess water in the container doesn’t just affect the acid itself, but also makes it harder to weigh and manipulate accurately in the lab. I’ve seen theoretically "dry" samples turn into sticky messes after just a few days out in humid air – and that’s a mess no one wants to clean up.
Cool, dry, and dark: this mantra goes far. Room temperature usually works, but avoid storing bottles near heating vents, on sunny windowsills, or close to anything that cycles hot and cold. Temperature swings can accelerate decomposition. Some labs have purpose-built chemical refrigerators for particularly sensitive compounds, but for this acid, a steady, moderate temperature achieves the right balance. I keep mine on a designated shelf, away from volatile bases or strong acids, since accidental spills or fumes could start unwanted reactions. Segregating chemicals by compatibility isn’t bureaucratic fuss – it’s saving your sanity long-term.
Labeling matters. I’ve pulled down too many mismarked bottles in shared labs, only to find someone has scribbled "TFBA" in sharpie and called it a day. Clear, legible labels with full names, concentrations, and hazard warnings help everyone working in the space make good choices. If you have to ask what’s in the jar, you’re taking unnecessary risks, no matter how much experience you’ve got under your belt.
For disposal or accidental spills, talk to trained safety staff. Trifluorinated acids require specific protocols; normal waste streams won’t do. Don’t pour them down the drain or toss them in regular trash. Local environmental laws guide how to dispose of organofluorine compounds because of their persistence in the environment.
Good storage protects your results and your health. Small habits—sealing bottles, monitoring the climate, labeling correctly—add up to a safer, more reliable lab environment. Storage isn’t just about keeping chemicals from going bad. It’s about respecting the work and everyone who’ll use these reagents after you. By treating each vial with mindfulness, we all make scientific progress possible—not just for ourselves, but for the next person who opens that door.
Anyone who has spent time near a research lab knows chemicals come with their own set of risks. Some demand little attention beyond basic precautions, but some need real care and respect. 2,4,5-Trifluorobenzoic acid doesn’t leap out of the catalog as a notorious hazard, but easygoing attitudes toward lab compounds can bite back fast if safety questions get ignored.
This compound often finds itself as a building block in the world of organic synthesis and pharmaceutical research. It helps chemists stitch together molecules needed for drug design, agrochemical research, and polymer work. People handling it aren’t always production workers in hard hats—it’s just as likely to turn up in a teaching lab or a custom synthesis order.
The structure of 2,4,5-Trifluorobenzoic acid includes a benzene ring with fluorine atoms and a carboxylic acid group. Many in the benzoic acid family are low-to-moderate irritants. While this compound doesn’t pack the punch of industrial acids or old-school pesticides, repeated exposure brings its own set of problems. Anecdotally and from manufacturer data sheets, these compounds can irritate the skin, eyes, and respiratory tract. Accidentally inhaling the dust or letting it sit on the skin for long periods may cause those familiar itchy eyes or coughing fits people in the lab have felt at some point.
Fluorine-containing chemicals can be sneaky. Even though this acid isn’t as reactive as others, the presence of fluorine groups nudges chemists to think twice. Perfluorinated substances get a lot of press for persistence in the environment—the so-called “forever chemicals”—but trifluorinated benzoic acids aren’t quite in that notorious league. They don’t break down quickly, so careless disposal with regular laboratory waste might start to add up over time. Here’s where responsible chemical stewardship matters.
Having worked in academic labs, I’ve watched new students learn fast that “not outright dangerous” doesn’t mean “harmless.” Gloves, eye protection, and fume hoods are the daily uniform for a reason. Small spills tend to bring out complacency, but cleaning up quickly and using ventilation stops irritation before it gets worse.
One near-miss with a tipped flask of a similar benzoic acid years ago left several of us coughing, eyes stinging from a cloud of dust. It’s easy to scoff at compounds that lack skull-and-crossbones warnings, but every seasoned researcher knows: the ones you ignore are the ones that end up teaching the hardest lessons.
Regulators in the US and Europe do not flag this compound as an acute toxin, carcinogen, or environmental disaster, so people sometimes brush aside the need for a second look. But good habits last a career. Always avoiding skin contact, using a fume hood, labeling containers clearly, and treating waste as chemical waste—including leftovers and paper towels—builds a margin of safety into lab operations. Keeping a clean workspace and washing up after handling chemicals make a big difference over the long haul.
For readers who manage chemicals for a living or just do the ordering, it never hurts to pull up an SDS before new experiments. If a new version gets published, it’s worth scanning through for updated toxicity or environmental guidance. Training and common sense go much further than any label warning in keeping bad outcomes off the news—and out of your own workplace stories.
Many platforms promise just about any material you can imagine, from rare enzyme substrates to obscure organics. Even so, buying something like 2,4,5-Trifluorobenzoic Acid doesn’t work the way online shopping usually does. This isn’t a set of headphones; chemicals like these travel a different road, with more rules, more gatekeepers, and a lot less convenience than most buyers expect.
In my years around labs and university storerooms, orders for specialty chemicals always began with paperwork. There’s a good reason. Chemicals containing fluorine, and benzoic acid derivatives generally, raise red flags within regulations. No trusted supplier wants to risk fines, or worse, by shipping to the wrong place or person. For 2,4,5-Trifluorobenzoic Acid, legitimate vendors usually request buyer identification, the purpose for which the compound will be used, and sometimes proof that you have proper facilities. Licenses or permits might come into play in certain jurisdictions, especially if the destination is outside the US or EU.
Online chemical catalogs aren’t like regular e-commerce storefronts. Outfits like Sigma-Aldrich and TCI America ask buyers to set up verified accounts. These accounts link back to businesses or institutions because street addresses, not post office boxes, matter. Students and researchers find that purchase requests route through procurement offices by default. Hobbyists run into locked doors, for reasons rooted in both safety and accountability.
Phone calls from suppliers are still fairly common after big orders or rare items. I’ve watched colleagues field questions about their intended experiments and lab safety standards. Once trust is established, future purchases tend to move faster, but nobody skips this first layer of due diligence.
Quality turns into a serious conversation with specialized molecules. Reagent and analytical grades matter because a contaminated batch can sabotage months of work. Chemists pay more for chemical purity backed by certificates, and companies usually guarantee analysis with every shipment. There’s peace of mind in seeing actual spectrograms attached to your receipt instead of ambiguous grade designations.
Some buyers try to sniff out deals from middlemen or e-auctions, especially overseas. I’ve seen purchases melt into frustration this way — chemicals stuck in customs for months, disposal headaches after opening canisters with unidentified residue, or outright scams. The risk just doesn’t match the savings, especially for labs or startups with limited budgets. Direct from authorized distributors or certified resellers usually ends up cheaper and safer, even after factoring in compliance costs.
Every chemical shipment triggers more paperwork. Couriers trained in hazardous material protocols handle this kind of delivery. Big cities see more frequent runs, but smaller labs outside urban areas might wait longer. Customs holds can tangle even the most routine purchase, especially after new regulations come online. I tell newcomers to plan for delays, especially if they’re ordering in the spring for summer research.
Plenty of researchers hope to see streamlined vendor portals and harmonized safety requirements. Some countries now offer digital licenses or single-window clearance systems; in practice, progress feels piecemeal. Vendor partnerships and transparent guidelines would help scientists avoid black-market temptations and help keep everyone safer.