Looking back at fluorinated aromatic compounds, folks in chemistry circles have always shown a special fascination for how fluorine changes a molecule’s character. 2,3,5,6-Tetrafluoro-4-Methylbenzyl Alcohol didn't crop up out of nowhere. Chemists started tweaking aromatic rings with fluorine atoms in the mid-to-late 20th century, experimenting with their electrical punch and how those tweaks shaped reactivity. Once, these compounds were tricky and sometimes too costly for everyday use. Over time, improvements in synthesis let research groups reel them in at a reasonable scale without bankrupting their budgets. The development of selective fluorination gave birth to molecules like this: robust, odd-sounding, but packed with promise, especially whenever durability and chemical resistance mattered.
Anyone who holds a vial of 2,3,5,6-Tetrafluoro-4-Methylbenzyl Alcohol knows it doesn’t scream for attention. This aromatic alcohol usually appears as a colorless liquid or a solid with a faint whiff, typical of many benzyl-based chemicals. The strategic positioning of four fluorine atoms on the benzene ring leads to a chemical that doesn't just resist attack; it often challenges scientists to dig deep if they want to coax new reactions out of it. This unusual stability under harsh conditions stands out in an industry weighed down by reactive or unstable feedstocks. The methyl group adds a slight twist—nudging its melting point and boiling point, tailoring solubility in select solvents, and giving it just enough hydrophobicity to pivot from traditional alcohol chemistry.
Fluorination shakes up aromatic chemistry more than casual observers expect. Here, the four fluorine atoms pull electron density from the ring, dampening the usual reactivity seen in plain benzyl alcohols. This isn’t just a textbook point; it matters big-time during both storage and use. The compound resists oxidation better than its non-fluorinated siblings, holds steady in the presence of acids, and only lets certain reactions happen. The methyl group sits quietly until someone pushes it with strong bases or oxidizers, and even then, the molecule keeps most of its original backbone. Water doesn’t dissolve it eagerly, while organic solvents like ether and chloroform will take it in gladly. In my lab days, we learned quickly that heating this stuff brought predictable results—no surprising color changes, no strange smells. Only in the company of energetic reagents would it bother to react.
Chemists can't afford confusion, so they look for clear standardization on purity, appearance, and handling. Most reputable suppliers stake their reputation on quantifying purity to 98 percent or higher, flagging any contaminants by gas chromatography. Labels aren’t just stickers—they underline flash points, melting points, and regulatory signals set by groups like OSHA and REACH. People handling these compounds demand real numbers and unambiguous names, not cryptic codes or marketing nonsense. Even storage advice isn’t buried in jargon: keep it cool, dry, capped tight, and out of oxidizing agents. This clarity doesn’t just keep researchers safe; it actually boosts confidence when shifting between labs, regulatory environments, or even between industries.
Making 2,3,5,6-Tetrafluoro-4-Methylbenzyl Alcohol prompts a grin from seasoned organic chemists. Most synthetic strategies rely on selective nucleophilic substitution—replacing hydrogen atoms with fluorine—or reductions starting from the matching benzyl halide. Some clever folks begin with tetrafluorotoluene, introducing a benzyl group with classic Friedel-Crafts chemistry, though often with specialized fluorination agents. In graduate school, I watched a senior grind through repeated vacuum distillations just to get the last trace of acid out, learning quickly that these syntheses test patience and equipment equally. More modern approaches push for greener alternatives, using milder reducing agents or catalytic fluorination to cut down on harsh side-products. Each step shuffles between efficiency and purity—a game every synthetic chemist knows too well.
If the molecule had a catchphrase, it would be “try me, if you dare.” Routine benzyl alcohol oxidations barely register, so anyone seeking derivatives needs to ramp up the stakes with tougher reagents. The fluorines ward off electrophiles and nucleophiles, turning most classic substitutions sluggish or impossible. For those who crack the code, though, neat tricks emerge. Conversions to esters, ethers, or acid derivatives spark new life in the molecule, extending its reach into specialty polymers, agrochemicals, or advanced materials. Over the years, people have tinkered with its core to generate photo-resistant films or robust intermediates in drug synthesis. I’ve seen more than one patent hinge on tweaks to this backbone, the fluorines providing a stability no plain, perhydrogenated cousin could match.
Academic articles toss different names at this molecule: tetrafluoro-methylbenzyl alcohol often comes up, though purists prefer IUPAC's clunky “(2,3,5,6-tetrafluoro-4-methylphenyl)methanol.” Trade catalogs choose clarity, listing only the positions so chemists match the compound to their diagrams. Shorthand slips into casual talk—TFMBA, or just “the tetrafluoro alcohol,” hinting at how insiders save breath in group meetings or whiteboard scribbles. The many names echo its frequent appearance in journal articles and patent documents, often as an intermediate lost in a longer pathway.
Nobody should take safety for granted, no matter how stable a molecule seems. Fluorinated aromatics usually avoid drama—unless mishandled. Most exposure risks involve splashing, inhalation of mists, or skin contact, and so the basic glove-and-goggles routine never feels optional. I remember a colleague who underestimated the volatility—a minor spill became an hours-long clean-up, made more stressful thanks to the low but real risk of hydrofluoric acid formation in the presence of strong acids. Venting, fume hoods, and strict housekeeping matter just as much as knowing your MSDS details. Those in industry pay close attention to off-gassing, container compatibility, and routine inspections. The point: good habits matter most with chemicals that seem unexciting on paper.
Application areas reflect a chemical’s personality as much as any graph or certificate. 2,3,5,6-Tetrafluoro-4-Methylbenzyl Alcohol moves quietly into specialty coatings, the pharmaceutical world, and as a treasured intermediate for manufacturing high-end fluorinated polymers. Drug researchers use it when metabolic stability and electronic characteristics can’t be found elsewhere—often, it's tucked into an early-stage candidate molecule. Materials science labs reach for it when hunting for thermal stability, insight into protein-fluorine interactions, or exotic surface properties. In small-batch settings, it opens the door to new ligands or catalysts, with the fluorines handing synthetic control to researchers who crave predictability.
R&D shapes the molecule’s destiny more than marketing categories ever could. In the past decade, research groups have dived into how these fluorinated aromatics interact with enzymes, metals, or polymers. A flood of publications explores their use in designing molecules that outlast rivals in harsh environments or resist breakdown from light, oxygen, or chemicals. Modern work goes all-in on catalysis—connecting the unique electronic “shielding” from the fluorines to improvements in selectivity, yield, and recyclability. In my own reading, I've come across studies where this alcohol forms the backbone for imaging agents or advanced diagnostics, promising more sensitive detection in the medical field.
Safety assessments need to outpace curiosity. Toxicity profiles for closely related fluorinated benzyl alcohols suggest low acute toxicity for humans, but persistent exposure, bioaccumulation, or breakdown products raise legitimate concern. Over the years, researchers flagged that fluorinated aromatics resist traditional degradation pathways, a problem for both wastewater and long-term human health. Regular workplace monitoring and environmental screening take center stage, and the discussion doesn’t stop at human risk—fish, plants, and microbes all interact in unpredictable ways. I watched one university lab pivot toward greener alternatives after seeing just how hard it was to extract trace residues from soil. Recent animal studies help sharpen safe exposure limits, but the race to predict long-term effects keeps pressing for more data, not just assumptions.
Looking forward, this molecule sits at the crossroads of stability and versatility. Sustainable chemistry sets the pace, and future applications lean on ways to make the synthesis less wasteful and the byproducts more manageable. There’s an appetite for integrating the alcohol into biocompatible materials or as part of smart, responsive coatings in electronics and biotech. New catalysis methods could open uses in processes that today sit behind technical barriers or cost walls. Conversations in research circles revolve around balancing the desire for durability with the stern need to avoid persistent pollutants. The next big leap rests on scientists unraveling safe recycling or upcycling pathways, or even finding biological systems that can accept and break down these tough chemicals. For those invested in safe, forward-looking chemistry, the lesson remains clear: keep pushing boundaries, but never ignore the footprint left behind.
Chemicals with a dash of fluorine draw plenty of attention in both research and industry. 2,3,5,6-Tetrafluoro-4-methylbenzyl alcohol stands out in this crowd. You don't have to work in a lab every day to appreciate how powerful substituting hydrogen with fluorine atoms can be. Just ask anyone who's ever handled non-stick cookware or puzzled at the resilience of modern pharmaceuticals.
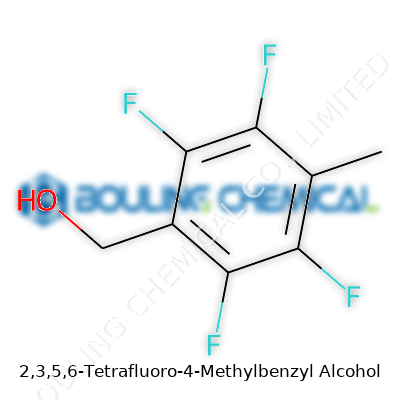
This molecule takes the basic benzyl alcohol backbone, swaps out four hydrogens for fluorines on the aromatic ring, and adds a methyl group. The core is a benzene ring; think of it as a six-carbon ring, an old friend to any chemist. At carbon 1 sits the benzyl alcohol group—CH2OH—jutting out like a limb. Moving through the ring, the 2, 3, 5, and 6 positions feature fluorine atoms tucked in neatly. White-out those hydrogens, stick on some fluorines, and you’ve changed both the physical and chemical game.
These changes aren’t just window dressing. Fluorines bend a molecule’s personality. Water and oils treat it differently. Enzymes in living things don’t break it down the way they might a plain benzyl alcohol. Imagine a lock and key—switch up the bumpy bits, and only some keys fit anymore. Chemists look for ways to dodge breakdown in the body to keep drugs working longer or to help pesticides stick around until pests are gone.
The methyl at the 4-position doesn’t just round out the symmetry. This group can subtly nudge the molecule’s shape, its electron cloud, and its chemical reactivity. Sometimes these tiny tweaks open new doors—a molecule once inactive in a process now jumps into action, or flips from being toxic to benign. Organic chemistry is storytelling at the atomic level. This is a story about tinkering with structure for real impact.
The combination of benzyl alcohol, four fluorines, and a methyl group gives rise to potential uses as building blocks for advanced polymers or medical imaging agents. Fluorinated aromatics show up in pharmaceuticals and agrochemicals, often as the piece that makes the rest of the molecule last longer or perform better. It’s no secret that billions have gone into finding clever ways to lock these unusual structures into large molecules.
Synthetic chemists have always faced a challenge: arranging fluorines in just the right spots without making the process hazardous or wasteful. The market doesn’t reward accidents or runaway costs. Efficient surfactants, specialty coatings, or next-generation drug molecules could all use pieces like this, but only if they can be made safely and responsibly.
Big risks face everyone working with fluorinated molecules. Fluorine chemistry often ends up producing stubborn byproducts that stick around in the environment. Even if a compound works wonders in a lab, careless manufacturing can poison groundwater or expose workers to danger. Green chemistry offers hope here—engineers are searching for pathways that snap in fluorines cleanly, reuse reagents, and leave behind manageable wastes.
What changes things is a commitment to responsible sourcing, transparent data-sharing, and honest evaluation of risks before new chemicals land in the open. Both regulatory agencies and the chemical industry must stop and think critically before diving headlong into every shiny new fluorinated compound.
This is more than academic curiosity. Understanding the structure—and the ripple effects—can make all the difference between a technical miracle and a costly mess. The shape and behavior of 2,3,5,6-tetrafluoro-4-methylbenzyl alcohol offer a revealing glimpse into how real-world impact often lies in the fine details.
2,3,5,6-Tetrafluoro-4-methylbenzyl alcohol rarely makes headlines, yet the world quietly relies on molecules like this one to keep moving forward. My own time in applied chemistry taught me that the smallest tweaks to a molecular structure can open the door to major technological shifts. This compound serves as a testament to that idea. It's not the front-page hero but fills indispensable roles in pursuits ranging from drug research to electronics production.
The pharmaceutical world spends a lot of effort tracking down molecules that deliver quirky, specific properties—and 2,3,5,6-tetrafluoro-4-methylbenzyl alcohol shines in that search. Its particular arrangement of fluorine and methyl groups changes how it interacts with other chemicals. Medicinal chemists look for structures like this because those tiny changes can turn an average prospect into a working medicine by improving stability, adjusting how long a drug stays in the body, or guiding it to the right target. Even a minor component in a synthetic route deserves attention since small differences sometimes mean the difference between a stable, effective treatment and one that falls short. Each functional group plays its part; in this case, the four fluorine atoms often increase resistance to metabolism without bringing extra toxicity.
Fluorinated compounds keep electronics and advanced coatings ahead of the curve, and this benzyl alcohol steps up as a building block for such work. My experience tells me that custom-designed molecules are what fuel the race for ever-smaller, more dependable devices. The unique properties that fluorinated aromatics bring—like low reactivity and high resistance to heat—have made them a mainstay in the search for better semiconductors and display technologies. Whenever the end goal is a surface resistant to both moisture and wear or a piece of circuitry that keeps up under stress, advanced intermediates like 2,3,5,6-tetrafluoro-4-methylbenzyl alcohol set things in motion. Research journals frequently note these structures as scaffolds to fine-tune polymers or modify surfaces for specialized equipment.
Chemistry doesn’t just push medicine and technology along; it quiets fears about global food security too. In the quest to develop new agrochemicals with specific activity and longer shelf lives, chemists lunge for molecules bristling with fluorines. The backbone crafted from this methylbenzyl alcohol can provide a platform for building more selective and durable crop protection agents. I remember watching teams in agricultural labs work through battery after battery of tests to make sure new molecules not only hit their target pests but held up under tough field conditions. Stability, selectivity, and environmental half-life all trace back to starting materials, and fluorinated intermediates keep cropping up as first choice.
Every leap forward in chemicals brings the question of sustainability and safety. The same properties that make fluorinated compounds precious—durability, reactivity resistance—often make them tough to break down once their job ends. Scientists and policymakers need to keep their eyes on research that builds greener methods for both making and disposing of these compounds. Industry partnerships with university labs can smooth out hurdles to recycling and reuse. As more data comes in about bioaccumulation and environmental impact, adjustments in both design and policy should follow.
I remember the sharp, sterile scent of the chemistry lab as my professor handed me a vial of clear liquid. Even then, she stressed, checking purity is as important as knowing what’s in the bottle. In the world of chemicals like 2,3,5,6-Tetrafluoro-4-Methylbenzyl Alcohol, purity isn’t a trivial detail. This small fluorinated alcohol plays a big part in niche synthesis and research. Getting the specs right and keeping contaminants low goes well beyond lab neatness; it keeps research on track and end products safe.
For a compound like this, a purity of at least 98 percent tends to be the norm in research settings. Hitting this mark means less interference from impurities in any further reaction, which directly affects the accuracy and reliability of results. Think of it like following a recipe: adding salt instead of sugar wrecks the cake. Even the smallest impurities can send reactions sideways, giving researchers headaches or dead-ends rather than breakthroughs.
Looking closer, those buying or using this alcohol in the lab rely on supplier transparency. Reputable companies back up their purity claims with data from gas chromatography, NMR, or mass spectrometry. Researchers might double-check with their own instruments just to be sure. The goal is simple: keep the unknowns out, so the science stays strong.
Impurities can mean more than a failed experiment—they can be dangerous. In pharmaceuticals, contaminant traces sometimes react in unexpected ways. Just last year, a widely used chemical intermediate was recalled after an impurity went unnoticed, leading to off-target effects in an early-stage drug trial. These aren’t just theoretical risks. In my own work, a poorly sourced reagent once tanked a month’s worth of effort thanks to a contaminant that wasn’t listed on the data sheet. The lesson stuck: always demand trustworthy data and rigorous controls.
Purity matters not only to the chemist or the pharmacist. It touches the consumer, the patient, or the environmental regulator down the line. Fluorinated compounds often end up in specialty materials, agrochemicals, or pharmaceuticals, where side reactions or byproducts could make their way into the supply chain. Addressing purity from the start reduces long-term problems, recalls, or regulatory headaches.
Practical Steps ForwardThose designing protocols for handling 2,3,5,6-Tetrafluoro-4-Methylbenzyl Alcohol can make headway by requiring certificates of analysis and investing in their own verification tools. Open communication with suppliers builds trust. Finding a balance between cost and quality isn’t easy; skimping on purity might save money up front but will likely cost more in lost time or botched results.
Some labs pool resources to buy analytical equipment, sharing the operational load. This approach helps both new startups and longstanding institutions stretch their budgets and stay confident in the chemicals they use. Over time, the upfront effort pays for itself through avoided mistakes, cleaner data, and safer products.
Purity isn’t just jargon—it’s a guarantee that research, industrial synthesis, or any application depending on a chemical like 2,3,5,6-Tetrafluoro-4-Methylbenzyl Alcohol, stands on solid ground. As science keeps marching forward, vigilance about purity lets discovery happen on purpose, not by accident.
I’ve lost count of how many storerooms I’ve seen with poorly labeled bottles and mystery spills. Chemistry runs on details—both in theory and practice. When you handle chemicals like 2,3,5,6-tetrafluoro-4-methylbenzyl alcohol, the room for error shrinks. This isn’t just some textbook warning; it’s the kind of lesson I picked up both from a formal education and from watching colleagues scramble after overlooking what looked like small risks.
Storing specialty chemicals goes far beyond throwing a bottle on any shelf. With 2,3,5,6-tetrafluoro-4-methylbenzyl alcohol, you’re dealing with an aromatic alcohol that can bring a few hazards to the workbench if it leaks or reacts. This compound should live in a tightly closed container, far from direct exposure to light and heat. Glass bottles with proper seals beat plastic every time, especially if the substance has the potential to soften or degrade common polymers over time. I’ve seen old stoppers eaten away in as little as a year, leading to dangerous surprise leaks.
I’ve worked in shared labs where acids, bases, and reactive alcohols ended up on the same shelf out of laziness or because someone ran out of room. Here’s what matters: 2,3,5,6-tetrafluoro-4-methylbenzyl alcohol doesn’t belong next to acids, oxidizers, or open reactive metals. The risk of unwanted reactions goes up with proximity. You can learn this the easy way—by following common-sense segregation—or the hard way, which usually involves an evacuation and a rush for the spill kit.
Moisture in the air can quickly change the safety profile of many chemicals. Experiences in overly humid storerooms have shown me how labels fade, caps corrode, and crystals sometimes appear inside containers. Dry storage helps avoid all sorts of headaches, especially if you include a desiccant packet inside the storage enclosure. For volatile materials, storing the chemical under nitrogen or another inert gas adds a layer of protection, particularly if you keep the bottle around for years at a time.
Labeling seems obvious, but some of the most dangerous mix-ups I’ve seen stem from sloppy handwriting or missing hazard information. I’ve walked through labs where the storage standards get thrown out as soon as things get busy. Following basic rules—clear labeling, up-to-date chemical inventory, routine checks—saves time, product, and sometimes your safety. Nothing beats a routine scan of storage conditions, especially after power outages or building maintenance. If the fridge or storage cabinet goes down, you want your chemicals accounted for and undamaged.
No one likes paperwork, but a chemical safety protocol makes everything easier if something happens—spills, leaks, or surprises during inventory checks. Real accountability grows from repeated habits, not from reading a policy once. At its core, storing 2,3,5,6-tetrafluoro-4-methylbenzyl alcohol means keeping others safe, respecting the material, and not taking shortcuts, especially as older bottles sit for years waiting for just one more reaction scheme.
Anyone who’s spent time working with chemicals in a research lab or production setting knows that every compound has its own personality. Some give off nasty fumes, some burn through gloves, and others just love to surprise you with hard-to-clean spills. 2,3,5,6-Tetrafluoro-4-Methylbenzyl Alcohol isn’t the flashiest chemical on a shelf, but treating it with the attention it deserves can keep science and scientists healthy.
This compound, like many organofluorine derivatives, isn’t the kind of thing to splash around without a plan. Breathing in the dust or vapor often leads to irritation in the nose or throat. Getting it on your skin isn’t a better deal, either. In my experience, once your skin starts itching from a bit of solvent or an alcohol derivative, you only need that lesson once. So, taking a few moments upfront to avoid messy mistakes saves hours of regret later.
Long sleeves, goggles, and gloves aren’t overkill—they’re the basics. Think about wearing a lab coat as a lightweight insurance policy; a few seconds to button up can stop a world of hassle down the line. Nitrile gloves handle most exposures, but I always check that they haven’t started thinning or tearing. Simple slips open the door to more dangerous accidents. And, frankly, washing hands after leaving the bench is an easy habit that pays off every time.
No one likes to work in a cloud of chemical vapor, fluorinated or otherwise. A properly running fume hood is the backbone of safety here. I’ve seen folks try to cut corners, working outside the hood “just this once.” That gamble often ends badly, with surprise headaches or trips to the health office. Hoods catch spills and keep unwanted fumes out of your breathing space, which is a luxury more than a rule. If you catch even a whiff of chemical scent, it’s a sign that something’s off—stopping work and figuring out why always trumps pushing through the smell.
Leaving bottles on a cluttered bench only takes one elbow bump to turn tidy labs into drama zones. Sealing up stock bottles and keeping them in ventilated, clearly labeled cabinets makes returns easier and reduces the odds of a mix-up. Light, heat, and moisture all work to degrade sensitive fluorinated compounds. Extra vigilance on where and how bottles rest between uses ensures safer chemistry and more reliable results.
Spills get everyone’s heart rate up, even seasoned chemists. Tackling a spill with a cool head and the right kit—like absorbent pads, a sturdy broom, and the willingness to ask for help—keeps small accidents from growing into big stories. For waste, nothing beats clear labels and dedicated containers. Complacency around chemical waste has haunted everyone who’s swept floors in research spaces, leading to costly disposal or even bigger hazards. Regularly scheduled waste pickups and training on disposal rules let science march forward without unnecessary risks.
Safety slips rarely come from ignorance. Most often, they grow out of shortcuts, tiredness, or the pressure to finish a long experiment. Open conversations about near-misses and realistic, lived advice help everyone get home the same way they arrived—healthy and ready to tackle another day of challenging chemistry. Putting care into every step, not just textbook compliance, turns the dangers of handling 2,3,5,6-Tetrafluoro-4-Methylbenzyl Alcohol into manageable parts of a rewarding career.