The story of 2,3,5-Trimethylphenol, a trimethylated phenol, stretches back to the drive for better chemical building blocks during the mid-20th century. Before sophisticated separation technologies, researchers found the process for isolating individual methylphenol isomers both draining and expensive. Advances in organic synthesis opened up routes to access these precise positional isomers, responding to the growing need for fine-tuned antioxidants, specialty resins, and tailored intermediates. Early references in academic literature tied 2,3,5-trimethylphenol to phenolic resin research and to the search for benzene ring derivatives with better performance in heat or oxidative environments. Given the increasing scale of industrial chemistry post-World War II, the compound’s commercial story rose alongside the expansion of petrochemical refineries and specialty chemical manufacturers.
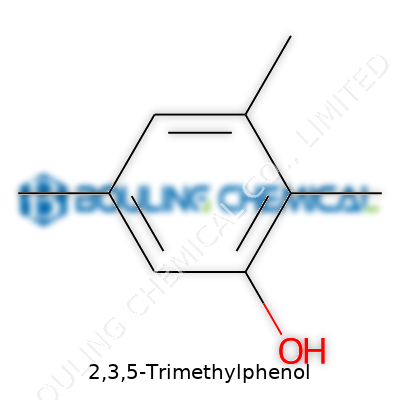
At its core, 2,3,5-Trimethylphenol looks unremarkable: a benzene ring hung with a hydroxy group, and methyl tails at the 2, 3, and 5 positions. But the smallest tweaks to molecular structure often open surprising doors. By adding three methyl groups to phenol, chemists modify everything from boiling point to reactivity. Products like this don’t demand attention in headlines the way flashy pharmaceuticals might, but their roles in glue lines, fuel additives, and specialty coatings drive the performance of the consumer products we see every day. Under names like Mesitol or 2,3,5-trimethylhydroxybenzene, this molecule often sits at the core of larger blends or helps fine-tune performance in complex chemical reaction networks.
One thing about 2,3,5-Trimethylphenol stands out beneath the microscope and in the lab notebook: bright white crystals with a sharp, medicinal odor. Its boiling point sits north of 220°C which means it can survive higher temperature manufacturing processes without losing its punch. The methyl groups block certain reaction sites, dialing down its reactivity compared to plain phenol. This lower reactivity gives it the edge where thermal stability means less breakdown and less off-gassing. Solubility stands moderate in water but increases sharply in organic solvents, broadening where chemists can use it just by picking the right blend.
Industry circles toss around the word ‘Mesitol’ for this chemical, and if you read through a materials safety data sheet, you’ll spot the CAS number 697-82-5. Companies usually ship it in sealed metal drums, with labeling keyed to international transport standards and clear hazard warnings. Batch purity runs above 98%. Every batch run gets checked for color, melt point, and levels of trace isomers. Labels must follow Global Harmonized System guidelines; pictograms for combustibility and skin irritant top the list. These technical specs aren’t bureaucracy for bureaucracy’s sake—they stop cross-contamination and make sure downstream users stay safe.
Synthesis of 2,3,5-trimethylphenol tracks back to methylation of phenol. Some producers rely on alkylation with methanol in presence of catalysts, often using cuprous or aluminum-based supports to coax methyl groups into position. Selectivity isn’t trivial—the trick lies in getting three methyl groups onto the ring at the right points without filling all six slots or overreacting to tetra- or pentamethylated byproducts. Advances in catalyst design let operators steer the reaction more finely. After reaction, fractional distillation pulls the target compound away from a mess of close-boiling relatives and tars that gum up columns. Early processes produced a chemical soup; new ones deliver a crystalline product that meets both purity and cost needs.
On the benchtop, 2,3,5-trimethylphenol plays as a source material for more ambitious syntheses. The methyl groups steer substitution reactions, so chemists often protect them or exploit their electron-donating behavior to drive reactions at precise ring positions. This opens a path to produce bespoke antioxidants, flame retardant precursors, and even some agrochemical intermediates where a bit more stability pays off. Electrophilic substitutions run smoother at the remaining ortho and para sites not crowded by a methyl. The compound rarely undergoes direct oxidation unless forced by harsh reagents, so it offers backbone stability in oxidative environments. It also takes up space in resin chemistry, lending itself to structures that need bulkiness and rigidity without losing phenolic activity.
Outside textbooks, 2,3,5-Trimethylphenol can show up as Mesitol, methylphloro, or 2,3,5-trimethylhydroxybenzene. Across languages or different labeling laws, those names let buyers, sellers, and end users know that they’re working with T-cresol’s heavily methylated cousin. Accuracy in names matters here; with dozens of phenol isomers in supply chains, a slip-up swaps more than just a letter and can shift the entire behavior of an end product.
With compounds like 2,3,5-Trimethylphenol, the hazards aren’t always obvious. The medical odor warns of volatility and possible irritancy. Direct skin contact triggers redness or mild dermatitis, so operators count on gloves, goggles, and good ventilation even in bench work. Industry labeling standards shine here: flammable liquids mark the label, and most operations handle it only inside explosion-proof rooms. Waste catches special handling as per local chemical codes, both to avoid accidental fires and to prevent runoff into waterways. Recovery and recycling cuts waste and keeps costs in check. By protecting workers and keeping emissions low, companies comply with regulations and protect their process from downtime.
This molecule doesn’t grab attention on supermarket shelves, but tug on the supply chain and you’ll spot it everywhere from adhesives, made tougher by methylphenolic backbones, to antioxidants for rubber blends. It gives phenolic resins just enough steric bulk to resist heat without going brittle. Lubricant makers add small quantities to oil blends to improve performance at temperature extremes. The electronics sector leans on methylated phenols in photoresist chemistry. Outside these big names, smaller niches like custom pigment production and agricultural intermediates see their own demand spikes, depending on the year’s technological cycle.
Chemists in both industrial and academic labs keep tuning 2,3,5-trimethylphenol’s properties. They’re testing catalysts that boost yield or slash reaction times, always pushing for cleaner, greener synthesis. Research groups keep investigating its antioxidant properties in new settings—think food preservation and polymer stabilization—while computational chemists model its breakdown pathways for safer, more controlled use. On the analytical side, new methods for detecting ppm-level contamination help keep finished goods clear of regulatory headaches. Over the last decade, pressure for bio-based production continues rising. Using renewable feedstocks for its synthesis would reduce reliance on petroleum, cut greenhouse emissions, and open the product to more markets facing tighter environmental rules.
Work on toxicity circles back to concerns with other lower molecular weight phenols. Acute exposure looks unlikely to cause long-term harm in controlled settings, but chronic exposure stories—dermatitis from repeated contact, or headaches from inhalation—remind anyone handling it to respect the science. The compound’s breakdown in the environment can produce smaller phenols, which carry their own baggage for water and soil contamination. European regulatory bodies already set watch limits on phenolic compounds in workplace air and wastewater. Green chemists keep searching for degradation pathways that minimize risks and for filtration solutions that catch trace amounts before they hit municipal water systems.
Looking out over the next ten years, 2,3,5-Trimethylphenol’s prospects tie closely to shifts in regulations and the pace of specialty materials science. Sectors like high-performance polymers and niche consumer chemicals thrived on molecular customization, and tweaks to phenolic building blocks set the stage for the next generation of coatings, electronics, or adhesives. Supply looks solid so long as upstream feedstocks stay steady—any hiccup in petrochemical supply chains could spur a search for greener or more resilient alternatives. Wider public and regulatory attention on chemical safety means every new application fights for approval based on solid toxicological data. Ongoing research is set to clarify both its environmental breakdown and safe use across complex production cycles, while advances in production tech could unlock efficiency leaps that both save costs and lower emissions. The real legacy for 2,3,5-trimethylphenol, sitting on lab shelves and in freight containers worldwide, rests in the hands of chemists who see it as both a tool and a puzzle—one with just enough mystery left to keep the field innovating.
In my experience, it’s easy to watch science documentaries and think the world runs on dramatic discoveries or massive breakthroughs, but the truth: Life relies on a long list of humble chemicals that never show up on TV. One that often flies under the radar is 2,3,5-Trimethylphenol. Just hearing the name, most folks would think it belongs locked away in some research lab. But the roots of modern industry stretch into some strange corners.
So what does 2,3,5-Trimethylphenol actually do? To someone who’s mixed chemicals on a bench and watched the market for specialty chemicals, the answer feels straightforward: This compound arrives in the chain of making things like vitamin E, plastics, colorants, and some pretty specific chemical intermediates. In simple terms, companies turn this phenol derivative into building blocks for other valuable stuff—sort of like flour for bread, but for industry. Instead of ending up in a bagel, it winds up in health supplements and everyday materials.
One of the classic uses of this molecule is as an intermediate in vitamin E synthesis. Large chemical makers take it, react it with other chemicals, and out comes vitamin E, which millions of people swallow every morning. There’s something strangely satisfying in knowing that a small tweak in a chemical structure can eventually end up doing good for human health.
You’ll also spot traces of 2,3,5-Trimethylphenol in production of resins and antioxidants. Certain plastics and coatings wouldn’t work the same way without these chemical helpers. The material world runs on a hidden supply of specialty compounds. A friend of mine once pointed out that even if you skip all supplements, you’re still surrounded by these chemical “middlemen” all day long—your electronics, furniture, even parts of your car.
Industrial chemistry has a reputation for being cold and abstract, but having watched the manufacturing world up close, I’ve learned it’s filled with small pieces, each carrying big responsibility. The industry’s reliance on things like 2,3,5-Trimethylphenol opens the door to all kinds of challenges—cost, sourcing, safety, environmental impact. One bad link in the chain, and the ripple spreads fast.
Environmental concerns don’t just exist as footnotes, either. Production and use of phenol derivatives demand tight controls. Workers need proper training and safety protocols. Regulations show up for a reason: to steer these compounds away from causing trouble downstream. Companies who value safety and compliance generally see fewer problems, but lapses aren’t unheard of.
Instead of ignoring how critical these chemicals are, it seems smarter to shine a light on them. Chemical manufacturers could invest more in greener synthesis and better recycling. Universities continue searching for cleaner routes and drop-in alternatives, but the real momentum happens when industry wraps safer practices into daily operations. Cutting back on waste and keeping a close eye on workplace exposure makes a difference—not just for profits, but for the community surrounding these plants.
The lesson from 2,3,5-Trimethylphenol isn’t about memorizing obscure chemical names. It’s about understanding how much of our modern life springs from unsung compounds, created and handled by people whose choices matter. If the industry keeps listening to safety experts, regulators, and the communities that share the land, these building blocks can keep serving us all—this time, with everyone’s wellbeing factored into the recipe.
People often glaze over when hearing a name like 2,3,5-Trimethylphenol. It sounds like something locked away in a far corner of a chemistry textbook. Still, behind the tongue-twister sits a connection to both the structure of our world and the products that fill shelves and labs. The chemical formula for 2,3,5-Trimethylphenol is C9H12O. That string of letters and numbers gives it a place in the big universe of organic compounds called phenols.
C9H12O seems straightforward, but even after years in science classrooms, seeing those notations doesn’t always switch on a lightbulb. Breaking it down piece by piece makes all the difference: nine carbon atoms, twelve hydrogen atoms, and a single oxygen atom form 2,3,5-Trimethylphenol. Each methyl group—the CH3 chunks—attaches itself to carbons two, three, and five on a phenol ring. What makes this layout special? The placement of those groups changes how the compound behaves and how people use it.
This chemical, like many phenols, matters most behind the scenes. You won’t find it painted brightly on supermarket packages, but it plays a role in the development of antioxidants, pharmaceuticals, and specialty resins. Research points to its usefulness in making stabilizers that keep things fresh and in certain drug-making steps. As someone who’s spent time in research labs, I’ve seen just how a single shift in molecular arrangement impacts the effectiveness of what comes off the benchtop.
Rarely do we pause to think about the people behind laboratory doors, but trust and transparency matter. There’s comfort in knowing the science community sticks to guidelines set by groups such as IUPAC, which help prevent mislabeling or misuse. When talking about compounds like 2,3,5-Trimethylphenol, following these naming and sourcing standards avoids confusion and strengthens consumer confidence.
Discussions around synthetic chemicals always bring up health and environmental concerns. While direct toxicity data for 2,3,5-Trimethylphenol isn’t splashed across headlines, its chemical relatives come with known risks to aquatic life and sometimes people. That’s where experience in handling chemicals, wearing protective gear, and safe disposal practices come into play. Education about proper lab techniques and prompt reporting of spills or leaks lessens risks to people and planet.
Chemical jargon shrinks the distance between experts but blocks out everyone else. As someone who once puzzled over such terminology, I recognize the importance of digestible explanations. Bringing transparency to compounds like C9H12O helps people outside the field make better decisions about safety, research, and policy.
When we talk openly about what’s in a bottle, how it gets used, and potential impacts, everyone benefits. Scientific rigor partnered with honest communication creates trust—something the world needs as chemistry keeps shaping daily life.
People hear a name like 2,3,5-Trimethylphenol and their eyes might glaze over. In practice, though, this chemical brings real challenges. Working in labs and factories, I’ve seen how even small mistakes with such compounds create lasting headaches—from skin burns to lung problems and even longer-term health risks. Nobody walks into work hoping for a chemical accident, but those moments happen fast, especially with organic solvents and aromatic compounds.
If you’re dealing with 2,3,5-Trimethylphenol, skin and eyes usually see the trouble first. Sometimes you notice irritation quickly, like a stinging sensation, or sometimes it creeps up later. That oiliness you feel on your fingers—that’s not just a minor annoyance. Chemicals like this soak right into the skin and then into your system. Fumes can build up too. Even if the smell isn’t strong, it doesn’t take much for vapors to cause headaches, dizziness, or worse after breathing them in for an hour or more. I’ve had coworkers brush off a “little whiff,” only to regret it the next day.
Nobody wants to suit up like they are stepping into a space capsule, but basic personal protective equipment saves more than just laundry bills. Nitrile gloves do a better job than latex for restricting passage of aromatic solvents like this one. A solid pair of safety goggles matters as much as your eyesight. If you can, go with splash-proof goggles instead of those cheap open-corner glasses. For people who work around this chemical for hours, respirators rated for organic vapors provide real protection, especially inside rooms lacking solid ventilation.
In real-world settings, there’s always somebody who believes they are too fast or too careful to worry about spills. Trouble is, spills catch everyone off guard. Having absorbent pads and chemical spill kits sitting by the workbench sounds obvious, but most accidents expose how rarely people keep those close by. My own lab improved safety when the chemical fume hood stopped being just for mixing big batches. Everyday measuring, weighing, and pouring worked better under a fume hood—less vapor, less risk. Regular clean-up and checklists help too, especially after a long shift when mistakes sneak in.
A proper storage plan goes beyond putting containers in a cabinet and calling it a day. This chemical reacts with strong oxidizers, and sometimes, it causes problems with metal containers. Storing chemicals in designated, labeled bottles—never food containers—cuts down confusion. Regularly checking expiration dates may seem like busywork, but old containers spell trouble. For disposal, many places treat phenolic compounds as hazardous waste, so dumping leftovers down a sink isn’t just risky—it’s illegal in plenty of places.
No workplace or academic lab should expect newcomers to “figure it out” by watching others. Hands-on training, clear signage, and step-by-step walk-throughs transform daunting protocols into habits. New staff benefit from walking through what to do during a minor spill or splash—not just reading a poster. Supervisors and coworkers must set the standard every day, because safety guidelines only work if everyone takes them seriously even during busy stretches.
In my experience, safety comes down to habit, trust, and a willingness to speak up if something feels off. There’s never a substitute for preparation. No job or experiment runs smoother by skipping gloves or guessing at proper cleanup. It’s the boring routines—the right masks, working fume hoods, prompt spill responses—that prevent injuries and, honestly, keep everyone’s day on track.
Chemistry has a way of sneaking into daily routines, even for folks outside the lab. Take 2,3,5-Trimethylphenol. It's one of those small-aroma phenols that show up in research, manufacturing, and even environmental talks. The key properties everyone asks about? Melting point and boiling point. For 2,3,5-Trimethylphenol, the melting point lands around 71°C, and the boiling point clocks in near 238°C. These aren’t just trivia for a flashcard set—they actually shape how companies and researchers use, store, and assess this chemical.
I can recall my first year in a college lab, sweating bullets as I did a distillation. Temperatures matter. If a compound melts at a certain temperature, anyone using it knows where solid turns to liquid. With 2,3,5-Trimethylphenol, that 71°C threshold means you can store it at normal room temp and expect a solid, not a puddle. That fact makes packaging and transport a breeze compared to something that would ooze out of a container.
Jump ahead to the boiling point, and you’ve got 238°C—past the mark where water is long gone as steam. For folks manufacturing fine chemicals or working in labs, this tells you where the substance leaves the beaker as vapor. Synthetic chemists rely on these numbers to set up their gear, adjust heating rates, and avoid nasty surprises or equipment failures. Try heating it without respect for that boiling point, and you get loss of product, ruined apparatus, or worse.
There's a big push in the specialty chemicals sector for efficiency—fewer wasted batches, fewer emissions, safer handling. Details like where a compound melts or boils help companies decide how to scale up from a glass beaker to a few tons in a drum. Lower melting points can mean energy savings, while higher boiling points allow for broader reaction conditions. For 2,3,5-Trimethylphenol, that middle-of-the-road melting and high boiling point open up use in manufacturing antioxidants, resins, and pharmaceutical intermediates.
If you’ve ever visited a mid-sized chemical plant, you might have noticed the focus on safety. Proper knowledge of melting and boiling points prevents pressure build-up and controls fire hazards. The American Chemical Society stresses that properties like these get punched into safety plans and process simulations. People base entire reactor protocols around these two numbers.
Runoff and waste management depend on how stubbornly a chemical hangs around in solid, liquid, or vapor form. Anything that boils at a lower temperature risks evaporating into the air during routine use, raising chances for worker exposure or atmospheric pollution. With a high boiling point like 238°C, 2,3,5-Trimethylphenol doesn't just float off into the environment under normal conditions. Still, the industry needs to keep an eye on waste streams, accidental releases, and storage near high-temperature equipment.
It’s easy to shrug off details like melting and boiling points, but that’s the stuff that directs safe, responsible chemical use. Environmental agencies and even smaller labs are pushing more rigorous training about substance properties. In my own experience, getting these details right makes the difference between a smooth project and a late-night panic phone call to the safety officer.
By grounding decisions in actual data—like the numbers for 2,3,5-Trimethylphenol—science becomes less mysterious and a whole lot safer, for both the people in the lab and the wider community.
Browsing through safety sheets in my chemistry coursework once, 2,3,5-Trimethylphenol caught my eye—not because it’s everywhere in our homes, but because how we treat chemicals at the margins often shows how well we protect our health. People rarely talk about this compound outside industrial circles, but that doesn’t mean it deserves less scrutiny.
2,3,5-Trimethylphenol is no household name, but its close relatives, the methylphenols, pop up in everything from resins to pharmaceutical production. The standard story goes that many phenols irritate eyes and skin. A few are worse, hitting the liver or kidneys after too much exposure. But finding extensive, up-to-date details on this one proves tricky. That’s part of the problem: policy and science tend to pay less attention to chemicals that haven’t yet made headlines for causing trouble.
Looking at the research available, exposure to 2,3,5-Trimethylphenol usually happens in places where workers handle chemical manufacturing. Existing safety literature groups it with substances that can be harmful if inhaled or touched in high concentration. The irritation to the eyes, skin, and respiratory tract does not set it apart from many other aromatic chemicals. These effects show up after direct contact at higher exposures.
The catch is uncertainty. There’s limited published research on long-term or low-dose exposure to 2,3,5-Trimethylphenol in people. Older studies on similar methylphenols suggest liver and kidney stress after consistent overdose, but rarely focus on this specific compound. One can’t rule out more serious effects without more study. Toxicity data from animals suggest caution, but public sources do not confirm whether it accumulates in the body or how it might interact with other regular exposures in daily life. That knowledge gap puts a premium on careful handling.
Having handled chemicals in a university teaching lab, I’ve seen how a small lapse—forgetting gloves or working without enough ventilation—turns a routine task into a personal hazard. Safety isn’t a box to tick on a form; it’s real protection against known and unknown risks. For workers using 2,3,5-Trimethylphenol, personal protective equipment makes a difference. Good lab practice, sealed containers, and effective extraction fans take priority. Outside the plant gate, this chemical should never end up in groundwater or air near homes. Strict disposal is basic common sense, not just regulatory red tape.
Nobody should shrug off the hazard just because detailed toxicity profiles don’t fill a binder. Chemical safety relies less on waiting for disaster and more on applying what we do know: minimize skin and inhalation exposure, avoid spills, and keep usage away from food, water, or home environments. Of equal importance, ongoing monitoring and public transparency matter. Industries and regulators need to share exposure records openly—not as after-the-fact damage reports, but as routine communication.
Public health depends on bridging the gaps between expert circles and the rest of us. That means keeping a critical eye on old compounds—and respecting that “not proven hazardous” is never the same as “perfectly safe.”