Materials that have multiple fluorine atoms attached to a benzene ring didn't enter labs and chemical supply stores on a whim. The path of tetrafluorinated benzyl alcohols began with the broader rise in interest toward fluorinated organics during the latter half of the twentieth century. After DuPont's discoveries that linked organofluorines to valuable refrigerants and polymers, the next wave of curiosity hit synthetic chemists eager to test how else stable C–F bonds could shift reactivity and durability in organic molecules. As a result, compounds like 2,3,4,5-tetrafluorobenzyl alcohol slowly worked their way from niche intermediates in specialized papers to larger commercial catalogues. This expansion didn’t just stem from profit but from curiosity about what fluorine substitution can really do to common structural motifs in synthesis, pharmaceutical research, and materials science. In my own route through synthetic organic labs, I watched as academic and industrial chemists began tinkering with fluorinated benzyl derivatives, not only as curiosity projects but as essential parts of wider studies around drug scaffolds and new materials.
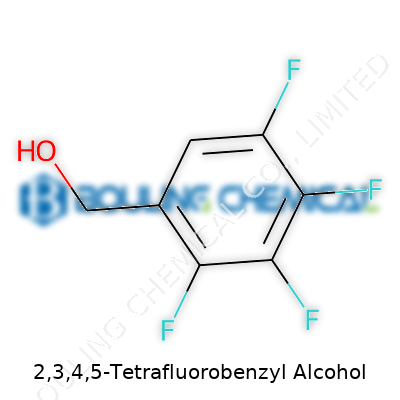
2,3,4,5-Tetrafluorobenzyl alcohol stands out for its structural simplicity and utilitarian edge. It holds a benzene ring clothed in four fluorines, leaving one arm open as a benzylic alcohol. Some may ask, why does a fluorinated benzyl alcohol matter? It turns out adding multiple fluorines on a benzene ring doesn't just toughen up the backbone—it fundamentally shifts the reactivity, boiling point, and even the way it smells and feels to handle. This molecule becomes a gateway to learning how heavy electron-withdrawing groups can nudge ordinary alcohols to act in extraordinary ways during synthesis. Chemists appreciate how modifying single atoms in an otherwise common structure can push the limits of what certain reaction pathways might tolerate, and what functional groups can survive harsh environments. So, 2,3,4,5-tetrafluorobenzyl alcohol isn’t just another point in a catalog; it represents a practical intersection of academic curiosity and real-world application.
People who have poured and weighed 2,3,4,5-tetrafluorobenzyl alcohol see the difference between this compound and its non-fluorinated kin. Tetrafluorination of the benzene ring suppresses its aromaticity, gives it a higher density, and pushes its boiling point higher than benzyl alcohol without substitution. The presence of four fluorine atoms renders the molecule more lipophilic and at the same time, tougher against metabolic or oxidative attack. Most pure samples present as a colorless to slightly yellowish liquid, modestly viscous. The compound’s resistance to many common degradation pathways opens it up for challenging conditions, like those needed for medicinal chemistry scale-ups or tough industrial polymerizations. From a safety perspective, that same chemical stability means storage containers don't yellow or build up residue as quickly as with less robust benzylic alcohols.
Standard reference sources such as peer-reviewed chemical journals and safety data sheets highlight the precision needed when labeling and quantifying fluorinated alcohols. Consistent naming, batch-specific purity data, and clear hazard identification remain non-negotiable standards if you want to avoid confusion in the lab or miscommunication with downstream users. In academic and pharmaceutical contexts, purity often exceeds 98 percent, with trace impurities tightly screened by NMR, GC-MS, and high-performance liquid chromatography. Because of the toxicity risks associated with fluorinated benzylic compounds, handling protocols are noted in detail, reminding chemists to use gloves, protective clothing, and proper ventilation at all times. Many researchers track their stock and usage logs closely, since even minor cross-contamination can throw off a sensitive biological assay or lead to complications in later synthetic steps.
Taking a closer look at the ways 2,3,4,5-tetrafluorobenzyl alcohol is made offers a case study in modern synthetic ingenuity. Early attempts to attach four fluorines to a benzene ring required harsh direct fluorination, but safer strategies soon emerged. The more reliable approach involves starting with a tetrafluorinated benzene derivative and introducing a benzylic alcohol motif through lithiation, formylation, and reduction processes. Skilled preparative chemists often turn to directed ortho-metalation using specialized bases, then take advantage of the ring's increased electrophilicity to install the alcohol-bearing group with high selectivity. Through trial and error, process chemists eliminated side reactions that plagued initial protocols, gradually moving toward higher yields and fewer undesirable byproducts. It’s genuinely satisfying to watch older, risky chlorofluorination reactions replaced with milder options that keep yields up and hazards down, especially in crowded shared facilities.
Once you have a well-characterized batch of 2,3,4,5-tetrafluorobenzyl alcohol, it quickly turns into a workbench for chemical creativity. Standard functional group interconversions—oxidation to aldehydes or acids, protection-deprotection strategies, nucleophilic substitutions—take on new twists in the presence of four electron-withdrawing fluorines. Reductive and oxidative conditions that easily chew up less stabilized benzylic compounds often leave this one standing. Medicinal chemists have explored how the tetrafluorobenzyl motif resists metabolic breakdown compared to its mono- or difluoro relatives, which can translate to longer-lived candidates in drug action studies. Coupling protocols like Suzuki or Buchwald-Hartwig take advantage of the ring’s altered reactivity profile, allowing for cleaner cross-coupling outcomes. Contrastingly, the fluorines occasionally complicate lithiation or nucleophilic aromatic substitution, which demands thorough planning and characterization. For students and researchers alike, working with this compound provides an education in how ring substitution steers chemistry far more than textbooks can convey.
Names pile up quickly in the world of specialized organics, causing headaches for both veterans and newcomers who try to keep records or search literature. 2,3,4,5-Tetrafluorobenzyl alcohol may appear as "TFB Alcohol" or "tetrafluorinated benzyl alcohol" in some resources. Less often, suppliers give it catalog codes that hide its identity behind numbers, which slows down literature searches and increases the risk of costly misorders. Using proper IUPAC naming and checking CAS numbers helps keep everyone on the same page. In academic writing and patent filings, sticking with standardized chemical names has prevented more than a few headaches.
Anyone who has worked with fluorinated organics recognizes the need for caution. Even as 2,3,4,5-tetrafluorobenzyl alcohol’s boiling point and low volatility make it less prone to accidental vapor exposure than lower-molecular-weight analogs, direct skin contact or inhalation can have negative effects, including irritation and, in rare cases, systemic toxicity. Established operational procedures stress eye protection, use of chemical fume hoods, and avoidance of open flames due to the compound’s tendency to decompose at high temperatures, releasing corrosive fluorinated gases. Waste disposal routines can't cut corners either; improper discarding of fluorinated organics leads to environmental harm, as fluorine-rich compounds resist breakdown in natural settings. The tight regulations around fluorinated chemical handling come not only from agency rules but from the collective experience of researchers—hard lessons from past mishaps underlining the importance of meticulous practice and regular safety audits within the lab environment.
Organic synthesis, materials science, and pharmaceutical research have all found uses for 2,3,4,5-tetrafluorobenzyl alcohol. In my laboratory years, I encountered this compound most frequently as a synthetic intermediate: either as a way to introduce the tetrafluorobenzyl group into more complex frameworks, or as a probe for studying electron-deficient arene reactivity. The pharmaceutical sector invests in these compounds because fluorine atoms can dramatically improve a molecule's stability in human metabolism, often extending the drug's effective half-life. Material scientists have shown that incorporating tetrafluorobenzyl groups changes the electronic and mechanical performance of polymer backbones, especially in advanced coatings or specialty membranes. Researchers tend to get excited when these modifications lead to measurable gains in chemical resistance or tunable surface properties. Analytical chemists use it as a derivatizing agent to improve the detectability of certain analytes, transferring the benefits of the fluorinated tag to biological and environmental assays.
Research on 2,3,4,5-tetrafluorobenzyl alcohol rarely stands alone. Instead, it slots into broader questions about how polyfluorinated aromatics behave in living systems, how well they can carry drugs through the bloodstream, and how strongly they affect the flow and resilience of coatings or specialty plastics. Academic groups, especially those vested in pharmaceutical lead optimization, run structure–activity studies with the tetrafluorobenzyl motif to see whether the heavy fluorine load translates into real performance gains. Companies exploring next-generation materials cast a wide net—testing this and other fluorinated intermediates in new adhesives, electronics substrates, and even in energy storage. Nearly every R&D team with a foot in organofluorine chemistry keeps their eyes open for new synthetic tricks that can clean up side reactions, boost yields, and cut down hazardous byproducts. Over the years, these projects have improved operational efficiency, making the production of such advanced intermediates less expensive and more consistent.
Toxicological data on highly fluorinated benzylic compounds keeps expanding, given their growing use in pharmaceuticals and materials. Repeated studies point to the need for caution, as these compounds, including 2,3,4,5-tetrafluorobenzyl alcohol, resist breakdown, potentially accumulating in tissue or the environment. Acute toxicity in mammalian systems tends to be lower than for smaller, more volatile fluorinated aromatics, which doesn’t translate to safety if exposures persist or doses increase. Regulatory agencies maintain strict upper limits on workplace air and surface concentrations because of concerns about long-term health impacts, especially in poorly ventilated or low-oversight settings. My colleagues in environmental toxicology found that quantifying and minimizing accidental discharge from factories can reduce downstream contamination risks. Communication between chemical manufacturers, users, and regulators built the current foundation for worker and environmental safety—and that's a foundation worth defending as research into these materials continues.
Looking ahead, demand for specialized building blocks like 2,3,4,5-tetrafluorobenzyl alcohol isn’t likely to fade. As industries press further into high-performance electronics, targeted therapeutics, and advanced filtration membranes, each application brings new requirements for stability, selectivity, and ease of synthesis. Ongoing work by chemists and engineers often circles back to the need for greener, more efficient preparation methods, and for continuous improvement in handling, purification, and waste management. The possibility that this compound—or relatives modified with even more complex functional groups—might lead to safer, longer-lasting, or more effective materials drives real investment and collaboration. Sharing my own experiences working near the edge of synthetic methods development, it's clear that these challenges motivate teams to reach across disciplines, pooling expertise to develop solutions that don't just tick boxes for performance, but do so responsibly. The story of 2,3,4,5-tetrafluorobenzyl alcohol, like so many specialty chemicals, underscores how human ingenuity, practical problem-solving, and a little caution underpin every leap forward in modern science and industry.
In a lab packed with strange beakers and busy researchers, someone once asked me why we bother using such targeted chemicals like 2,3,4,5-tetrafluorobenzyl alcohol. Some see it as just another tongue-twister on a bottle. I see something practical: a building block that keeps showing up where precision really counts.
Chemistry is like cooking—sometimes you toss in a dash of something special to get the reaction just right. 2,3,4,5-Tetrafluorobenzyl alcohol works as one of these trusty ingredients. Its structure, loaded with four fluorine atoms on the benzene ring, helps scientists tweak molecules in a way that wouldn’t be possible with a plain old benzyl alcohol. You find it showing up in pharmaceutical research and agrochemical labs. Researchers want molecules that behave in very specific ways, and those fluorines nudge chemistry in directions that open up new possibilities.
Pulling examples from published work, researchers use this compound to craft intermediates for more complex drugs or bioactive agents. Fluorine changes a molecule’s metabolism, stability, and how it engages with its environment. Medicinal chemists count on it for making drugs that stick around long enough in the body to do their job, without turning toxic.
Several years ago, I watched a presentation on polymers that could survive in harsh industrial settings. The speaker talked about how basic aromatic alcohols were outclassed by their fluorinated cousins. 2,3,4,5-Tetrafluorobenzyl alcohol became their go-to solution for polymers used in electronics and coatings. The fluorine atoms boost resistance to solvents, acids, and heat—a single change with big consequences for consumer safety and product durability. The same trend shows up in the push for new liquid crystals in electronic displays, where precise control over physical properties means sharper screens and longer-lasting devices.
In environmental labs, accurate detection of trace chemicals can spell the difference between safety and disaster. Technicians sometimes use this alcohol to derivatize samples—that means chemically “tagging” other molecules, so they show up more clearly during gas chromatography testing. Strong electron-withdrawing from the fluorines brightens the signal. Suddenly, substances that would hide at the bottom of the detection chart pop up clearly for analysis. This matters for everything from checking pollutants in water to ensuring food safety.
I’ve seen the flip side too. Fluorinated chemicals sometimes stick around in the environment far longer than anyone intended. The same stability that makes them useful also means they don’t break down easily. Some developed countries now want tighter tracking and more responsible disposal to avoid polluting water or soil.
Solutions can take many forms. Green chemistry movements push for alternatives, recycle fluorinated wastes, or build safer molecules with similar benefits. Research teams focus on using these powerful little blocks only where their performance truly stands out, not just out of habit. Real transparency about where these chemicals go, and meaningful investment in waste treatment, would give science and industry space to innovate without repeating the mistakes of other persistent chemicals.
2,3,4,5-Tetrafluorobenzyl alcohol might look like a mouthful, but its chemical makeup tells a straightforward story. With its four fluorine atoms sitting on a benzyl backbone, plus a single alcohol group, the formula is C7H4F4O. The molecular weight clocks in at about 180.10 g/mol. Chemists who work with aromatic alcohols and halogenated compounds know well that swapping out hydrogen atoms for fluorines changes more than just the number on the scale. Fluorination can give compounds unique reactivity, higher stability, and in many cases, a different safety profile.
In my days working alongside organic chemists, I saw the value that fluorine substitutions bring to the table. The presence of several fluorine atoms in a benzyl alcohol framework nudges the molecule’s reactivity, not in a minor way but in ways that matter in both laboratory and industrial applications. Substitution like this can help make molecules more resistant to metabolic breakdown—a trait that’s important not just in pharmaceuticals but in specialty polymers and materials. Fluorinated compounds often don’t just survive tougher chemical environments; they can enhance the performance of entire systems, from agrochemicals to advanced electronics.
Drawing on the distinct chemical features of 2,3,4,5-tetrafluorobenzyl alcohol, researchers can fine-tune reactions that wouldn’t pan out with non-fluorinated analogs. Fluorine’s small size and high electronegativity shift electron density in the aromatic ring, shaping how the compound behaves in cross-coupling reactions, nucleophilic substitution, or even in more advanced transformations. This creates a niche where chemists can design new molecules, catalysts, and end-use products. In my experience, this ability to steer reaction paths can be a game-changer for creating next-generation chemicals.
Chemicals with multiple fluorine atoms don’t just offer up new science; they also add new questions about safety and sustainability. The environmental persistence of fluorinated aromatics can raise eyebrows, especially in regulatory circles. Many scientists remember how long-chain perfluorinated compounds triggered global concern due to their resistance to breakdown and their tendency to accumulate in the environment. With increasing knowledge and regulatory scrutiny, it has become clear that we need smarter ways to design, use, and dispose of these materials. Chemists have to think beyond immediate performance. Any widespread adoption demands responsible handling, clear disposal protocols, and rethinking waste streams.
While innovation compels us to use 2,3,4,5-Tetrafluorobenzyl alcohol for its chemical advantages, it’s just as crucial to address the environmental baggage that fluorinated molecules sometimes bring. Encouraging green chemistry practices, such as using more benign starting materials and greener reaction conditions, helps minimize risk. Supports for improved waste management and recovery systems in both laboratory and production settings can make a real difference in limiting environmental exposure.
Chemistry shouldn’t just chase the next big breakthrough. The responsibility sits with everyone in the lab and beyond, to balance innovation with caution, and recognize that every new compound comes with both promise and responsibility.
Ask anyone who has worked in a lab or a workshop, and you’ll get the same story: keeping chemicals in the wrong place, or letting things slide just a little, often backfires fast. One time, a bottle of sulfuric acid sat a bit too far from the vent. The fumes ate through the poorly sealed cap, and the next morning, everything smelled like rotten eggs mixed with trouble. Plenty of disasters begin this way—usually more from human error or overconfidence than from wild accidents.
With chemicals, vague advice leads to trouble. Let’s talk specifics. Take strong acids or bases. These always demand containers made of glass or certain plastics, not just any jug lying around. Store them cool and dry, away from direct sunlight. A little heat or UV light, and suddenly, unstable reactions or leaky bottles become the afternoon’s headache. Hydrochloric acid, for example, thrives on corroding everything nearby—workbench, floor, metal shelf. There’s a reason schools and industrial sites set aside a separate, ventilated spot just for this stuff.
Nobody in their right mind stacks oxidizers near flammables. Mix those by mistake, and you’re basically setting the stage for fireworks—of the worst kind. The fire marshal’s reports often read like a checklist of “what not to do” borrowed straight from hasty storage mistakes: bleach close to ammonia, fuel cans sharing a shelf with acids. Experience teaches fast. Keeping categories far apart may mean running up the stairs a few extra times, but the peace of mind outlasts any inconvenience.
Labels save lives, not just reputations. After a few years surrounded by rows of bottles, the habit of double-checking every label sticks. Color codes, warning signs, and even hand-scrawled block letters prevent confusion. Unmarked bottles end up the wildcards in every incident report. It’s easy to think, “I’ll remember which one is which.” That kind of thinking only works right up to the first close call.
Handling brings its own risks, even if storage checks every box. The story of a neighbor trying to pour paint thinner back into the can with bare hands quickly turned into an ER visit for chemical burns. Gloves, goggles, and well-maintained spill kits aren’t for show. Mistakes often bundle together: poor lighting, interrupted concentration, or a missing funnel. Every shortcut adds up, but not in your favor.
Better policies come from real pain and smart adjustments. Storing only what’s needed cuts clutter and confusion. Training matters—a quick refresher every few months goes further than pages of policies tucked away. Ventilated cabinets and lockable doors don’t just meet regulations; they keep the morning routine from turning into an emergency. Good lighting and clear signs change the pace and confidence in handling the most unpredictable solvents or powders.
Most mistakes in chemical storage and handling come from hurrying or assuming nothing could go wrong. Learning hands-on and adjusting habits beat reading another manual. In chemical work, small changes prevent big regrets—and those lessons never get old.
Talk about “2,3,4,5-tetrafluorobenzyl alcohol” and you’ll probably see some eyes glaze over, yet this isn’t some far-off lab curiosity. It comes from the world of fluorinated chemicals, a space that’s left its mark on everything from nonstick coatings to the latest in pharmaceuticals. Whenever you see the benzyl alcohol part of a chemical, you know you’re looking at something designed to fit into many different chemical reactions. Fluorinated versions, like the one here, often bring higher stability—a quality handy for certain products but also a concern for the environment and human health.
Knowing what you’re dealing with isn’t about fear; it’s about respect. I remember my college chemistry lab, working with solvents that smelled like trouble. A few years of education drilled into us: gloves, goggles, and fan always running. No shortcuts. Why? Because even chemicals that don’t shout danger can turn a day in the lab into a day at the clinic. 2,3,4,5-Tetrafluorobenzyl alcohol falls in this “don’t assume it’s safe” bucket. There's no widespread consumer product using this alcohol outright, which probably says something about its handling needs.
Fluorinated organic compounds have earned a reputation for causing headaches in toxicology circles. I’ve seen data on benzyl alcohols in general: moderate toxicity, especially if inhaled or ingested, irritation to eyes, skin, and respiratory tract. Add four fluorine atoms to the ring, and risk often goes up. Fluorinated substances can linger in the environment and are hard to break down, building up in animal tissue over time. We call this bioaccumulation, and it matters because it’s not just about the person doing the mixing or the employee at the plant.
With this specific alcohol, scientific literature gives only sparse information, but the warnings about similar chemicals echo here. The chemical can irritate your skin and eyes, and if it turns to vapor, breathing it for any stretch of time could make you dizzy or sick. Swallowing it? Don’t test that theory.
Regulators often take time to assess specialty chemicals like this. The slow pace doesn’t excuse taking risks. Chemists and workers trust their lives to data sheets, but real safety comes down to culture. When safety goggles become routine—not a formality—we start winning. Companies and labs should enforce these rules, no matter how minor the exposure seems. Everyday folks may never buy a bottle of this alcohol, yet our rivers, air, and food webs pay the price of careless disposal.
Personal connection helps motivate change. I watched cleanup bills soar after a minor leak of a different fluorinated compound—they don’t play fair when released. Extra diligence in storage, clear labeling, and on-site spill kits prevent many problems. Policies on disposal should fit the stubborn, persistent nature of these chemicals, too. Industry must invest in research for less persistent alternatives, support tracking and inventory controls, and avoid “fire and forget” waste dumping. For anyone at home, any chemical experiment—even “harmless” ones—deserves gloves, goggles, and fresh air.
The tools and solutions exist. We just need to remember that no one wants to be the story in next week’s news because of a preventable chemical accident.
Out in the world of fluorinated chemicals, 2,3,4,5-tetrafluorobenzyl alcohol doesn’t always grab the top headline, but this stuff brings a lot to the table—especially when you look at its solubility. The story really starts with its structure. Adding four fluorine atoms to a benzyl alcohol backbone flips the standard playbook: you get a less polar molecule with a stubborn resistance to mixing with some of the liquids its plain cousin blends with easily.
Memories from long afternoons in the lab come back when I smell the sharp, clean scent of these benzylic alcohols. Classic benzyl alcohol dissolves like a charm in water and just about anything organic. Not here. The extra fluorine atoms in 2,3,4,5-tetrafluorobenzyl alcohol block that trait. They create an electron-rich ring that's surprisingly resistant to hydrogen bonding, so water doesn’t welcome it much. Nonpolar solvents like dichloromethane or ethyl acetate often do the job better. This isn’t just a footnote; it shapes how researchers and manufacturers approach synthesis or purification. Skip this step, and there’s a good chance you’re left with oily residues, failed extractions, or wasted time.
Good solubility in organic solvents makes this compound a quiet hero in the right hands. People working on new drugs or designing specialty polymers like working with fluorinated aromatics like these because their properties can boost thermal stability, reduce surface energy, or offer strong resistance to degradation. Picture a chemist in drug discovery: they keep samples dissolved in DMSO or acetonitrile, not water, to keep solutions homogeneous. I've seen first-hand how poor solvent choices often mean more troubleshooting and delays, not just for bench chemists, but across an entire R&D pipeline.
Research shows that fluorination usually drops water solubility, and this holds up here. Think of it almost like waterproofing. The more fluorine, the more hydrophobic the compound becomes. For that reason, these molecules don’t wash down the drain easily. Waste streams need special handling. Environmental concerns grow—fluorinated by-products have staying power in soil and water. A handful of studies point out how persistent these molecules can be, creating pressure for chemists to think through solvent-recycling steps or safer alternatives, long before scale-up.
The question isn’t just about what mixes and what doesn’t; there’s a larger responsibility at work. Real-world chemistry often falls short of the “greener chemistry” promised by modern slogans. I’ve watched teams struggle as they try to find greener solvents or work up protocols that comply with new environmental rules. Tetrafluorobenzyl alcohol, like many of its cousins, nudges labs and manufacturers to innovate beyond the standard solvent list. Some groups have started exploring water-miscible organofluorine chemistry. Others turn to continuous-flow reactors, which cut down on waste and allow for tighter control over how these stubborn molecules interact with solvents.
Solubility isn’t just a single data line for a chemical like this. It shapes workflows, impacts environmental choices, and can add unexpected roadblocks or open doors in a research setting. Getting curious and questioning those daily solvent choices lands you further ahead than sticking with old habits. For people who work with chemicals every day, learning the real behavior of a small molecule—like 2,3,4,5-tetrafluorobenzyl alcohol—has ripple effects beyond the bench into greener practices and smarter workflows.