Looking back at the chemical industry, few compounds tell as complicated a tale as 2,2-dichloroacetophenone. This compound traces its roots to early twentieth-century research searching for powerful tear gases suitable for use in conflicts. During the two world wars, chemists scoured for substances that could disperse crowds, incapacitate individuals, or clear restricted areas, all without long-term lethality. The legacy of this chemical traces back to such ambitions, with records dating to 1922 when it first surfaced as a potent irritant. Governments and scientists saw in it a means of quickly controlling situations that spiraled out of control. Chemists of that era didn’t always grasp how rapidly science can outpace society’s ability to mitigate risks, so many of these compounds—2,2-dichloroacetophenone included—entered arsenals without thorough study of lasting effects.
2,2-Dichloroacetophenone is no stranger in the catalogues of research laboratories and specialized manufacturers. It presents itself as a crystalline, solid compound with a sharp, piercing odor. What sets it apart is its high effectiveness as a lachrymatory agent, meaning it causes tears and eye irritation almost instantly on exposure. Law enforcement agencies coveted such properties through much of the last century and, for a time, 2,2-dichloroacetophenone saw adoption as a principal component in various “tear gas” solutions. As technology evolved, so did scrutiny, and today discussion around this compound focuses less on riot control and more on chemistry and safety.
In the lab, this compound stands out for its moderate melting point and limited solubility in water. Its solid form makes it easy to transport and store, but that form also brings its own hazards, as it can become airborne with little provocation and irritate eyes, skin, and respiratory systems. Its molecular formula—C8H6Cl2O—marks it as a chlorinated ketone. The chlorine groups play a crucial role both in its reactivity and its irritant properties. Unlike some volatile compounds, it sticks around on surfaces and in enclosed areas, complicating cleanup efforts after dispersal.
Precise handling and clear labeling cannot be overstated for compounds like 2,2-dichloroacetophenone. Any bottle or shipping container must clearly show not only the chemical name, but also hazard warnings related to its irritant effects. Users must recognize the danger of acute exposure—eyes that sting, lungs that feel raw, and skin that may blister with extended contact. Storage guidelines recommend fully sealed containers, protected from light and moisture to prevent degradation. Regulatory frameworks—especially in the US and Europe—now demand special labeling and documentation anytime this compound circulates beyond closed labs.
Chemists typically prepare 2,2-dichloroacetophenone by chlorinating acetophenone with chlorine gas in the presence of light or suitable catalysts. This method introduces two chlorine atoms to the alpha-carbon next to the carbonyl group. The technical knowledge required to make this transformation is not trivial; the conditions must be tightly controlled because uncontrolled chlorination could produce unwanted byproducts. This synthesis reminds us how chemistry can harness powerful transformations, but those reactions often leave residues and emissions that create further management challenges.
What makes 2,2-dichloroacetophenone so significant in organic chemistry is its versatility in further reactions. The ketone core allows for nucleophilic addition, while the chlorines offer points for substitution. Under the right conditions, this compound can serve as a building block in the synthesis of pharmaceuticals, agricultural chemicals, or dyes. Unfortunately, its pathway to more benign or specialized derivatives doesn’t erase its hazards. Efficiency and safety both demand skilled chemical intuition. Modification work reminds seasoned chemists that every change invites a cascade of new effects, sometimes desirable, sometimes troublesome.
This substance goes by several names in academic and industrial circles, and these aliases often appear on databases. The most common alternative is CN—a shorthand rooted in its use as a riot control agent. Other synonyms include phenacyl chloride and chloroacetophenone, along with various numbers and letter codes assigned over decades of shipping and regulation. Keeping these straight matters not only for clarity but also to prevent dangerous mix-ups, especially in environments where multiple tear gas agents reside side by side.
Tough standards guide every step with 2,2-dichloroacetophenone, from synthesis to final application. Personal protective equipment becomes a must: chemical-resistant gloves, goggles, and well-designed ventilation systems all form the first line of defense. Many stories circulate about careless handling leading to accidental exposures, with resulting rash, coughing, and even long-term respiratory issues. Laws now require clear procedures for storage and disposal. Companies that manage hazardous materials audits know that complacency can seed disaster quickly. Over the years, more countries have moved to restrict or ban its use entirely, recognizing the balance between security goals and respect for individual health.
Few compounds better illustrate the fraught line between chemical innovation and ethical responsibility. During much of the last century, law enforcement and military outfits counted on 2,2-dichloroacetophenone as a standard “tear gas.” Its effectiveness at clearing crowds is well documented, though its legacy bears scars of misuse and accidental injury. Beyond police and defense, it also carved a niche in chemical synthesis, finding roles as an intermediate for more complex organic molecules. Today, teaching laboratories might stock it more to demonstrate chemical properties than to apply its irritant effects, and even then, only behind layers of safety precautions.
Research momentum shifted as new priorities emerged. Early studies focused on increased potency and lower cost. Now much of the scientific attention spotlights how exposure impacts human health, how chemical properties might be repurposed for less hazardous synthesis, and what alternatives can match its utility without its drawbacks. The evolution of tear gas development mirrors changes in the broader scientific worldview over the last fifty years—shifting from operational effectiveness to minimizing collateral damage and environmental persistence. Teams across the globe continue to tease out new chemical transformations sparked by 2,2-dichloroacetophenone, some hoping to build more specialized or less toxic offspring from its structure.
Toxicology tells a cautionary story. Small doses can trigger severe discomfort, even lasting damage to eyes and lungs in sensitive individuals. I’ve seen credible studies where exposure led to corneal injury, blistering skin, and inflammation deep in the lungs. Chronic cases sometimes left workers with ongoing respiratory issues, and mishandling in the past led to a handful of documented deaths. Disposal turns into its own challenge, with breakdown products that don’t always vanish into harmlessness but sometimes linger in the water or soil. Modern labs increasingly turn to animal-free models or detailed computer simulations to reduce risk. The research points in one direction: overexposure remains a real danger long after the initial sting fades.
Society’s relationship with 2,2-dichloroacetophenone continues to move away from direct application and toward lessons learned. Synthetic chemists are hard at work on alternatives that break down faster, deliver results with lower collateral damage, or shift entirely away from irritant properties. On the regulatory side, laws are getting stricter every year—a direct result of what decades of field and lab use have revealed. Research still extracts insight from its core structure, spinning off new compounds that suit pharmaceuticals and agricultural research. For those who remember its troublesome legacy, the future rests in open-eyed debate, honest assessment of risks, and a renewed focus on safer science. Continued transparency about health impacts, clear labeling, and strict operational protocols will shape how, or even if, this compound stays relevant in the years to come.
No one enjoys learning that the same chemical can find a place in medicine, industry, and law enforcement. 2,2-Dichloroacetophenone is a real example of this. This compound was first introduced in research circles before the middle of the last century and later earned recognition under the name CN tear gas. The reason it's important to know where and how it's used comes down to public safety, ethics, and health.
Anyone who has watched footage of protests or riot control might already be acquainted with what this chemical can do. Police forces around the globe once deployed CN gas to break up demonstrations and maintain public order. Tear gas shells and handheld spray devices used to contain CN filled the gap before CS gas became the military and police favorite. People exposed to CN recall symptoms like burning eyes, coughing fits, and skin irritation—effects designed to disorient and disperse crowds.
This application has always raised questions. Tear gas use isn’t just a footnote; it throws up a real debate about balancing order and the right to protest. Inhaling this chemical hurts. Anyone with asthma or other respiratory conditions may face real health risks. Over the years, medical researchers have documented significant injuries and rare fatal outcomes, especially in poorly ventilated spaces. The chemical sticks around as residue, contaminating clothes, street corners, and even food carts.
Outside control tactics, 2,2-Dichloroacetophenone holds value in laboratories. Chemists use it as a building block for research—preparing more complex organic molecules, pharmaceuticals, or materials. The presence of the two chlorine atoms gives researchers flexibility for developing new compounds. Even if it doesn't attract headlines, this role pushes forward medical and technological innovation.
But labs are tightly regulated. Researchers must handle the substance with protective equipment and must document each application thoroughly. Training ensures that lab workers understand the risks and how to avoid unnecessary exposure.
The continued use of any tear gas should stay under the spotlight. Lawmakers and agencies should listen to research and public testimony about the health impacts. Urban centers need more transparency on when and why police use these devices. Limits must be enforced, especially in enclosed spaces or near vulnerable people.
If society trusts science and regulation, then labs must keep monitoring exposure limits and disposal processes. No one gains when hazardous chemicals leach into water supplies or linger in public parks. Stronger training, periodic review of protocols, and open reporting make misuse harder.
Tear gas once marked authority, but experience shows that trust grows from engagement, not fear. Each choice about chemicals like 2,2-Dichloroacetophenone reflects bigger questions—safety over force, and curiosity over carelessness. That conversation never grows old.
I once witnessed a chemical mishap in a university lab that turned a promising morning into chaos. The culprit? A hurried moment near an irritating compound. 2,2-Dichloroacetophenone reminds me a bit of that experience; it’s not well-known outside specialized circles, but it brings danger to every workspace where it’s handled. Long used in riot control and some lab applications, this chemical earns its reputation from strong irritant properties—particularly affecting eyes, skin, and the respiratory tract.
Goggles don’t just sit on a checklist; with 2,2-Dichloroacetophenone, they become your first true shield. A face shield adds another barrier. Nitrile gloves, not thin latex, prove reliable against these organic compounds. I’ve seen colleagues skip lab coats on a hot day, but splashes from chemicals like this teach fast lessons; long-sleeved, chemical-resistant clothing serves more than lab fashion.
Anyone who’s spent an afternoon in a fume hood knows what a difference air movement makes. 2,2-Dichloroacetophenone vapors act swiftly, and headache or throat burn can follow even brief exposure. Modern labs rely on strong local exhaust and well-maintained hoods, but even in older facilities, cracking a window or working near makeshift exhaust setups beats toughing it out and hoping for the best.
Leaving a bottle unscrewed or failing to secure lids is a shortcut to problems. I keep contrasting tales in my mind: one colleague who methodically double-checks containers before re-shelving chemicals, another who shrugged off minor spills—until the day a cleanup triggered an asthma attack three benches down. Dry, locked, and well-marked storage matters. Absorbent materials for cleanup must handle both liquids and contaminated glass, and waste needs proper channels, not quick trips to regular garbage.
Some labs run hands-on drills, showing new staff exactly how to respond if a bottle tips over or fumes hit unexpectedly. Experienced chemists sometimes act casual, but even pros can get caught out. Nobody should face 2,2-Dichloroacetophenone alone; setting up work in pairs ensures someone else can reach for the eyewash station or call for help if needed.
According to published guidelines, 2,2-Dichloroacetophenone can cause respiratory distress and severe irritation after only short exposures. Eye contact presents a real risk for persistent injury. The National Institute for Occupational Safety and Health alerts us that immediate access to emergency showers and eye washes will help prevent lasting harm. No amount of experience with other chemicals prepares you for the punch this compound delivers.
Leadership in the lab isn’t only about technical skills; it’s about setting examples the rest of the team will follow. Respecting known hazards, keeping protective gear close, and establishing honest communication about close calls protects everyone. Good housekeeping habits push beyond compliance checklists—they build comfort, trust, and readiness if something goes sideways. That’s how you turn a risky environment into a resilient one.
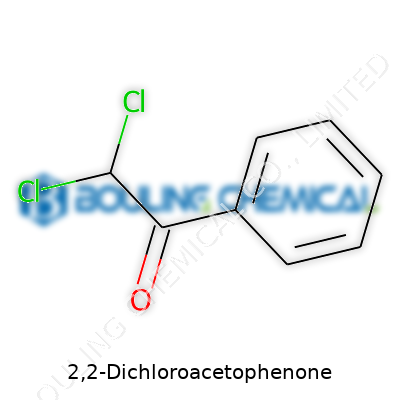
2,2-Dichloroacetophenone gives us a mix of chemistry rooted in both science and real-world impact. The molecular structure draws attention: a core benzene ring, a carbonyl group, and two chlorine atoms attached to the alpha carbon. Its chemical formula goes as C8H6Cl2O. Take a closer look at a diagram, and you’ll spot the layout quickly. On one end, a benzene ring radiates chemical stability common to aromatic compounds. One carbon next to the ring bears two chlorines, a detail that brings a sharp difference when compared to similar molecules.
The particular arrangement of atoms changes how the molecule interacts with biological tissues and the environment. In personal experience, working in labs that handled chemical irritants, I learned fast that even a simple shift in the position of a chlorine atom can make a world of difference in toxicity and persistence. 2,2-Dichloroacetophenone rests in the spotlight partly because of its use in crowd control as a tear gas component. The alpha, alpha-dichloro group on that acetophenone backbone boosts its ability to irritate mucous membranes. This isn’t a small detail; it’s the essence of why the chemical behaves the way it does upon exposure.
Fact stands that the structure explains more than just a chemistry quiz question. It puts focus on the responsibilities tied to such compounds. Chemists must think about the aftermath of every formula—who gets exposed, how persistent the chemical proves in air or water, and whether cleanup leaves lasting traces. I recall a spill response drill at my institute, underscoring the strict care needed for substances just like this one, due to properties like low volatility and resistance to rapid breakdown.
Scientific literature shows the molecule as a skin and respiratory tract irritant. Rapid hydrolysis does not occur, which lets it linger in certain settings unless addressed with strong oversight. Researchers have documented cases where exposure resulted in severe symptoms, confirming that the two chlorines bonded on the alpha carbon make a difference in how our body recognizes and reacts to the substance. The chemical isn’t as easily washed away as some hope.
Problems linked to 2,2-Dichloroacetophenone’s usage invite a closer look at risk management. Safer chemical alternatives exist for certain applications, some using less toxic or faster-degrading molecules. Real progress grows from research into these new structures. Regulatory bodies continue to refine exposure guidelines based on emerging data. The onus falls on every stakeholder—from manufacturers to first responders—to approach compounds like this with caution and transparency.
Knowledge about chemical structure doesn’t live only in textbooks. Whether developing new policies or handling day-to-day lab work, understanding how each atom sits in a molecule and what impact that brings can change the outcome for entire communities.
I’ve spent enough time around chemistry labs to know that storage decisions can make or break a workplace. Even the most mundane bottle of solvent can become someone’s headache after a careless mistake. I’ve seen cardboard boxes collapse under leaking acids, and watched the aftermath of vapors warping plastic shelving. Experience tells me: chemicals demand respect right from the shelf up.
Now, 2,2-Dichloroacetophenone doesn’t make the headlines like fluorine or mercury, but this compound earns caution. Labeled as a lachrymator, it can cause tears and irritation fast. Its track record as a tear gas ingredient in law enforcement might sound old-school, but the health risks stick around if you ignore the basics. Anyone storing this chemical outside a controlled setting is accepting more risk than necessary.
No one should bet on luck with hazardous chemicals—not even with small amounts. With 2,2-Dichloroacetophenone, chemistry guides the storage call: keep it in a tightly sealed container, preferably glass or high-grade plastic that resists corrosion. The compound can react with metals, and leaks spell both workplace hazards and regulatory headaches. The point isn’t paranoia; the point is cutting every chance for a spill or chemical reaction that turns routine into emergency.
Cool, dry, and out of sunlight sets the baseline. Heat and light are notorious for stirring up unwanted reactions, and moisture can break down sensitive chemicals in odd ways. If you’ve seen what humidity does to a forgotten bottle, you know the label sometimes holds less information than the smell. A chemical storeroom should never run warmer than necessary, and the less sunshine creeping between boxes, the better. These conditions help keep 2,2-Dichloroacetophenone stable and limit the chance of accidental vapor release.
Bottle labels matter—something as simple as a missing expiration date or smudged hazard symbol can invite mistakes. People change jobs, paperwork vanishes, and yesterday’s organization slips away faster than most realize. If a co-worker can’t spot what’s inside at a glance, some day that could cost real money or worse. In my own experience, thorough labeling with clear warning symbols is the difference between organized shelves and the mess that sets off an evacuation drill.
Ventilation gets overlooked plenty—storage rooms doubled as broom closets or printer nooks shouldn’t house toxic compounds. Airflow pulls away vapors and sets up another layer of protection between chemical and lungs. Even a faint smell drifting past storage tells you something’s gone sideways. Regular checks, not just when audit season rolls around, help spot early signs of damage: corroded caps, warped lids, or accidental splashes dried like ghostly fingerprints.
Regulatory bodies don’t hand out storage advice as suggestions. Federal and local guidelines grow out of lab disasters and workplace injuries stretching back a century. I once worked in a facility that shrugged off secondary containment—until one accident led to a shutdown and a bill for environmental cleanup. Even a small leak can trigger legal and health consequences far bigger than the hassle of a lockable chemical cabinet.
Secondary containment trays, fire-resistant cabinets, and never mixing incompatible groups in shared cupboards sound like overkill until you see what happens after someone stores solvents next to acids. The right habits, built into daily routines, save both time and lives. At the end of the day, solid storage for compounds like 2,2-Dichloroacetophenone isn’t about extra rules—it’s about respecting the science and experience built up through trial, error, and sometimes, painful lessons.
Handling chemicals like 2,2-Dichloroacetophenone feels routine in labs that see lots of organic syntheses. Peeling open a container of this stuff, you know you’re dealing with a solid that doesn’t pretend to be friendly. The compound shows up as white to off-white crystals. Sitting in the palm, the substance has a faint musty odor. It dissolves well in organic solvents like ethanol and chloroform—not so much in water, though, so spills don’t just rinse away.
Chemically, 2,2-Dichloroacetophenone has a straightforward formula: C8H6Cl2O. That puts two chlorine atoms onto the acetyl group, attached to a benzene ring. Why do those details matter? Chlorination changes everything. The molecule packs a bigger chemical punch, and that’s what gives it real bite—literally, if you catch a whiff or it ends up in the wrong place.
Most folks know 2,2-Dichloroacetophenone as "CN gas," the original military tear gas. Countless riot control stories have its sting—the classic stinging eyes, burning skin, nasty coughing. That’s because CN reacts quickly with the eyes and mucous membranes. It’s much less volatile than modern irritants like CS gas, which means it lingers on surfaces and in the air if not cleaned thoroughly. I've scrubbed lab benches that still reeked of the compound hours after a spill. It’s less forgiving than its newer cousins.
Heating brings risk. This compound melts around 54°C. Above that, it boils near 245°C, but the real headache comes before those numbers: heated CN gas releases toxic fumes including hydrogen chloride and phosgene. It stresses how easily some chemicals cross from lab curiosity to environmental hazard. Without a good fume hood or proper extraction, working with it feels uncomfortable at best, genuinely risky at worst.
Earlier chemical defense and policing doctrines relied on substances like CN before shifting to what we now consider safer or less persistent agents. Even so, certain countries stock it as a backup due to its cost and stability in storage. No one calls it gentle—it’s corrosive to skin, triggers sneezing fits, and can poison you if inhaled at high enough doses or without protective gear.
Chemical properties aside, the compound sticks as a reminder about responsibility. (That’s not just a line.) Accidents happen, sometimes from underestimating storage needs. Keep CN somewhere too warm, and you can lose a whole storeroom to caustic vapors. A sealed, ventilated cabinet well away from heat stops most issues before they start. Responsible disposal also matters—neutralizing solutions, not pouring down drains. Environmental persistence isn’t just a regulatory buzzword; I’ve seen groundwater tests light up years after an improper dump.
Good ventilation, clear labeling, organic-resistant gloves—these aren’t just for OSHA inspectors. Training fresh researchers or security staff about the hazards and limits around CN is the best form of harm reduction we have. The chemical’s presence in old munitions and historical stockpiles reminds us not all risk vanishes with new standards. Routine checks and strict inventory controls keep even old stocks from causing problems.
There’s no escaping: 2,2-Dichloroacetophenone is no relic. It’s a wake-up call. Every property—from its crystalline form to its stubborn smell—underscores the importance of chemical literacy, vigilance, and practical respect in labs, storerooms, or anywhere the substance sits waiting to be used.