Chemists have long had an eye for the transformative power of fluorine. Back in the early 20th century, introducing fluorine into aromatic rings stirred up excitement in laboratories. But it wasn’t until advances in synthesis, especially electrophilic and nucleophilic aromatic substitution during the mid-1900s, that compounds like 2,3,4,5-tetrafluorobenzonitrile started appearing beyond patent blueprints. Researchers found that stacking fluorine atoms onto a benzonitrile core delivered not just academic curiosity, but real-world utility — all in service of pharmaceuticals, agrochemicals, and high-performance materials. By the ‘80s and ‘90s, improvements in catalytic fluorination meant large-scale synthesis lost much of its cost and complexity, and the door opened for regular use outside specialist circles.
Most chemicals don’t leave an impression on people outside a small group of experts. 2,3,4,5-Tetrafluorobenzonitrile is a bit different, showing up in custom synthesis catalogs and under fluorescent lab lights. It’s not just a raw material, it’s a building block — a stepping stone for scientists developing the next class of antibiotics, pest controls, or liquid crystals. Researchers are drawn to its versatility. The combination of four fluorine atoms and a nitrile makes it a candidate for reactions that demand both electron withdrawal and functional group selectivity. This isn’t a catch-all chemical; it functions as a foundation when creating value-added molecules in serious research and industrial labs.
2,3,4,5-Tetrafluorobenzonitrile tends to look like a colorless to pale yellow crystalline solid, with a sharp, sometimes pungent odor that hints at the nitrile group and fluorine’s presence. The compound typically boasts a melting point in the mid-50s Celsius range, but what truly sets it apart lies in its chemical stability. High electronegativity from the fluorine atoms means resistance to oxidative and acidic conditions that would chew up less robust molecules. Such stability opens new doors for storage and shipping, reducing risks of decomposition. The low polarity typical for heavily fluorinated benzenes helps it dissolve in organic solvents, but getting it into water remains tough.
Chemists handling this compound rarely depend only on a label. Instead, they look for corroboration: LC-MS analysis, NMR spectra, purity percentages from suppliers. Usually, purity around 98% serves most applications in synthesis. Safety labels highlight the toxicity associated with nitrile groups and the persistent environmental nature of multi-fluorinated aromatics. National and regional hazard regulations guide packaging, so transportation aligns with good laboratory practice. Responsible suppliers provide all information for safe handling, storage, and emergency response.
Building 2,3,4,5-tetrafluorobenzonitrile hasn’t always been simple. Early routes often relied on direct fluorination — a risky maneuver, given fluorine’s reactivity. Over time, safer and more predictable methods gained favor. These days, most production uses stepwise nucleophilic aromatic substitution, starting from pentafluorobenzene or similar precursors with a controlled introduction of the nitrile group, sometimes through a diazonium salt pathway or copper-catalyzed Sandmeyer-type reactions. Innovations continue to reduce hazardous by-products and improve yields, but full green chemistry alignment still feels a stretch due to the stubbornness of those carbon-fluorine bonds.
This molecule may seem chemically quiet thanks to its shield of fluorines, but under the right conditions, the nitrile group springs to life. Coupling reactions with amines and the transformation of the nitrile into carboxylic acids or amides showcase its flexibility. Selective defluorination or hydrolysis offers chemists a challenge, but those who succeed unlock access to closely related compounds — crucial steps in making new pharmaceuticals or agrochemical scaffolds. The electron-deficient ring keeps reactions directed and chemoselective, ideal for those designing precise synthetic routes.
Inside research papers and catalogs, you’ll see a handful of synonyms: 2,3,4,5-tetrafluorobenzenecarbonitrile, TFBN, or CAS 324-84-3. Each shorthand fills a need, sometimes emphasizing the molecular structure, sometimes just providing a quick handle for those in a hurry. Across languages and research cultures, the root is unmistakable — fluorines at every point but the para-position, with a biting nitrile punctuating the chemistry.
Experienced chemists treat multi-fluorinated aromatics differently than standard benzene compounds. Skin contact, inhalation, and ingestion risks drive strict adherence to lab protocols. The nitrile group demands good ventilation due to the risk of forming toxic gases during decomposition. Many labs assign these chemicals to specialized fume hoods, double gloves, and careful eye protection, especially during heating steps. Regulatory agencies like OSHA and ECHA issue guidelines to prevent chronic exposure, emphasizing environmental controls — spent reagents and residuals need careful disposal, given their resistance to standard degradation pathways.
This compound finds its value in more places than most specialty chemicals. Pharmaceutical chemists use it as a masked handle for further transformations, building molecular candidates in the fight against infection and cancer. In agrochemical companies, the compound serves as a warhead in the quest for potent, selective herbicides and insecticides. Material scientists value the unique combination of rigidity, hydrophobicity, and thermal stability, key qualities in designing advanced polymers or specialty coatings for high-value electronics. Despite its relative obscurity in the broader chemical market, its niche presence signals genuine utility for researchers striving to solve tough challenges.
Behind every breakthrough involving 2,3,4,5-tetrafluorobenzonitrile, countless hours of research fill notebooks. Scientists continually push for catalysts that lower reaction temperatures, improve selectivity, and limit waste. Some groups experiment with microwaves or alternative solvents to cut down on reaction time and environmental footprint. University labs chase new ways to activate or defluorinate the ring, hoping to unlock even more downstream chemical diversity. Industry partnerships often fund longer research tracks, betting that fine-tuning this chemistry leads to valuable patents or new product lines.
For all the progress fluorinated aromatics have paid forward, concerns about their toxicity and persistence shadow their success. Toxicologists dig deep into both acute and chronic effects: the risk of skin and lung irritation, long-term accumulation, and changes to genetic material after chronic exposure. Some animal studies flag the possibility of bioaccumulation, especially in aquatic environments. Regulatory bodies step in with restrictions, often requiring extensive safety data before any product containing such rings exits the lab and enters the marketplace. Striking a balance between innovation and safety shapes every debate about fluorinated intermediates today.
Society doubles down on both technological progress and environmental safety as more compounds enter global supply chains. For 2,3,4,5-tetrafluorobenzonitrile, the demand for new medicines, greener agrochemicals, and high-performance materials guarantees continued attention. Researchers must find production methods that lower waste, energy input, and environmental persistence. Pressure mounts for chemical companies to register solid safety data and support programs for responsible disposal. Opportunities to develop selective deconstruction chemistries stand out as one path toward balancing synthetic utility with sustainability. With each new paper, chemists learn more not just about fluorinated chemistry, but about shaping progress in a way that respects both innovation and the world around us.
Chemistry feels overwhelming, especially with names like 2,3,4,5-tetrafluorobenzonitrile. Strip the jargon, and it’s a benzene ring decked out with four fluorine atoms and a nitrile group. Anyone who’s taken a turn with a molecular kit in college knows how a single functional group can flip the script for a compound’s behavior. Here, swapping hydrogen for fluorine makes a serious difference, both in reactivity and in how this stuff might affect the environment.
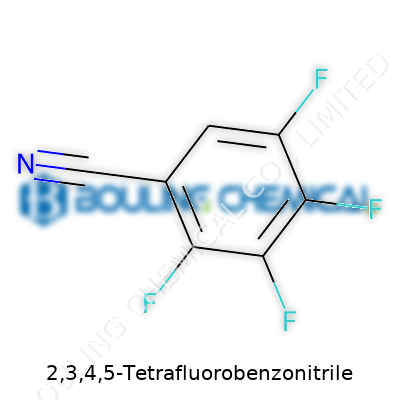
For those mapping it by hand, the structure builds on a benzene ring, which forms the backbone. Picture carbon atoms laid out in a flat, connected hexagon—a staple for many organic molecules. In this compound, four of those carbons—at positions 2, 3, 4, and 5—carry a fluorine each. The number sequence counts around the ring starting from the one attached to the side group. Fluorine’s presence isn’t just about swapping out atoms; it transforms benzene into something with new properties, like resistance to heat, chemical attacks, and environmental breakdown.
The nitrile group (-C≡N) sticks itself onto the ring, usually at position 1, because chemistry likes to give side groups top billing with its numbering scheme. This group acts as a strong electron sink, pulling charge away from the rest of the molecule. Together with the fluorines, the whole combination gets even more intense: high stability, low reactivity, hard to break down. Companies lean into these features for specialty chemicals, but these traits don’t always work out for ecosystems.
Fluorinated organic compounds often resist breakdown—sometimes by design, sometimes unintentionally. These kinds of molecules show up in fields as different as pharmaceuticals and materials science. Chemists use 2,3,4,5-tetrafluorobenzonitrile to build up more complex fluorinated materials, especially in electronic and agrochemical industries. The electron-withdrawing power of both fluorine and the nitrile group creates starting points for more intricate chemistry down the line. People who’ve spent hours on organic synthesis quickly spot just how useful that can be for making new molecules that need a stable, inert base.
The trouble lies in what happens after use. Fluorine atoms don’t let go easily; waste products laced with these structures can stick around for years. The same strength admired in a lab morphs into a lingering headache for communities and regulators. Nobody wants compounds that persist in water, air, or food, especially with research showing how some fluorinated chemicals affect wildlife and health in ways still being unraveled.
People in the field understand the worry. Universities now encourage research into ways to break down or recycle fluorinated compounds. Enzyme-based approaches or new chemical pathways hint at progress. Regulators push for stricter handling and monitoring, and industry buys in because nobody wants a repeat of past environmental mistakes with persistent pollutants. Researchers want to keep using the strengths of fluorine chemistry without stacking up invisible risks for later generations.
Chemistry doesn’t happen in a vacuum. As someone who’s seen both the excitement of a new reaction and the complexity of dealing with chemical waste, I see the need for conversations that connect molecular structure to the choices we make in labs, factories, and policy meetings. Understanding and respecting molecules like 2,3,4,5-tetrafluorobenzonitrile helps spark the next round of solutions—because whether we craft, use, or study them, the impacts last long after the ink dries on a research paper.
New molecules often change the way manufacturers build better products. Take 2,3,4,5-tetrafluorobenzonitrile—a chemical that doesn't get mainstream attention but pulls a lot of weight in the background of everyday life. If you walk through a research facility or specialty chemical plant, you quickly see how often the backbone of an advanced molecule connects back to simple, reliable building blocks like this one.
Most of the pharmaceuticals on the market start with smart chemical strategies. In drug discovery and design, chemists need to introduce tightly controlled variations into molecules to meet strict safety and performance standards. The four fluorine atoms on this compound pack quite a punch, helping new drug molecules resist breakdown, stick around in the body for longer, or fit into the right pocket of a protein target. With each new antiviral or anti-inflammatory agent, small modifications mean big changes for patient outcomes. There’s a reason the pharmaceutical world trusts fluorinated aromatics: better absorption, stronger stability, and improved targeting.
In agriculture, chemical innovation keeps crops safe and yields high. Many pesticides and herbicides begin life as put-together structures featuring tough, stable groups like this benzonitrile. Plants and fungi struggle to break the molecule down, giving protection that lasts long enough to see a real difference in harvest results. Farmers may never know the name, but they depend on crops protected by molecules built off this dependable foundation.
Strong, lightweight, and resistant to the elements—these are qualities prized in high-performance plastics and polymers. Engineers rely on fluorinated aromatics like 2,3,4,5-tetrafluorobenzonitrile to give plastics extra resistance against heat, UV, and chemical exposure. Aerospace panels, electronics casings, and components that work under harsh factory conditions use polymers engineered for reliability. In my own work, I’ve helped test coatings for sensitive equipment; every time, additives like this help protect surfaces where other plastics would fall apart.
Electronics need insulation and protection too. The same properties that shield a microchip from humidity also let electrical contacts hold up in hostile industrial spaces. Building these features into the polymer backbone saves money and trouble down the line, letting devices last longer and suffer fewer failures.
Environmental concerns only grow more important every year. The chemical industry faces pressure to reduce waste, handle safer processes, and switch away from legacy compounds with toxic byproducts. Versatile intermediates like this one simplify synthesis routes—fewer steps, less hazardous waste. This compound often replaces more problematic ingredients, shrinking hazards for workers and the wider public.
Building the next generation of greener pesticides, longer-lasting electronics, and breakthrough medicines means thinking carefully about every molecule in the chain. Compounds like 2,3,4,5-tetrafluorobenzonitrile matter not just for what they do, but for how their use can reshape the bigger picture across multiple industries. As we keep pushing the limits of technology and safety, smart chemistry sets the pace.
Some people hear terms like “2,3,4,5-Tetrafluorobenzonitrile” and start searching for their old chemistry notebooks, feeling that familiar headache creep up. But this isn’t just another tricky compound name—understanding its details, especially its molecular weight, shapes practical decisions in labs and factories around the world. So let's take a practical look at what the molecular weight of this compound means and why it deserves a closer look.
2,3,4,5-Tetrafluorobenzonitrile carries the formula C7H2F4N. Modern organic chemistry relies on basic arithmetic. Find the atomic weights: carbon (12.01), hydrogen (1.008), fluorine (18.998), and nitrogen (14.01). Add up each atom's contribution:
Tally the total: 84.07 + 2.016 + 75.992 + 14.01 = 176.088 g/mol. Knowing this value saves headaches for anyone running a synthesis, calculating yield, or checking if the scale’s reading makes sense. In my own research on aromatic nitriles, tracking molecular weights early keeps your project timeline from slipping later on.
A lot of people shrug off molecular weights as background trivia for tests, but every step in industrial chemistry, from purchasing raw materials to setting safe exposure limits, leans heavily on that one number. Chemists need to weigh out solid samples with surgical precision. Small mistakes in the decimal bring real headaches to quality control. Many years ago, I watched a colleague waste a full week chasing a mystery impurity, only to realize bad calculations led to skewed proportions. Nobody wants to admit a simple math slip caused hours of repeat runs.
In pharmaceutical research, the story grows sharper. The FDA wants confidence that chemists know what dose they’re handing out, not runaway error margins. If drug developers botch the backbone calculation, the error ripples all the way to the clinic. Tetrafluorobenzonitrile’s unique substitution pattern—four fluorines on the ring—alters its electronic characteristics, impacting reactivity in later stages of synthesis. That means you can't guess at the molecular weight and hope it’ll work out.
It sounds simple: just add the numbers, right? But the reality slips. Sometimes, analysts work from incomplete or outdated data sheets. Digitizing reference data in common lab software has helped, but errors linger—copy-paste slip-ups, rounding, and outdated libraries can send a ripple across multiple projects. Taking a few minutes to re-check the calculation prevents wasted time and faulty results. When new team members start in the lab, one of my early lessons involves showing how to build molecular weight from scratch, pen and paper. It’s more than a calculation; it’s a confidence-builder.
Every organization using or manufacturing tetrafluorinated aromatics needs a direct line to trustworthy chemical data sources. Reliable databases, routine training refreshers, and real “pencil-and-paper” checks prevent the kind of mistakes that escalate. Encouraging a habit of verifying key data points like molecular weight—especially at the start of every new project—saves more time and frustration than almost any fancy lab gadget. For chemists, students, and plant managers alike, knowing the number isn’t just about trivia. It’s about doing the job right, keeping standards high, and building trust in every result.
Anyone who’s worked in a lab learns pretty quickly that small mistakes with chemicals can turn stressful in a hurry. Take 2,3,4,5-Tetrafluorobenzonitrile for example. It’s a mouthful to say, but in practice, it’s just another specialized compound that shows up in research, chemical manufacturing, and sometimes pharma projects. Using it right hinges on paying attention to storage and handling, since safety and performance rely on getting those details right.
We’re not talking basic pantry storage here. 2,3,4,5-Tetrafluorobenzonitrile, like many aromatic nitriles, brings its own quirks. Moisture creates problems — even low humidity in the air can react with nitriles over time, leading to decomposition or changes in chemical purity. A dry environment, with a tight-sealing container, stands as the sensible choice. I always opt for an amber glass bottle if there’s any chance of light sensitivity, since a few compounds in this group do break down in sunlight.
Temperature control plays a huge part. Colleagues have argued about fridge versus room temperature, but I’ve seen the best results keeping these materials cool, but above freezing. A climate-controlled cabinet, with restricted access, keeps inventory accurate and deters inquisitive hands from tampering. No one wants a nitrile spill.
Reading SDS sheets is a habit, but relying on your senses matters, too. Many nitriles, especially fluorinated ones, give off sharp odors and can irritate the nose and eyes. I’ll never forget the time a friend let vapors drift while pipetting on an open bench; one cough had the whole room scrambling for fresh air. Safety goggles and gloves block the most obvious risks. A chemical fume hood earns its keep with every container opened.
Spills never go well with these compounds. Fluorinated aromatics can stick around in your workspace or react with cleaning agents. I stick to absorbent pads, then follow up with neutralizing solutions approved for cyanide derivatives. Breaking old habits, like using paper towels, can make all the difference if your priority is both safety and keeping residue out of the trash.
I’ve seen more labs get dinged on waste disposal than just about anything else. 2,3,4,5-Tetrafluorobenzonitrile shouldn't end up in sinks or regular trash. Most local authorities treat fluorinated and nitrile wastes as hazardous due to persistence and toxicity. Keeping a clear, labeled waste stream isn’t just paperwork — it cuts costs by avoiding violations and keeps the workspace running smoothly.
No single product training covers every scenario. I keep a log of who’s used which batch, route new team members through a refresher with every new compound, and double-check containers regularly. Labels fade, storage rooms get reorganized, and it’s easy to overlook a half-empty bottle left on someone’s bench after a late night. Building real safety comes from routine, reminders, and occasionally stopping to rethink our storage setups.
Safe handling isn't glamorous, but it keeps labs productive and people out of trouble. The extra effort upfront in storing and handling complex chemicals like 2,3,4,5-Tetrafluorobenzonitrile pays off by stopping problems before they start.
Ask a chemist about 2,3,4,5-Tetrafluorobenzonitrile, and the first thing you might hear is group sighs about handling fluorinated organics. Academic papers mention this nitrile because it's useful in pharmaceutical and agrochemical labs, where every molecule earns its keep. Not many outside those labs talk about it, yet it keeps popping up on chemical order lists.
Most users, myself included, have faced the classic riddle: if something isn't splashy enough to make headlines, should we worry less? Short answer—no. Ever since the rise of perfluorinated compounds, folks in science and regulation have learned to treat any heavily fluorinated substance with suspicion. Subtle differences in structure can push a compound from "nuisance" to "toxic hazard," so anything with both fluorine and cyanide groups needs extra scrutiny.
The presence of a nitrile group rings alarm bells. Cyanide derivatives are notorious for their toxic potential, mainly from their ability to upset cellular respiration. Though not all aromatic nitriles release cyanide readily, their risk profile sits above that of plain hydrocarbons. In 2,3,4,5-Tetrafluorobenzonitrile, four fluorine atoms packed onto the ring further increase concern. Fluorinated aromatics are often persistent in the environment, and persistence sometimes brings bioaccumulation.
Without direct studies on the human health effects of this specific compound, risk assessments lean on analogues. Many similar fluorinated nitriles show skin and eye irritation and can damage aquatic life. Some laboratory safety sheets recommend lab coats, goggles, and gloves, and urge the use of chemical fume hoods when working with this substance. It's not just academic caution—this is a real risk born from chemistry's darker side.
Stories about PFAS chemicals tell a cautionary tale: resistance to breakdown in nature compounds the risks for groundwater and living things. Regulators once saw these molecules as harmless—decades later, entire communities now wrestle with contaminated water supplies. 2,3,4,5-Tetrafluorobenzonitrile hasn't made it into that infamous club, but its structure signals the need for upfront respect.
Research from the European Chemicals Agency shows that many aromatic fluorinated compounds trigger long-term concerns for both toxicity and environmental fate. That might mean trouble for anyone using this chemical without proper waste collection and destruction protocols. Many industries are moving away from persistent chemicals, not only out of regulation, but from public pressure as well.
Anyone who spends time in a laboratory learns that it's not just about avoiding acute toxicity—chronic exposure, accidental releases, and improper disposal add new layers to the equation. Companies who work with advanced chemicals, like 2,3,4,5-Tetrafluorobenzonitrile, have a duty to rethink how they protect both people and the planet. Ventilated spaces, adequate PPE, and strict disposal rules reduce the chance of incident.
Policy-makers and researchers should work together to close the information gap. That means further studies on toxicity, tracking environmental footprints, and public reporting of safety data. As chemists, we can push for greener alternatives and urge suppliers to phase out chemicals with unclear risk. The broader community benefits when the hazards are seen and managed before real harm arrives.