Functionalized phenols grabbed the spotlight in organic chemistry as soon as the field began tuning benzene rings for reactivity and stability. In the hunt for stronger nucleophiles and electrophiles, chemists found that swapping out hydrogens with halogens could shift electronic properties dramatically. 2,3,5,6-Tetrafluorophenol stands as a product of this journey. By mid-twentieth century, academic groups reported fluorinated phenols in their search for substrates with tailored activity. Real strides picked up steam as better methods emerged for selective fluorination; the electronegativity changes brought by fluorine atoms continue to fascinate both academic and industrial chemistry labs. Today, tetrafluorophenol is just as likely to pop up in a big pharma synthesis as in a graduate student's toolbox, a testament to decades chasing new reactivity in the aromatic world.
With its four fluorine atoms hugging the benzene ring, this compound draws attention for more than its synthetic challenges. Not every phenol sporting four fluorines gets grouped the same way. 2,3,5,6-Tetrafluorophenol doesn’t just look different; its electron density, hydrogen bonding, and volatility all shift from traditional phenol. This matters when you want to activate an intermediate or tune a molecular scaffold. Instead of unpredictable behavior, you get repeatable patterns. Folks working in analytical chemistry or industrial manufacturing know that materials like this move the needle, raising the floor of reproducibility in advanced material science or medicinal chemistry. I remember the time a colleague struggled to block an unwanted oxidation with standard phenols—switching to tetrafluorinated material made the unwanted side reaction drop off completely.
At room temperature, you find 2,3,5,6-Tetrafluorophenol as a solid, with a distinct, sharp odor. Melting and boiling points land lower than non-fluorinated cousins because those fluorine atoms disturb intermolecular forces, disrupting the typical hydrogen bonding pattern seen with plain phenol. Its solubility leans where you'd expect—the presence of both hydrophilic and hydrophobic sides makes it stubborn about mixing with water but more welcoming to organic solvents. Once you handle it, you don't forget the sensation; the volatility and odor alone call for respect in the lab. The acidity of the phenolic hydrogen drops compared to regular phenol, thanks to the electron-withdrawing pulls from the tightly clamped fluorines. Reactivity profiles shift as a result; you can expect reduced nucleophilicity at the ring, but electrophilic palladium or copper complexes still see this substrate as a lively partner.
Labs expect high purity for bench work and scale-ups, and suppliers typically guarantee a minimum of 98% for 2,3,5,6-tetrafluorophenol. Analytical labs regularly run NMR or HPLC tests for verification. Researchers pay close attention to the batch and lot numbers, verifying against certificates of analysis, since even minute contamination from water or residual acid can throw off sensitive reactions. Even the storage label matters—amber glass vials help minimize photo-degradation, and desiccants keep moisture out. Safety warnings show up right away on bottles, reflecting the compound’s strong smell and skin-irritation potential. Any user with a bit of lab experience spots the warning pictograms, reminding them this isn’t just another anodyne reagent.
Getting to this fluorinated phenol isn’t straightforward with raw reagents and glassware. Direct fluorination of phenol almost guarantees a mess of isomers and tar. Syntheses usually start with pentafluorobenzene, selectively knocking out one fluorine with hydroxide under controlled conditions. Hydrogen fluoride or potassium fluoride often turns up as the fluorine source for precursors at an earlier step. The reaction setup demands equipment resistant to strong bases and acids—Teflon becomes a go-to material for anything touching the reaction mixture. Careless work can lead to corrosion or even fires. In my own research experience, careful control of temperature and slow addition saved hours of cleanup and heartbreak, especially on the day high-purity product came out instead of the usual brown sludge.
Organic chemists favor 2,3,5,6-tetrafluorophenol as a leaving group in activation strategies. For example, forming esters or ethers with this phenol backbone boosts downstream reactivity, because the resulting tetrafluorophenoxy group exits much more easily than most. This property surfaces in complex molecule synthesis, peptide coupling, and activation of carboxylic acids or phosphate esters. Coupling agents like DCC and EDC rely on such properties to drive difficult reactions forward. The aromatic ring, bristling with four fluorines, resists most electrophilic substitution, staying inert against most acids and oxidants. But nucleophilic aromatic substitution can take place at the remaining hydrogen or at open positions if the right conditions show up. Scientists keep tinkering, finding new ways to use those unique electronic quirks for more selective transformations and clean isolations of target molecules.
Chemists love their shortcuts, and names for this compound reflect that. You might catch references to TFP in synthetic journals or safety sheets—shorthand for 2,3,5,6-tetrafluorophenol. Systematic names highlighting the positions of the fluorines help with clarity, especially for regulatory purposes. Naming conventions can get confusing, especially since other isomers with different fluorine arrangements appear in specialty catalogs. Analogues pop up in pharmaceutical or agricultural patent filings, sometimes with obscure code names, underscoring the broad-based interest from specialty chemical manufacturers all the way to regulatory bureaus tracking toxicology data.
Labs don’t take this material lightly. Experience counts for plenty in handling tetrafluorinated compounds, since even a drop can cause lasting irritation to eyes, skin, or lungs. Most chemists double up on gloves and work strictly in ventilated hoods. Waste handling follows set protocols—solvent rinses and sealed waste bins prevent accidental exposures and environmental release. Facilities that process significant quantities set up dedicated storage and have spill-response kits at the ready. Regulatory standards for reporting and handling hazardous materials apply here; anyone who’s lost lab time to a spill or unexpected exposure knows the cost of underestimating such chemicals. Instead of cutting corners, most research groups establish written handling steps and training for every new user.
Organic synthesis grabs much of the spotlight, but applications for 2,3,5,6-tetrafluorophenol spread out. In my own time on multi-step synthesis, I saw this compound trim hours off purification steps and improve yields for sensitive intermediates. Medicinal chemists prize the phenol for making pro-drugs or fine-tuning the release rates of active pharmaceutical ingredients. Material scientists mix tetrafluorophenol derivatives into polymer backbones to tweak mechanical strength or thermal resistance. Analytical chemists select it as a derivatization agent for GC-MS studies where low background and clean fragmentation matter. Even semiconductor manufacturers look at it for surface treatments that reduce contamination in chip fabrication. Each user group finds some unique upside, whether it’s reliability or specialized performance.
Academic and industry labs keep pushing for greener, safer ways to make and use tetrafluorophenol. Directing research dollars toward milder fluorination methods and recyclable catalysts boosts sustainability and cuts waste. Some groups tinker with enzymatic analogs, looking for bio-inspired transformations that skip strong acids and high temperatures. I’ve seen funding agencies pay close attention to proposals that tackle these bottlenecks, especially with pushback on hazardous reagents and environmental impact. The research output keeps growing: new publications describe milder syntheses, lower waste generation, and even scalable processes for making pharmaceutical building blocks using tetrafluorophenol as a starting point.
Safety reviews keep growing as use expands. Inhalation and dermal contact both trigger acute irritation in exposed workers. Longer-term studies track organ effects and metabolic breakdown. Data shows that the phenolic hydrogen’s increased acidity and the fluorines’ presence change how this chemical interacts with biological systems. Toxicologists look at acute and chronic exposure, studying pathways for organ damage or long-term health risks. There’s a growing collection of rodent studies and occupational monitoring reports. Labs treat even low-level accidental exposures with caution, reinforcing the need for robust PPE and engineering controls. Regulatory bodies have started taking a closer look at cumulative exposure data as production scales up, especially in countries rolling out stricter chemical safety laws.
Demand for clean and tunable building blocks in pharmaceuticals and materials science shows no sign of slowing. Chemists continue searching for ways to improve on old standards, and tetrafluorophenol sits in a strategic position for those chasing incremental gains in reactivity or safety. Better preparation methods—cheaper, milder, and less wasteful—would lower the barrier to broader adoption. As more is learned about potential risks, smarter safety protocols and toxicity studies will help maintain trust between chemists, workers, and the public. For those willing to innovate, subtle changes to the molecular scaffold could unlock whole new classes of catalysts or materials, pushing boundaries where unfussy performance and fluoroaromatic chemistry overlap. The future pays attention to both efficiency and responsibility.
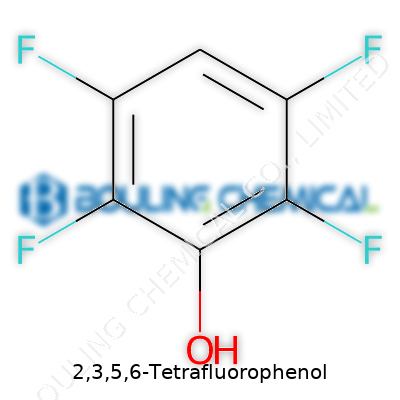
There is something oddly fascinating about how the smallest changes in a molecule can give a compound entirely new abilities. Take 2,3,5,6-Tetrafluorophenol, for example. The name itself might remind people of high school chemistry, sticky benches, and goggles, but its chemical formula, C6H2F4O, holds its own significance far beyond the classroom. Under this arrangement, there’s a single benzene ring, four fluorine atoms snuggled in just the right places, two remaining hydrogens, and one oxygen connected as a hydroxyl group.
I remember the first time I came across a fluorinated phenol late during graduate work. The way this combination of elements changes the reactivity and physical properties grabs you. Adding fluorine atoms to a simple phenol ring isn’t just an exercise in naming conventions. It transforms the boiling point, the solubility, and even how the compound interacts with biological systems. Fluorine is a stubborn element; it digs its heels in and resists reactions, which is both a blessing and a challenge, depending on your goals in the lab.
Ask any researcher why the exact structure and formula matter, and one theme always comes up: predictability. A formula like C6H2F4O gives us a blueprint, which then allows companies, chemists, and even students to infer how this phenol might behave. Several drugs and industrial materials draw inspiration from these patterns. Imagine making a pesticide or a pharmaceutical compound. Altering a single position on an aromatic ring, like this one, sometimes means the difference between a life-saving treatment and an ineffective byproduct.
Working in the lab with fluorinated organics, you learn they often stick around in the environment for a long time. Their resistance to breakdown can serve a purpose—think coatings that never stain or medicines that aren’t digested too quickly. On the flip side, these same traits have led to concerns about bioaccumulation and toxicity. Several fluorine-containing molecules have earned a bad reputation over the past few decades for just this reason. It’s a double-edged sword: the very thing that makes them useful can become an environmental headache once they slip outside intended boundaries.
There’s no magic bullet for taming persistent chemicals. Experience shows that real progress lies in transparency and sharing results. Before every new fluorinated compound launches into the marketplace, conversations about its persistence and effects need to happen. Chemists can get creative about greener approaches by focusing on degradation pathways and using less-pervasive alternatives where possible. Regulations and open publication help keep bad outcomes in check—when everyone can see the risks and benefits, it’s harder for problems to sneak up on us.
C6H2F4O isn’t just an abstract curiosity. Its arrangement on paper means real impacts in the world, and the responsibility sits with us. Anyone who’s spent enough time poring over molecular structures learns to appreciate the ripple effects. Whether for a drug, a coating, or a specialty additive, the meaning behind “2,3,5,6-Tetrafluorophenol” and its chemical formula reaches beyond the lab, touching health, environment, and innovation alike.
Chemistry sometimes feels distant from daily life, but every so often, a compound shows up that makes a real difference beyond the lab. 2,3,5,6-Tetrafluorophenol is one of those hidden players. This molecule, thanks to its unique four-fluorine structure, shows up in several applications that touch everything from pharmaceuticals to specialty materials. In R&D circles, its uses stand out not for flashiness, but for quiet reliability.
Take drug development. Medicinal chemists rely on building blocks that speed up synthesis and help create complex molecules. 2,3,5,6-Tetrafluorophenol gets picked for activating carboxylic acids, forming what are called “active esters.” These active esters turn normally sluggish reactions into efficient processes, bringing down the cost and effort of developing new medicines.
Think about cancer research, where speed and purity drive every stage. By improving the coupling efficiency between peptides and proteins, this fluorinated phenol is part of the quiet machinery making experimental drugs possible, especially those using peptide and protein therapies. Without these connections, some treatments would stay stuck on the bench instead of reaching patients.
Materials science depends on a supply of compounds with unusual stability. 2,3,5,6-Tetrafluorophenol, thanks to those fluorines, resists heat and harsh chemicals. In the world of polymers, it finds its place as a modifier for making materials with impressive chemical inertness and mechanical strength. Electronics companies lean into these types of performance polymers to push boundaries in displays, batteries, and even protective coatings.
Most people never handle this compound directly, but the gadgets and devices showing up in homes and workplaces often benefit from it. As devices shrink in size and raise demands on heat and chemical resistance, fluorinated phenols step forward.
Agricultural labs searching for efficient synthesis routes draw on Tetrafluorophenol’s activating ability. Picture the pursuit of crop protection agents with precise activity and longer lifespans; small changes in chemical structure deliver big differences for farmers. Here, the compound helps shape molecules that stand up to sunlight and tough weather while remaining safe enough to protect the food chain.
Custom synthesis shops keep 2,3,5,6-Tetrafluorophenol on their shelves for making specialty chemicals, dyes, and pigments. The compound’s knack for activating and stabilizing, without leaving unwanted byproducts, keeps it valuable for small-batch projects.
The growing use of fluorinated organics, including 2,3,5,6-Tetrafluorophenol, brings a responsibility to consider environmental and health impacts. Persistent fluorinated chemicals can build up in the environment, raising questions about long-term effects. Handling this compound asks for careful safeguards, from lab protocols to large-scale manufacturing.
Research into safer disposal, greener alternatives, and better containment is starting to ramp up, driven by both regulation and public scrutiny. The best way forward involves real transparency in research and a strong commitment to safety at all stages.
2,3,5,6-Tetrafluorophenol might never become a household name, but its influence stretches across high-impact fields. Its specific chemical features allow scientists and engineers to build solutions faster and better, while also calling for smart choices about environmental risk. How we balance progress and responsibility matters just as much as the next breakthrough.
Anyone who has spent a day in a chemistry lab quickly picks out the quirks of different compounds. Some chemicals catch your nose first, others stain your gloves, but a select few have a character you remember long after the experiment is over. 2,3,5,6-Tetrafluorophenol, a mouthful to say and a bit of a tongue-twister when labeling vials, falls into this last category. It’s more than just another halogenated phenol; its appearance and behavior with solvents set it apart.
Pour a small amount from its bottle and it's clear: this is no run-of-the-mill white solid. Most reputable suppliers stock it as an off-white to slightly beige crystalline powder. It’s not snow-bright, and you may find a subtle tint or change in shade if your sample has hung around moisture for too long. Many in the lab have described the crystals as waxy or flaky—there’s a texture reminiscent of paraffin but with a finer, more fragile break. This fragility makes sense. The presence of multiple fluorine atoms changes the intermolecular interactions compared to a basic phenol, and you’ll spot this difference right away when you handle it.
The way a chemical mixes with common solvents makes or breaks its usefulness in synthetic routes. Tetrafluorophenol has a real identity here—it’s got a foot in two worlds. Compared to phenol, it often feels like an oddity: not shy around organic solvents, but not wild about water. Drop it into ether, acetone, or dichloromethane, and it dissolves smoothly. Chemists are no strangers to chasing after reluctant powders, coaxing solutes with sonication or heat, but here, things go much easier. Water, on the other hand, doesn’t want much to do with it. The compound shrugs off attempts at dissolution in anything too polar, reminding us how replacing hydrogen atoms with fluorine shifts even the basics like solubility.
The structure explains a lot. Four fluorine atoms on the benzene ring create an environment that resists hydrogen bonding with water, even though a hydroxyl group still clings to one side. The carbon-fluorine bond is famous for being stubborn—the most tenacious link in organic chemistry—and this stubbornness shows up in almost every property, including how this compound chooses its solvents.
Some might say this is just trivia, but understanding how and why molecules appear and dissolve matters. I’ve worked through enough failed syntheses to know that a subtle difference like this can turn an experiment from frustration to a clean result. If you know where and how a compound likes to dissolve, you save time, avoid contaminating batches, and sidestep expensive purification steps.
There’s another angle. Increasing demand for specialty fluorinated compounds in pharmaceuticals and agrochemicals keeps chemists reaching for these building blocks. If you handle 2,3,5,6-tetrafluorophenol, recognizing its waxy, flaky appearance and its solubility in organics helps you spot quality at a glance and prepare proper working solutions. Mistakes in understanding these properties can snowball—products fail QC, yields suffer, and research gets delayed.
Building experience with chemicals like this teaches skills beyond the textbook. Pay attention to storage. Excessive moisture can clump the powder and alter its handling. Label and dry it well. Choose solvents not just by what’s at hand but based on a real understanding of the compound’s preferences. Sharing these tips among colleagues keeps the next person from learning the hard way and is how most of us stay one step ahead in a busy lab.
Walk into any lab and you’ll quickly realize safekeeping of reagents isn’t just paperwork—it's a daily habit shaped by a good measure of caution and a few war stories. 2,3,5,6-Tetrafluorophenol, a staple in organic synthesis, brings its own set of challenges and risks. I’ve seen firsthand how ignoring safety with these “ordinary” chemicals leads to extra costs—ranging from ruined batches to evacuation alarms that break your routine and your nerves.
2,3,5,6-Tetrafluorophenol may just look like any other crystalline compound, but it comes with a reputation for volatility and toxicity. Breathing its dust or vapor can irritate lungs and eyes; splashes sting on contact. Accidents often don’t announce themselves with drama—it’s the slow buildup of fumes in a warm storage room or a dusty glove after a rushed transfer where most real-world exposures start. Over time, these mistakes chip away at health and confidence.
Every chemical has its quirks. This one breaks down faster in humid or hot conditions—dropping it near a sink, or tucking it on a sunny shelf, steps up the pace of decomposition. I’ve seen colleagues try to save shelf space by stacking bottles, only to realize later that the heat under the fume hood or next to a radiator can spark problems faster than anyone thinks. A locked, dry, and cool cupboard goes a long way. Moisture-tight containers keep air and water out, so those white crystals stay pure and stable. Simple steps, often skipped out of convenience, prevent ruined stock and sloppy spills.
Many underestimate what good air flow does in the chemical storeroom. A decent fume hood isn’t just a box with fans—it’s the front line when dealing with strong-smelling, sting-inducing chemicals. Years ago, a neglected vent in our storage led to a room full of acrid fumes after a lid went back crooked. A five-minute air flush fixed it, but the lesson was clear: check your hoods and vents, especially after moving bottles in and out. Simple maintenance staves off trouble and keeps everyone breathing easy.
Some roll their eyes at goggles and gloves, but I’ve watched burns and rashes put people out of work for weeks. Nitrile gloves, a thick lab coat, and eye protection cost pennies compared to medical bills and lost productivity. Small habits—like checking gloves for holes or having a spill kit within reach—build a culture of safety that keeps people out of trouble. Accidents show up when someone tires of following the rules, not because the rules are useless.
Experience shows that the best defense is teaching good habits and encouraging folks to ask when unsure. Refresher sessions aren’t just red tape; they stop shortcuts from creeping in and let everyone hear about close calls before they turn into headlines. Safety improvements—from better containers to updated hoods—often come from those who work with these reagents daily. Listening to those voices, and backing up their requests, builds a better environment for everyone who steps into the lab.
With chemicals like 2,3,5,6-Tetrafluorophenol, it's not about overreacting—it's about understanding real risk and addressing it before it becomes a story worth retelling for the wrong reasons. Thoughtful storage, proper gear, and honest training make a huge difference. That’s the lesson science gives, and it pays to listen.
Every few years, another chemical compound makes its way onto the radar of health-conscious workers and researchers. Lately, 2,3,5,6-Tetrafluorophenol – a mouthful, but also an essential intermediate in the pharmaceutical and specialty chemical industries – has started to show up in more laboratories and factories. The rising interest in this compound means more exposure among workers and the environment, which demands a hard look at what that means for health.
People who’ve spent even a little time handling chemicals know that working with phenols never goes smoothly. Tetrafluorophenol belongs to this rough-and-tumble family, known for irritating skin, eyes, and respiratory tracts. Just a drop on unprotected skin can trigger burning, redness, or swelling. Get a splash in your eyes and pain hits fast. Breathing in the vapors for any length of time? Think coughing, sore throat, or far worse, depending on how long you’re exposed.
Back in grad school, a friend of mine forgot his lab goggles for just a minute and paid for it with days of eye pain. Phenols don’t take prisoners. Tetrafluorophenol, thanks to its fluorine groups, seems even nastier. Animal studies for related compounds have suggested potential for liver and kidney harm, and the possibility of damage to the central nervous system with repeated exposure. I’ve seen seasoned technicians develop rashes or complain of nausea after just a brief stint working with substances from the same chemical family.
Some may focus on acute effects – the burning, the blisters, the stinging – but the worries don’t end there. Many phenolic compounds have shown lasting toxicity. Some research papers and regulatory agencies point to chronic hazards such as blood disorders, organ damage, or risks to reproductive health. While there's limited public data about the long-term effects specific to 2,3,5,6-Tetrafluorophenol, it’s reasonable to treat it as a serious threat until more studies offer clearer answers.
Another overlooked angle is environmental risk. Like a lot of synthetic fluorinated chemicals, this one can stick around in waterways and soil. Fluorinated compounds are notoriously tough to break down, and some have earned the label "forever chemicals" for the way they persist in the environment and show up in unexpected places years later. That spells trouble for anyone worried about a clean water supply or food safety.
Real safety with compounds like 2,3,5,6-Tetrafluorophenol begins with straight talk and education. Rushed training or "just-use-gloves" thinking doesn’t cut it. Proper chemical fume hoods, goggles, and gloves made from materials resistant to fluorinated phenols go a long way in cutting down risk. Waste handling needs to be more rigorous, with less dumping and more containment.
Managers need to invest in air monitoring and health screenings for anyone working with these chemicals, not because regulations say so, but because the science points to unknowns that can turn ugly. Substitution with safer alternatives, where possible, should always be on the table. Workers deserve transparency and decision-makers shouldn’t wait for disaster before acting.
Experience says most people learn these lessons the hard way—through mishaps, scares, or trips to the doctor. It shouldn't stay that way, and it won’t if we put in the work to respect chemicals even before every last risk is mapped out.