Fluorinated aromatic nitriles didn’t pop up by chance. They grew out of a persistent quest in organic chemistry to create stronger, more versatile materials. Scientists saw the benefits of fluorine long ago: add a few fluorine atoms to a benzene ring, and the compound resists heat, corrosion, and chemical attack far better than what you started with. 2,3,5,6-Tetrafluoroterephthalonitrile entered the scientific conversation once public and private labs started looking for monomers capable of building the next generation of polymers. In the backdrop of rapid advancements in electronics and engineering plastics after the 1950s, fluorinated building blocks became the star materials for insulation, membranes, and tough coatings.
This complex-sounding molecule mostly looks like a benzene ring with two nitrile groups and four tightly-clustered fluorine atoms carefully arranged. Chemists appreciate this structure for its tenacious stability and its knack for doing chemical work without giving up its core qualities. From what’s seen in research labs and manufacturer portfolios, these nitriles show up in specialty plastics, cross-linked membranes, and some advanced electronics. Fluorinated aromatics tend to cost more than their non-fluorinated cousins, yet the performance gains in harsh settings justify the investment for industries that take durability and precision seriously.
No matter how tough or exotic a chemical may seem, simple traits decide where it lands—solubility, melting point, how it behaves with heat or shock. Tetrafluoroterephthalonitrile gives a crystalline solid at room temperature, typically pale or colorless. Fluorine atoms shrink the molecular "bulk," pulling electrons tightly enough that moisture doesn’t break it down. Expect high thermal stability: it doesn’t melt or decompose easily, making it a favorite in applications that stretch from cryogenic temperatures to low-end pyrolysis. With strong electron-withdrawing groups (four fluorines and two nitrile claws), the molecule resists many bases and acids, making it promising for technical work in caustic environments.
In my work, technical documentation seldom stays dry; it shapes how chemists and engineers handle tricky solids and solvents. Regulatory demands push for purity markings—usually at or above 98 percent for serious research and industrial runs. Labeling means clarity on hazards: the irritant potential, fire risk, and exactly how to store away from moisture or incompatible solvents. For shipping, people mark fluorinated compounds with UN numbers, hazard symbols, and clear warnings to avoid accidental release. Good data helps keep incidents rare, saves time during inventory checks, and prevents pointless confusion at customs.
The route to 2,3,5,6-tetrafluoroterephthalonitrile follows basic organic rules: start with a tetrafluorinated benzene, introduce nitrile groups using copper-catalyzed coupling or Sandmeyer-type reactions, and purify by recrystallization or chromatography. This isn’t a backyard operation. Controlling temperature, scavenging side-products, and working under clean, dry conditions matter. If nitrile introduction goes astray, expect isomers or incomplete substitution, wasting expensive starting materials—a problem my colleagues and I have cursed more than once. Advances in continuous-flow synthesis and better catalysts have boosted yields and cut down on the harshest reagents, though these improvements don’t come for free.
Tetrafluoroterephthalonitrile isn’t just admired for what it is, but for what chemists can make it do. Nitrile groups provide versatile handles for further transformation. Under basic or catalytic conditions, each nitrile can become an amide or carboxylic acid, opening doors to more complex polymers or linkers for material science. Even the otherwise-unreactive ring can give way—fluorine’s electron-pulling power activates the molecule for aromatic nucleophilic substitution, which lets builders swap in a custom group where needed. That allows researchers to tune solubility, adhesion, or even add custom electronics features by introducing new substituents. Each successful tweak makes tomorrow’s membrane or sensor just a bit stronger or more selective.
In the world of specialty chemicals, naming confusion slows research and procurement. Synonyms often surface: “Tetraphtalonitrile, tetrafluoro-,” “TFTPN,” or by IUPAC standards, “2,3,5,6-tetrafluorobenzene-1,4-dicarbonitrile.” The market may use catalog identifiers, but for scientific work, precision matters more than brand. My experience tells me scientists spend far too much time double-checking synonyms and catalog listings, as a tiny slip brings the wrong compound and halts expensive research.
No amount of laboratory savvy replaces solid safety rules. Fluorinated nitriles don’t often react with air or moisture, sparing us violent surprises, but they still deserve respect. Dust can irritate skin, lungs, and eyes. Inhalation and direct contact call for goggles, gloves, and clean work surfaces. For storage, dry containers and low temperatures reduce breakdowns or accidental release. Spills demand quick cleanup—my old lab had staff drill for this, no matter how confident folks grew. Operators working with high-reactivity catalysts or pressurized vessels must follow local standards and carry fire-rated extinguishers nearby. Disposal should never cut corners: waste can’t just get tossed; licensed handlers must treat it as hazardous, even in small amounts, because fluorinated compounds linger in soil and water.
The places where tetrafluoroterephthalonitrile shows its strengths keep growing. Most people outside the field miss how vital these building blocks are for making membranes used in water purification, battery separators, fuel cells, or even next-gen microelectronics. I’ve seen it specified in cross-linked polymer matrices to produce coatings that shrug off concentrated acids or bases. Newer uses include optical sensors, membranes in medical diagnostics, and custom fluorinated resins for spacecraft wiring. What links these applications is a demand for precision and endurance—where failure costs more than the premium paid for advanced materials. If chemists can tweak physical properties just so, it becomes possible to design materials custom-tailored for jobs that old-school polymers can’t handle.
Ongoing research keeps feeding new uses and synthesis tricks for these fluorinated nitriles. Academic and private labs test new catalysts that let them run cleaner, faster reactions at lower temperatures—this cuts waste and improves worker safety. Analytical teams chase more exact ways to spot and separate isomers or byproducts. Some of the most exciting research explores blending tetrafluoroterephthalonitrile-derived polymers with nanoparticles to enhance toughness, electrical conductivity, or gas-selectivity. That means better filtration, stronger composites, or sensors that survive harsh environments. Funding tends to chase two big goals: lowering costs using greener methods and developing custom materials for ever-smaller, smarter electronic devices.
Safety in chemicals boils down to direct and long-term effects on health and nature. Fluorinated compounds raise tough questions, since they don’t break down quickly in landfills or water supplies. For tetrafluoroterephthalonitrile, direct toxicity in the lab remains manageable—irritant but not immediately lethal or carcinogenic at low exposures. Problems come from improper disposal or repeated exposure, especially through skin or inhalation in under-ventilated spaces. Animal studies point to possible organ effects at high doses. Environmental health experts flagged these and similar compounds for deeper review, urging stricter labeling and calling for research into safe breakdown or long-term containment, both during production and after the product leaves the manufacturer.
Looking ahead, fluorinated aromatic nitriles hold promise as the backbone for solving real-world problems—making seawater drinkable, shrinking electronic circuits, and building batteries that work across arctic and desert extremes. The cost and environmental footprint demand creative solutions, such as closed-loop manufacturing or engineerable biodegradation. If tomorrow’s regulatory pressure grows as expected, companies ready to develop greener syntheses and safer disposal pathways will lead the pack. That pulls scientists, engineers, and regulators together: sustainability will no longer be a buzzword but a hard requirement. In my own time at the bench and the whiteboard, I’ve watched researchers rally around these problems, and despite the hurdles, breakthroughs keep coming. Tetrafluoroterephthalonitrile, quietly, sits near the center of that progress—showing how even a tough, fluorinated molecule can carry so much of technology’s future on its sturdy, carbon-fluorine frame.
Most people have never heard of 2,3,5,6-tetrafluoroterephthalonitrile. Yet, the truth is, this finely-tuned molecule helps drive innovations behind the scenes, especially in the materials science world. Chemists prize it for the two nitrile groups locked onto a benzene ring, flanked by four fluorine atoms. This arrangement packs a double punch: chemical stubbornness that shrugs off corrosive influences, and a unique shape that lets it link up with other molecules in ways that less specialized compounds can't compete with.
New polymers don’t just stroll out of a lab; they need backbone molecules tough enough to stand up to mechanical stress, heat, and all sorts of nasty chemicals. 2,3,5,6-Tetrafluoroterephthalonitrile deserves a mention in the ongoing story of high-performance plastics because of its role as a monomer for crafting specialty polymers. These aren't your everyday plastics—we’re talking about materials that tackle some of the thorniest engineering and safety problems. For instance, the fluorine atoms stitched into the core of these products help them repel water and resist being broken down by acids or bases. That means less corrosion and more reliability from airplane fuselages to fuel lines.
Electronics manufacturers also rely on this compound's properties. Printed circuit boards, which form the “nervous system” of every modern device, benefit from high-temperature and flame-resistant layers. Sometimes, those layers start with molecules like 2,3,5,6-tetrafluoroterephthalonitrile. The safety margin goes up, and so does the device’s life span. The result: fewer failures in crucial equipment, from satellites to smartphones.
Anytime a chemical gets praised for being resistant to degradation, questions about environmental fallout aren't far behind. No one wants to see “forever chemicals” pile up in water systems or food chains. Polymers born from this compound are tough, but their persistence in the environment looks like a double-edged sword. Companies and regulators face a tricky balancing act—embracing the benefits while keeping an eye on what happens at a product’s end of life.
My own work with specialty chemical suppliers showed that waste management and safe processing don’t always keep pace with scientific progress. Some innovations skip the thorough due-diligence stage because the market pressures are relentless. Taking shortcuts might squeeze out more profit for a year or two, but it leaves society facing tougher questions down the line. Strong partnerships between industry watchdogs, environmental scientists, and manufacturers help stop corners from getting cut. Thoughtful rules—grounded in real risk assessments and not just knee-jerk reactions—will shape how these polymers can be safely used long-term.
With more public awareness of chemicals that persist in the environment, some promising strategies come to mind. Chemical engineers already hunt for alternative monomers that can break down after their service life, or they tweak existing molecules to nudge them toward safer degradation. Businesses that invest in closed-loop recycling take responsibility for their products well past the point of sale. As more people demand transparency about what’s inside the products they use, companies have a real reason to lead with integrity.
2,3,5,6-Tetrafluoroterephthalonitrile rarely makes news headlines, but its story touches industries, consumers, and the planet. Smart stewardship, more research, and practical policy can help us welcome the useful side of this chemical while keeping its impact in check. The challenge isn’t going away. If anything, that kind of vigilance will only become more important as science keeps pushing boundaries.
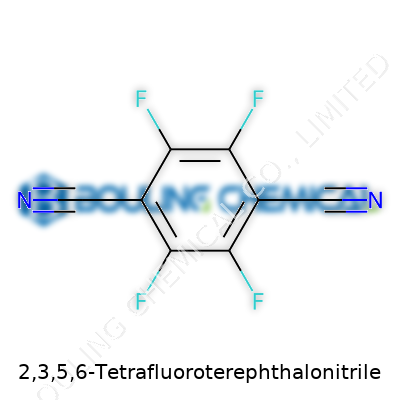
Every now and then, a name like 2,3,5,6-Tetrafluoroterephthalonitrile crops up and people either breeze past it or wonder what all those syllables mean. Underneath that intimidating label sits a molecule with real-world impact, especially in advanced material and chemical research. The molecular formula—C8F4N2—may look straightforward to a chemist, but its structure and significance deserve a closer look. Picture a benzene ring, but four hydrogen atoms have been kicked out and replaced with fluorine atoms at the 2, 3, 5, and 6 positions. On atoms opposite each other, a pair of nitrile groups set up shop at the 1 and 4 positions. This creates a flat, rigid molecule laced with electronegative fluorine and electron-withdrawing nitriles.
The fluorinated benzene core isn’t just a clever chemistry stunt. Swapping hydrogens for fluorines does more than change a name. Fluorine turns up the electron-withdrawing strength and boosts chemical stability. Polymers or specialty coatings built from this stuff tend to last, resist breakdown, and shrug off harsh solvents. The symmetrical dinitrile pairing offers sharp reactivity, letting the molecule connect into larger frameworks or react with select partners to form tough, heat-resistant materials.
Fluorination never comes cheap or easy. In my lab days, working with highly fluorinated feedstocks always meant managing extra steps and extra care. The tricky synthesis often stays tucked behind company doors, but researchers dug up reliable routes decades ago, often anchored to commercial fluorobenzene and smart nitration strategies.
This compound isn’t a household staple or something found in kitchen cleaners. Think high-performance plastics, selective membranes, or cutting-edge dielectrics. Researchers crave rigidity, stability, and unique electronic properties for things like lithium-ion battery membranes or resistant coatings for electronics. The molecular structure does real work: it hands out toughness and blocks water, oxygen, and relentless acids with more punch than most organic molecules can muster.
Development of robust nano-materials has leaned on the likes of tetrafluoroterephthalonitrile. A few grams in the right lab might become fragments in covalent organic frameworks—those with neat, grid-like architectures prized in gas separation and energy storage. That benzene skeleton, stripped and armed by fluorines, becomes a backbone for innovation. I’ve seen colleagues transform such molecules into high-surface-area scaffolds able to trap toxic gases or catch and release valuable rare-earth metals—simply because the structure lets them engineer precise pore sizes and stabilities.
Sourcing affordable, scalable fluorinated intermediates still pains many researchers. Environmental impact nags at the back of every chemist’s mind. Fluorinated compounds don't break down easily, and nobody wants to build tomorrow’s pollution. Labs and manufacturers need a circular mindset—close-loop processes, safe disposal, or recycling routes.
These aren’t abstract problems. Regulations grow tighter. Solutions might start with green chemistry tweaks, clever catalytic shortcuts, or sustainable fluorine sources, but every chemical built to last needs an end-of-life plan. Otherwise, practical value risks tumbling under environmental realities. The structure of 2,3,5,6-tetrafluoroterephthalonitrile hands us performance and durability, but the next step is building smart ways to make it and break it without leaving scars.
Not all chemicals demand the same level of respect, but anybody who’s worked in a lab for more than a minute knows that some compounds require extra thought in how you handle them. 2,3,5,6-Tetrafluoroterephthalonitrile falls into that territory. The name doesn’t exactly roll off the tongue, but what matters is what this stuff can do if you skip proper safety steps.
I learned early on in research that every compound has a personality. Some are just stubborn powders, others react with humidity or light, and some—like 2,3,5,6-tetrafluoroterephthalonitrile—threaten harm if you get too close without thinking ahead. This chemical can irritate skin and eyes and it doesn’t play friendly with your breathing if dust gets airborne. Anything with ‘nitrile’ in its name signals a need for gloves and goggles, no exceptions.
Direct contact leaves a mark. You don’t want that on your hands or anywhere near your face. Trusting the bare minimum is a shortcut straight to a health report nobody wants. We had a guy in our group who shrugged at gloves during a late-night experiment—ended up flushing his hands for way longer than he planned. The lesson stuck for everyone.
I never underestimate the importance of a good fume hood. If you think opening a window in your kitchen compares, you haven’t smelled a chemical accident. Powdery substances like this one have a way of lingering, and even tiny bits kicked into the air do damage with every breath. Always measure and transfer inside a hood. I treat it as a rule, not a suggestion, especially since many laboratories haven’t done a ventilation upgrade since the seventies. You can’t control everything, but you can make sure you’re not working in a fog of your own making.
Spills happen, and pretending otherwise is fantasy. I keep a plan in my back pocket for every material I touch. Getting lazy leads to contamination that spreads to shared equipment, sleeves, notebooks, and eventually other people. Think of your workspace as a living thing—it remembers every mistake.
The temptation to just toss waste in the wrong container is real. I’ve seen it done, and watched the fallout unfold. Compounds like tetrafluoroterephthalonitrile don’t just fade away once they leave your bench. They linger in the environment, and if not handled properly, can find their way into places no one intended. This isn’t about playing policeman; it’s about not adding to a growing problem. Follow institutional protocols, label everything clearly, and don’t leave mystery powders for the next shift.
Skepticism saves lives in the lab. Just because nothing went wrong today doesn’t mean tomorrow will be fine if you're careless. All those stories about someone skipping PPE or ignoring a spill—they aren’t rare exceptions, but reminders. Training and repetition make handling dangerous compounds automatic. Safety grows from the bottom up—via shared responsibility across the lab, not from a poster on the wall. So respect every compound, double-check your routine, and treat each bottle as if it’s more dangerous than you remember. In the end, the only good accident is one that never happens.
I remember the first time I opened a drum labeled 2,3,5,6-tetrafluoroterephthalonitrile—long name, potent impact if not respected. Back then, the notion of “just storing chemicals in a dry spot” seemed good enough. After a few close calls and one panicked search for the right SDS, my attitude changed. In the real world, smart storage prevents not only ruined research but health scares nobody wants to write home about.
This compound belongs to the family of fluorinated aromatics, and while its applications stretch from specialty polymers to emerging materials, the stuff’s more than just another white powder. Trace moisture or a cracked container can turn convenient storage into a cleanup headache. There’s a reason why chemistry teachers lose sleep over their storage cabinets.
It’s tempting to stash powders anywhere dark, but 2,3,5,6-tetrafluoroterephthalonitrile pushes back at carelessness. Over time, even minor humidity creeps in and triggers unwanted reactions, some releasing toxic byproducts. The safest labs I’ve seen always rely on tightly sealed containers that never see condensation or standing water. Think glass or high-grade plastic with flawless closures. Moisture stays locked out or problems sneak in.
Excessive heat risks slow decomposition, a process that might not smell or smoke but still wrecks material purity. Locked fridges or climate-controlled shelves keep the stuff at a steady, cool temperature that shields it from summer flares and winter chills. No surprises equals no accidents.
More than one chemist has reached for a white crystalline solid and triggered an evacuation because labels faded or fell off. For fluorinated compounds like this one, clarity is non-negotiable. I keep labels with bold print, waterproof ink, and storage dates. This might sound basic, but firms around the world document real incidents rooted in sloppy labeling.
Segregation gets overlooked by newcomers. 2,3,5,6-tetrafluoroterephthalonitrile doesn't play nice with strong acids, bases, or oxidizers. Storing it close to those just invites trouble. Shelves divided by chemical class save more than lab space—they keep people safe and the cleanup crew off overtime.
The American Chemical Society points out that improper storage stands behind a large share of laboratory accidents. Studies published in chemical safety journals back up this claim, showing common themes: humidity infiltration, temperature fluctuation, and misidentification pop up in post-incident reports far too often.
Real-world solutions look like regular audits, clear labeling policies, and proper investment in quality storage gear specific to sensitive materials. No one-size-fits-all approach beats dedicated chemical storage refrigerators or cabinets with controlled environments. When budgets run tight, prioritizing storage for higher-risk compounds pays off most.
No storeroom or lab is fail-proof, but every time labs treat 2,3,5,6-tetrafluoroterephthalonitrile with the caution and respect it demands, we step closer to safer science and stronger results. I’ve learned it’s never over-cautious to double-check that seal or reread a faded label. Better safe than part of a cautionary tale.
If you take a look inside a lab or production site working with advanced polymers or specialized electronics, chances are good you’ll spot some 2,3,5,6-tetrafluoroterephthalonitrile tucked away on a shelf. It grabs attention in research circles for a reason. This nitrite ring, decked with fluorines, winds up in everything from high-performance materials to specialty chemicals. But, as anyone who’s actually worked with the stuff will tell you, not all samples are made equal—purity matters tremendously in these applications.
From practical experience, chasing higher purity pays off—sometimes in ways you don't notice until something goes wrong. Solid-state synthesis craves clean starting materials. Residual metals or stray organics can wreck a catalyst or introduce stubborn color in a final product. You might find technical grade somewhere above 95%, but the bar often sits higher. For research or making electronic components, suppliers typically focus on 97% or better, and some go up to 98% or 99% to tackle the needs of more critical work. Purity doesn’t just keep a reaction on track; it’s the difference between repeatable results and costly surprises.
Bad batches or contamination aren’t just a nuisance—they slow down months of work. I’ve seen a simple trace impurity throw off analytical readings, plus wasted effort figuring out why. With tetrafluoroterephthalonitrile in particular, the double dose of nitrile and all those fluorines mean it’s less forgiving than other aromatic compounds. If you’re looking for fewer headaches and real consistency, it’s worth insisting on high numbers backed by a robust certificate of analysis.
We can’t talk chemistry without talking logistics. Packaging makes a surprising difference, especially in a compound as sensitive as tetrafluoroterephthalonitrile. Exposure to air, trace moisture, light—these nuisances wreak havoc on neat material and even cause long-term stability issues. In my own experience, samples left in poorly sealed glass vials turn clumpy or begin to decompose, and that’s the last thing you need mid-project.
The most common sizes I’ve run across are small, meant for research: 1 gram, 5 grams, maybe a 10-gram jar. These are usually glass bottles, amber or clear with PTFE-lined caps for a tight seal. The design keeps out the bulk of humidity and stops degradation. Go up a notch, and you’ll see 25-gram and sometimes 100-gram packs available for small-scale production or pilot work. Anything larger usually points to custom packaging, negotiated directly with chemical suppliers; you don’t find multi-kilogram quantities on standard shelves.
Supply reliability means getting what you ordered and actually being able to use it. Many researchers and engineers—myself included—lean toward vendors who put extra work into minimizing contamination and offer tough, protective packaging. Some labs go as far as transferring product to gloveboxes immediately or dividing big lots into smaller aliquots. Good handling doesn't only keep chemical properties intact—it keeps budgets in line and results reliable.
Straying away from the notion that chemistry is just a list of formulas helps focus on what really matters. Better transparency around purity data and packaging specs keeps folks in the lab from guessing. It strengthens collaborations and saves time. As more companies and academics zero in on advanced functional materials, there’s real momentum behind tighter quality controls—even for niche compounds. If suppliers keep developing more robust packaging and keep investing in analytical equipment, everyone benefits. It’s not about chasing fancy numbers, but about getting results that stack up, batch after batch.