Chemistry textbooks rarely lift the hood on the people and stories that drive the field forward. The backstory of 2,3,5,6-Tetrafluoro-1,4-Benzenedimethanol traces the rise of organofluorine chemistry. In the early days, adding multiple fluorine atoms to an aromatic ring took plenty of patience and some desert-dry humor. Fluorination started as a curiosity in labs chasing new ways to toughen up materials or rethink drug scaffolds. This specific tetrafluorinated aromatic diol came along in the late twentieth century, delivering a mix of stability, reactivity, and – for the seasoned chemist – a touch of pride at getting the synthesis right.
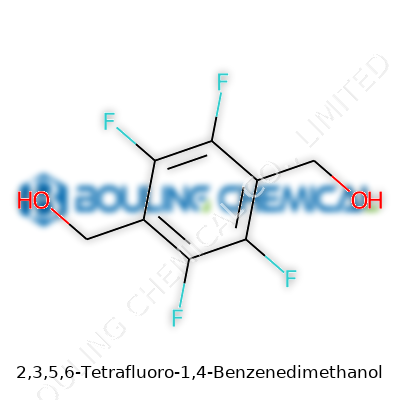
2,3,5,6-Tetrafluoro-1,4-Benzenedimethanol—rolls off the tongue about as easily as you’d expect—belongs to the family of aromatic diols. Picture a simple benzene ring, swap out four hydrogens for fluorines, and bolt on two alcohol groups at the para positions. Folks in the lab often call it by its abbreviated label, TFBM, or dig up synonyms like 2,3,5,6-tetrafluoroterephthalyl alcohol. Most publications stick to its IUPAC name, if only for the pomp of it.
You expect a tetrafluorinated compound to shun water, and TFBM delivers. As a solid at room temperature, it sits white to off-white, with a notable melting point that gives it shelf-stability. The fluorine atoms pull electron density, toughening up the aromatic ring and making TFBM more resistant to oxidative breakdown than plain para-phenylenedimethanol. These traits pull in folks working in specialty polymers, coatings, and high-performance materials. Chemical resistance comes as no accident; even strong acids and bases have a harder time bullying TFBM into submission compared to non-fluorinated analogues.
Suppliers tend to ship TFBM with high purity, usually over 98% by weight, and stress the control over moisture content, given that even a hint of water can throw off precise polymerization. There’s a need to lock it down from atmospheric moisture—fluorinated benzenes like to play it safe. Often, product labeling includes chemical structure diagrams for those who think visually, as well as CAS numbers and warnings about storage. As with all organofluorines, long-term stability hinges on avoiding strong nucleophiles during storage.
Syntheses rely on the selective introduction of fluorine atoms and the subsequent attachment of the alcohol functionalities. In labs, strategies split between direct fluorination and more polite halogen-exchange methods, given the hazards associated with fluorine gas. Most reliable methods begin with a suitably substituted aromatic precursor—think tetrafluoroterephthalic acid—and reduce it to the desired dimethanol. Catalytic hydrogenation usually steps in, but not without calling for exacting conditions and robust safety measures. Anyone with hands-on experience knows the unpredictability of aggressive fluorinating agents, and nobody misses the thick gloves and fume hood dances.
TFBM doesn't just sit around on a shelf. Its diol groups offer points for further functionalization. Researchers covalently link it into polyester chains, polycarbonates, or hop to more esoteric custom polymers. Introducing TFBM into a step-growth polymerization matrix can jack up the thermal and chemical resistance, handy for niche engineering plastics. The fluorine-rich ring acts as a tough core, while the alcohol groups bridge connections or open up to crosslinking reactions. A chemist’s toolbox brims with surefire reactions—esterifications, ether formations, and various coupling routines build complexity off this sturdy backbone.
Clear terminology pushes back against confusion. TFBM often appears under alternative names in specialty catalogs: tetrafluorinated 1,4-benzenedimethanol, 2,3,5,6-tetrafluorobenzene-1,4-dimethanol, and a few less common ones. Folks writing grant proposals tend to lean on the IUPAC label, while industry might shorten for convenience. This jumble of synonyms sometimes causes headaches searching databases, a reminder to cross-check any references.
Fluorinated aromatics demand respect, especially during syntheses. Liquid nitrogen traps, explosion-resistant shields, and precise ventilation come with the territory. TFBM’s toxicity profile doesn’t totally match up to some nastier fluorinated aromatics, but the dust or vapors can still get under the skin—literally and figuratively. Standard lab practice means gloves, goggles, and a fume hood at minimum, plus rigorous training on chemical spills or unexpected decompositions. Waste handling follows protocols for organic solvents and fluorinated residues, with waste streams often sent for specialized incineration. If experience teaches anything, it’s not to take shortcuts with fluorinated intermediates.
Industry wants materials that hold up against heat, solvents, and environmental stress. TFBM fits into this narrative, often woven into high-end polymer systems or as a building block for liquid crystal materials and advanced fibers. Specialty coatings drawing on TFBM resist scratches and chemical attack, which matters if you’re building labware, protective apparel, or display screens. Some labs look to TFBM for next-generation proton exchange membranes, hinting at a role in energy storage or conversion. The reach extends to certain pharmaceuticals, though the rarity of widespread use keeps it a specialty rather than a staple.
Labs keep tinkering. Emerging research focuses on customizing the physical characteristics of polymers by integrating more fluorine atoms into the matrix, and TFBM answers the call for high-performance materials. Academic groups present papers on crosslinking new resin systems for electronics, or try to engineer membranes that withstand extreme pH or temperatures. Focused research explores modifying TFBM to improve compatibility with other monomers, fine-tuning solubility or crystallinity for specific processes. Real innovation involves rolling up sleeves, running small-batch syntheses, and failing forward, learning from unexpected results.
Toxicity doesn’t get sorted by luck. Not every fluorinated aromatic causes immediate harm; TFBM sits somewhere in the middle of the risk spectrum. Studies suggest the molecule resists rapid breakdown, which means environmental persistence comes into play. Animal experiments highlight possible routes of exposure, particularly inhalation of powders or skin contact with solutions. Regulations push researchers to minimize releases to the environment, since long-lived organofluorines may bioaccumulate. Some labs now track worker health and waste streams with a seriousness that wouldn't have turned heads a generation ago. Lessons learned from persistent organic pollutants shape how TFBM gets handled—no one wants a repeat of past mistakes involving fluorine-rich compounds.
Future directions swirl around functionality, sustainability, and regulatory pressure. Many researchers hunt for alternative routes to TFBM that dump fewer hazardous byproducts or use greener reagents. Material scientists push to combine performance gains with easier recycling or eventual degradation. Data keeps stacking up around the environmental footprint of organofluorines, prompting some groups to look for biodegradable analogues. But the unique properties that TFBM offers—resilience, stability, and reactivity—underscore its continued relevance in specialty areas. The balance between innovation and safety, performance and environmental stewardship, defines the path forward. Chemists who understand the storied past of TFBM, not just its clean sheets and datasheets, shape how it will fit into the toolbox of tomorrow.
Most folks would never hear about 2,3,5,6-Tetrafluoro-1,4-Benzenedimethanol unless they work in a chemistry lab or in the manufacturing world. But this specialized molecule punches far above its weight in the big machinery of innovation. Its value starts with the unique structure: a benzene ring, four fluorine atoms, and two alcohol groups. That precise arrangement gives it a unique blend of strength, stability, and reactivity.
The core use for this chemical is in creating higher-performing plastics and resins. I’ve seen its effects most sharply in the electronics sector, where reliability often depends on materials that can handle stress, temperature swings, and exposure to all sorts of nasty agents. The fluorine atoms packed onto this molecule do heavy lifting—turning ordinary polymers into compounds that handle the pressures of cutting-edge gadgets or advanced industrial equipment.
Take liquid crystal displays. The push for slimmer and sharper screens means engineers need materials that don’t warp under heat or fade when hit with UV light. By incorporating this molecule into their backbone materials, manufacturers boost both strength and chemical resistance. I’ve met engineers in display manufacturing who credit tweaks like this for helping make screens last longer and perform better in everything from smartphones to medical monitors.
Another field that benefits is the world of specialty coatings. Fluorine-rich coatings made with this ingredient help keep surfaces non-stick and corrosion-resistant. If you’ve ever wondered how some industrial pipes keep running year after year with barely a dent or a rust spot, part of the answer lies in tough, engineered coatings that start with chemicals like 2,3,5,6-Tetrafluoro-1,4-Benzenedimethanol.
Then comes the role in making advanced adhesives and sealants. Regular glues fail quickly in demanding settings. Add a touch of this molecule, and the result is a stronger bond or a joint that doesn’t turn brittle over time. From aerospace applications to critical electronic assembly, dependable adhesives translated into real safety and reliability. On a more personal note, knowing that engineers obsess over every molecule in a car’s electronics or an aircraft’s cockpit has always made me feel a little safer on long trips.
This chemical’s features support the growth of sustainable manufacturing, too. For example, its high chemical resistance opens the door for products that last decades rather than years, helping reduce waste. Choosing materials that perform longer and require less frequent replacement offers a path toward reducing the environmental toll of rapid product cycles.
Of course, nothing’s free of tradeoffs. Handling and sourcing specialty chemicals can create challenges: cost, supply chains, and even health and safety risks for workers. The push for greener chemistry keeps researchers searching for ways to deliver the same performance with smaller footprints and fewer hazards.
What’s clear is that 2,3,5,6-Tetrafluoro-1,4-Benzenedimethanol enables tangible progress in electronics, coatings, and advanced adhesives. Investing in smarter chemistry—sometimes a tweak at the molecular level—translates into more reliable products and longer-lasting technology. It’s a reminder that big leaps often come from less-glamorous building blocks, quietly making our lives more efficient, resilient, and sustainable.
Looking at the name 2,3,5,6-tetrafluoro-1,4-benzenedimethanol, you can already sense some complexity. The chemical formula, C8H6F4O2, carries two methanol groups attached to the benzene ring, with four fluorine atoms pinned at predictable positions. In practical terms, this molecule is a mouthful and a deep dive into applied organic chemistry. From the start, the arrangement of these fluorine and hydroxymethyl groups shapes nearly every trait of the molecule—from how it dissolves to how it reacts with other chemicals.
Fluorinated benzene derivatives bring a certain edge to the table. Those four fluorines sitting across the aromatic ring do more than just decorate the skeleton. They pull electron density inwards, making the ring less reactive toward traditional substitution reactions—almost like turning down the noise in a crowded space. Adding methylol groups on the 1 and 4 positions turns this from a standard benzene variant into something that can hold its own in serious industrial and research uses.
In my experience with lab syntheses, getting a dialkyl benzene where a handful of sites are fluorinated means the resulting compound isn’t just chemically tougher—it stands up against strong acids and bases better than its plain cousins. That makes it a good foundation for polymers or specialties like high-performance polyesters, where stability under heat and resistance to breakdown count for a lot.
Engineers and chemists see tetrafluorinated benzenes as an entry point to high-end plastics. Take the construction of certain copolymers—replacing part of the backbone with something as robust as 2,3,5,6-tetrafluoro-1,4-benzenedimethanol can cut down on unwanted discoloration and improve water repellency. In applications where repeated sterilization or strong cleaning agents are common, fluorinated aromatics find themselves right at home.
On the flip side, issues with fluorinated aromatics have been cropping up in toxicology circles. Perfluorinated compounds, for example, have caught flak for environmental persistence and bioaccumulation. So while this molecule packs a punch for scientists and engineers, workers in the field pay close attention to safe handling, reduction of emissions, and disposal methods. Synthetic chemists have been hunting for greener routes all the while.
The long story of benzene derivatives swings between major discoveries and safety missteps. Fluorine’s special bond in these molecules delivers performance—and sometimes headaches. Regulatory momentum continues pushing chemical industries to reexamine life-cycle impacts. Academia, industry, and regulatory agencies share a duty to assess safety, share data transparently, and refine production. There’s healthy skepticism about how long these substances stick around in soil and water. Trust grows only from open discussion, smart policy, and clear evidence.
Chemistry runs in cycles, learning from old mistakes and scoring new wins. Molecules like 2,3,5,6-tetrafluoro-1,4-benzenedimethanol stand as a testament to how precise atomic tinkering can change what’s possible. They don’t exist in isolation. Each advance rests on a foundation of responsible research, environmental stewardship, and public engagement—grounded in both caution and creativity.
Most people glance past the ingredient lists on medicine bottles, cleaning sprays, or packaged snacks. The properties that shape these products—like melting point, solubility, and reactivity—aren’t just boring textbook trivia. They drive real-world impact, steering whether an over-the-counter medicine works safely, or if a detergent leaves skin unscathed. Years back, I worked with a team tweaking a common pain reliever. Our job was less about the formula’s active ingredient and more about how the finished compound dissolved. If something released too fast, it meant harsh side effects. Too slow, and pain stuck around. Hundreds rely on subtle tweaks in property to feel better.
Think about solubility for a moment. Take a drug that needs to dissolve quickly so the body can use it. If the compound’s solubility misses the mark, the drug might not hit your bloodstream in time. Aspirin offers a textbook case. It’s reasonably soluble in water, which means those little white tablets work rapidly. If you stumble onto a compound that clumps in water, chemists either go back to the drawing board or look for additives to rescue its performance. This isn’t just for drugs. Detergents, flavorings in food, even pigments in plastics depend on predictable, tested properties.
Instability sounds technical, but it lands with real consequences. Not long ago, a new food preservative caught my eye. It promised better shelf life, but its chemical stability couldn’t deal with light exposure. So snacks with this additive turned rancid under grocery store lights. If the compound could bond easily with oxygen, the food would spoil. We see similar issues in paints. A pigment that fades under sunlight spells disaster for homeowners or auto manufacturers. Knowing how a compound stands up to air, light, moisture, and heat forms the backbone of reliable consumer goods.
Diving deeper, volatility makes headlines in the world of fragrances and cleaners. High volatility means a compound evaporates quickly, giving air fresheners their punch—but also deciding how safe and sustainable the product is in daily spaces. If you’ve ever used a cleaner and felt your nose burn, a volatile compound likely played a role. Managing evaporation not only fine-tunes product performance but also drives health concerns, especially for asthma sufferers and children.
Properties like toxicity aren’t just abstract measurements. My uncle worked in facilities that stored industrial chemicals. He’d joke that folks needed more “nose” than a scientist—one whiff off meant more than a thousand spreadsheets. Hazards often come from simple misunderstandings of a compound’s properties—mixing one with another, or storing it at the wrong temperature. Latex gloves, safe handling procedures, and tight labeling rules owe thanks to teams that carefully checked each compound’s reactivity, flash point, and the risk of forming hazardous byproducts.
Progress keeps coming. Today, research communities and manufacturers team up to find greener chemicals with less environmental baggage. Developing compounds with better biodegradability or less tendency to build up in the food chain turns into global action—from home cleaning aisles to pharmaceutical labs. Open science and transparent data sharing mean experts spot dangerous trends sooner. Mistakes from the past, like leaded gasoline or toxic dyes, nudge each generation to balance performance with health and sustainability.
Many folks outside chemistry circles haven’t heard of 2,3,5,6-Tetrafluoro-1,4-Benzenedimethanol. It’s a specialty ingredient, often found flowing through labs or used in niche industrial processes. If you’ve ever worked with chemical research or dabbled in polymer science, you might already know about its role in specialty plastics or coatings. But when a compound like this starts popping up, people ask the right question: Is it hazardous? How do you handle it safely?
Walk through any well-run laboratory. Safety data sheets line the walls, and scientists wear gloves that match the threat of each project. Chat with a seasoned lab technician, and most will tell you: You don’t treat new fluorinated compounds lightly. That’s no overreaction. Many fluorinated organics can slip through normal skin barriers or deliver unexpected toxicity. Some can catch fire if stored near the wrong substances. Even in small amounts, vapor or dust can irritate eyes or lungs. My own work around similar compounds taught me quick respect. Sometimes, you can’t even smell or see the exposure happen. You just feel it later—nagging headaches, raw skin, unpredictable cough.
Dig through the literature and safety reports on 2,3,5,6-Tetrafluoro-1,4-Benzenedimethanol. You’ll notice words like “irritant” or “potential hazard to respiratory tract.” Inhalation and contact risks stand out. Those warnings are not just there for regulatory compliance. They save careers and livelihoods.
Occupational stories have a way of hammering these lessons home. A colleague once forgot gloves moving a container of a similar diol. Pink, stinging hands. Chemical burns that lingered for days. She never made that mistake again. Chemical compounds with fluorine substituents tend to work their way into tissues, meaning skin contact can deliver more of a punch. There’s also the ever-present risk of accidental spillage—onto clothes, benches, or worse, open wounds.
I’ve watched shop supervisors call out young researchers for open-toed shoes or missing eye shields. It can feel excessive until that one spill splashes up, and then everybody’s grateful for a tough old mentor looking over their shoulder. The most advanced chemical workflows fall apart in seconds without basic respect for hazard controls. Gloves rated for organics, splash-resistant eyewear, and regular hand washing become non-negotiable in real labs.
Some folks think only those who handle vats of industrial chemicals face real danger. Truth is, even milligrams of a misunderstood substance can cause harm. Proper labeling, clear ventilation, and training at every step help avert big problems. Storage matters too. Not sitting next to acids or bases that could trigger unexpected reactions. Disposal channels for fluorinated waste, handled through hazardous-waste programs, keep these compounds out of the trash and away from community water supplies.
If we respect the risks, we protect ourselves and those down the line—from colleagues to sanitation workers. Being cautious isn’t about slowing down progress. It’s about keeping families, careers, and communities safe in a world where advanced materials show up everywhere from smart coatings to designer electronics. Listening to the science, building solid habits, and sharing stories from those who have handled these compounds keeps everyone out of the emergency room and focused on discovery.
Chemicals like 2,3,5,6-tetrafluoro-1,4-benzenedimethanol have a way of reminding us that a little caution can go a long way. I’ve spent enough time in the lab to know that the fastest way for things to go wrong comes from letting routines get sloppy. This compound isn’t some run-of-the-mill solvent. Its molecular structure packs in four fluorine atoms, making its toxicity and reactivity real concerns for anyone who even thinks about using it in a project.
Safe storage comes down to a few basic rules: keep it cool, keep it dry, and keep it away from anyone with no business handling it. Nobody wants accidental exposure—think skin or eye contact, or worse, inhalation—so a tightly sealed container, labeled clearly, finds its place in a ventilated chemical storage cabinet. Nobody stores water-reactive or incompatible substances in the same space; that’s asking for reactions you can’t take back.
Talk with any chemist worth their salt, and you’ll hear about the frustration that comes from picking up a mis-labeled or poorly stored reagent. Data trips everyone up when safety slips. Temperature swings can mess with chemical stability, leading to dangerous breakdown products. And if you’re reaching for a bottle you haven’t checked in months, odds are it’s not in the same shape you remember. Old labels fade. Containers crack. These small details add up to a major hazard if left unchecked.
Anyone planning on working with 2,3,5,6-tetrafluoro-1,4-benzenedimethanol needs personal protective equipment. Gloves, eye protection, and lab coats form the first line of defense. I’ve seen folks skip the basics, only to regret it later after a splash or spill. Running work in a fume hood keeps vapors away from your nose and lungs. Simple habits, like never pipetting by mouth (yes, people still try it), mark the difference between rookie mistakes and professional work.
Working in teams with a safety-first mindset makes a difference. If someone sees something off—a container left open, a suspicious smell—speaking up keeps people healthy. It’s not about paranoia; it’s simply experience paying off in real time. Safety showers and eyewash stations nearby aren’t window dressing. I remember one moment of panic when a colleague got splashed with a strong acid. Without a working eyewash station, the story wouldn’t have had a happy ending.
Proper disposal proves just as important as storage or handling. Dumping chemical waste down the drain or mixing unknowns in a flask invites trouble. Waste containers, marked with chemical constituents, fill up fast in a busy lab, but that’s a much better outcome than risking environmental damage or chemical burns. Regulations around fluorinated compounds keep getting tighter, so falling behind on compliance isn’t just risky, it’s expensive.
For any lab or facility storing this compound, regular audits and training pay off. Whether you’re new or have decades under your belt, complacency never helps. If something feels off, it usually is. Staying up to date with material safety data sheets, keeping emergency contacts visible, and running drills can make the difference between a close call and a news story.