A lot of organic chemical tools have come and gone, but 1,4-Dimethylpiperazine has quietly kept itself relevant across decades of laboratory and industrial progress. Chemists first synthesized piperazine derivatives over a century ago, seeing their potential in pharmaceuticals and specialty chemicals. As researchers dug deeper into piperazine’s chemical structure, methylated forms like 1,4-Dimethylpiperazine cropped up. Early records in the chemical literature show this compound popping up as both a synthetic intermediate and an alternative building block for tablets and dyes. Its rise wasn’t flashy, but research communities recognized its flexible and reliable chemical backbone, which let it move steadily from tiny lab flasks into larger-scale production by mid-20th century.
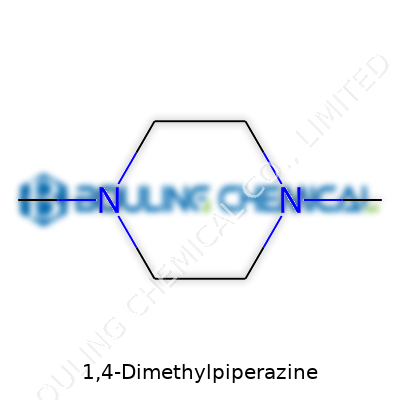
These days, 1,4-Dimethylpiperazine stands as a specialty amine that pops up in many chemical supply catalogs and research inventories. Its basic structure—a six-membered piperazine ring with methyl groups at nitrogen atoms 1 and 4—makes it more than just another organic building block. This simple change from the parent piperazine, by attaching two methyl groups, increases lipophilicity and changes how the molecule reacts with acids and electrophiles. Right now, it shows up both as a research reagent and as a stepping stone to other more complex chemicals. As a chemist, reaching for this compound means trusting its clean, predictable performance on the bench.
Looking closely, 1,4-Dimethylpiperazine takes the form of a colorless liquid or may show up as a low-melting solid, depending on storage conditions and purity. With a distinct amine smell, it easily dissolves in water and many organic solvents. Structurally, the methyl groups attached to the nitrogen atoms reduce hydrogen bonding compared to plain piperazine, meaning differences in melting and boiling points show up. Handling the chemical, it’s hard to miss the volatility and how it reacts to open air—think of the standard caution needed for small volatile amines. As a secondary diamine, the molecule’s backbone offers two reactive centers for further modification, giving it extra utility compared to single-functional molecules.
On a chemical label, one finds the familiar identifiers: CAS registry number, chemical synonyms, molecular formula. Purity often runs high, above 98%, since impurities can mess with reaction outcomes in sensitive syntheses. Storage instructions—keep the container tightly closed, avoid sources of ignition, and maintain a cool environment—reflect its volatility and reactivity. Standard labeling flags the potential health hazards, alerting users to risks if proper care slips. Years spent scanning labels train a chemist’s eyes to catch these details before a reaction begins, and 1,4-Dimethylpiperazine’s labeling follows protocols that protect people and projects.
Manufacturers usually produce 1,4-Dimethylpiperazine by alkylating piperazine with methylating agents like methyl iodide or dimethyl sulfate. This approach relies on controlling stoichiometry, temperature, and solvent choice to favor replacement at both available nitrogen atoms without over-alkylating or generating unwanted by-products. Careful monitoring helps trap off-gas and ensures clean separation of product from methylating reagents, which is especially important because these reagents can be toxic and sensitive to moisture. While alternative routes pop up in the literature, the methylation strategy continues to dominate, partly because secondary amines like this one selectively undergo dialkylation under the right conditions.
This compound’s two methyl-protected nitrogens remain ready targets for functionalization through acylation, alkylation, or even oxidation under strong enough conditions. Taken as a nucleophile, 1,4-Dimethylpiperazine reacts briskly with benzylic halides, sulfonyl chlorides, or activated acids to form a broad range of derivatives, many of which become active pharmaceutical ingredients or serve as ligands in metal complexes. Its relatively low basicity compared to unmodified piperazine shifts some reaction profiles, which matters when designing synthesis routes for new molecules. Over the years, I’ve seen it serve as both a protective group scaffold and a linking unit in heterocyclic synthesis, and each time its reactivity matches expectations—a boon for any lab chemist faced with unknowns.
Catalogs often list this compound under names like N,N’-Dimethylpiperazine, 1,4-Bis(methyl)piperazine, or simply DMPZ. The variety of names reflects its flexibility across different research contexts. Drug development teams and materials scientists each use their preferred terminology, but they all converge on its identifying codes and chemical structure.
Using 1,4-Dimethylpiperazine safely, one quickly learns to avoid inhaling its fumes and to wear gloves to block skin contact. Like other diamines, it can cause irritation, and there’s some potential for more serious health effects with high or repeated exposure. Standard safety guidelines push for good ventilation, chemical splash goggles, and prompt cleanup if any spills occur. In my lab days, the most important advice centered on treating even familiar amines with care: amines sneak through poor filtration, drift out of open bottles, and stick to gloves without warning. Good training and a cautious approach go a long way, and most incidents in academic or industrial labs come from ignoring established safety steps.
1,4-Dimethylpiperazine pops up across many applied fields. In pharmaceuticals, chemists use it as an intermediate when building active molecules, or tweak its structure to test new drug-like scaffolds. Polymer chemists reach for it when designing flexible chains or adding functional sites, especially for specialty adhesives or coatings. In agrochemical research, the piperazine motif helps generate new herbicides and insecticides, looking for better activity and easier processing. Analytical chemistry sometimes borrows the compound as a reference standard or as a modifying agent for chromatography reagents. In practical settings, its reliability under various reaction conditions explains why so many labs keep a bottle on their shelves.
Development pressures in medical, materials, and synthetic chemistry push researchers to keep exploring derivatives of 1,4-Dimethylpiperazine. Studies have tested its ability to add metabolic stability or modulate biological targets. Some developers work on using it as a core for smart polymers, exploring temperature-responsive gels and molecular sensors. Academics and industry players alike search for new reaction routes that cut waste, operate more safely, or support “green” chemistry principles. My own experience illustrates the value of bringing this molecule into project meetings—its simple structure often leads to surprising routes for drug candidates or materials with unusual flexibility.
No chemical comes without risk, and research into the toxicity of 1,4-Dimethylpiperazine has focused on acute and chronic exposure effects in animals and cell lines. Reports generally suggest moderate toxicity, with main concerns centered on respiratory and skin irritation as well as possible effects on central nervous function with heavy exposure. Regulatory attention means detailed data on safe limits for air and environmental disposal standards. Workers in production plants and academic labs need to keep up with evolving toxicological studies to guide safer handling and limit accidental exposure.
Looking ahead, expectations for 1,4-Dimethylpiperazine rest on its established track record and emerging potential in new fields. Growth in pharmaceutical R&D and specialty polymers shows no sign of stopping, and regulatory pressure to push for greener, safer syntheses highlights the need for versatile, well-understood chemical building blocks. As new technologies demand precise, reliable intermediates, this compound’s straightforward chemistry and predictable behavior likely boost its appeal. For me and colleagues, chemicals like 1,4-Dimethylpiperazine become trusted tools in the push for innovation. Its continued research, responsible use, and careful documentation make it one of those under-the-radar contributors that quietly power science forward.
1,4-Dimethylpiperazine sounds like something straight out of a college chemistry textbook, but it plays its own part in a world many people hardly see. I’ve watched many seemingly niche chemicals punch above their weight in daily industry, and this compound is a good example. It belongs to a family of molecules called piperazines, which are nitrogen-packed rings that chemists love for their versatility in reactions.
Years ago, while working alongside some pharmaceutical researchers, I found out just how much these basic molecules show up as building blocks for bigger, more complex drugs. 1,4-Dimethylpiperazine helps chemists tweak a drug molecule until it works just right in the body. You won’t find this chemical in your medicine cabinet, but you might see what it helps create. Many antihistamines, antidepressants, and anti-parasitic drugs rely on some branch of the piperazine family.
Beyond medicine, this compound helps with the manufacture of specialty chemicals that end up in things like plastics, dyes, and even cleaning agents. It gives materials different chemical properties, sometimes making them stronger or more durable. Companies that focus on specialties, rather than bulk chemicals, often turn to molecules like this for the edge they need to solve specific industrial challenges.
It’s easy to overlook chemicals that only experts talk about, but I’ve seen how something like 1,4-Dimethylpiperazine can change the path of product development. In pharmaceuticals, for example, its ability to form solid, stable crystals with other molecules sometimes shapes whether a drug passes the final stages of development. Cost and safety shape the decision to use it, along with how easily chemists can change its structure to get new effects.
While advancing materials and healthcare, chemicals like this also raise concerns when it comes to safe handling. During my time in the lab, even a tiny error around organic amines could cause problems — strong odors, skin irritation, or more serious risks if safety isn’t respected. Clear labeling and proper training separate successful, safe use from workplace accidents. Every chemist or technician working with such compounds learns to trust, but verify, each shipment and procedure.
Society benefits when chemical innovation balances productivity with care for health and the environment. From personal experience, the best environments involve transparency, constant training, and healthy skepticism of shortcuts. Regulators check for potential harm, urging caution with storage, transport, and disposal. Fact-based oversight creates a safer environment for both workers and neighborhoods near chemical manufacturers.
For future uses, the spotlight will remain on how researchers and companies handle environmental impact. Waste management and pollution control take center stage as these specialty substances leave the lab and enter industrial production. Investing in greener processes makes a difference. Some teams explore less hazardous alternatives, or ways to recycle and recover piperazine compounds.
Real progress happens when scientific advances run alongside hard questions about safety, waste, and long-term effects. As 1,4-Dimethylpiperazine helps push the boundaries of materials and medicine, keeping the conversation active ensures benefits reach people without sacrificing responsibility. The story of this small chemical sits at the crossroads of possibility and caution, always led by the choices people make every day at the bench and beyond.
Most people rarely give a second thought to compounds like 1,4-Dimethylpiperazine, and that’s understandable. On the surface, it has the ring of a niche lab reagent. For chemists, though, these little details—like the chemical formula—carry weight. The formula, C6H14N2, might look basic, but it points to a deeper story about structure, function, and safety.
Let’s break that formula down. Six carbons, fourteen hydrogens, and two nitrogens. This is not some exotic chain of atoms. What matters is the way those parts fit together: a six-membered piperazine ring holding two methyl groups at the 1 and 4 positions. Years working in research showed me that you can’t cut corners with structural identification. Misidentifying or mislabeling a compound can throw off days of experiments, skew safety data, and even cause dangerous reactions in the lab. Piperazine rings are nothing new in medicinal and industrial chemistry, but each substitution pattern changes the properties, whether in basic reactivity or how the compound behaves biologically.
Most fields that use 1,4-Dimethylpiperazine rely on institutional trust. If that formula gets listed wrong in a catalogue, the miss might not get caught until someone’s synthesis goes off track or a toxicity test returns unexpected results. I’ve seen seasoned scientists double-check shipments, relying on formula and CAS number as basic anchors. It doesn’t just save resources. It keeps working environments safer for everyone. A simple typo can mean the difference between a harmless experiment and one that creates hazardous fumes.
From a regulatory perspective, accurate chemical data guards people and environments. Many plant operators and EH&S professionals use chemical formulas to enter substances into local or national inventories. If the formula’s off, substances can wind up regulated under the wrong framework or slip through compliance checks. That creates a headache for people far from the bench—people who need clarity to do their jobs.
Look at established chemical databases such as PubChem and ChemSpider. Both list 1,4-Dimethylpiperazine with a formula of C6H14N2. Organizations like NIST back this up in their chemical reference tables. Academics publishing research in peer-reviewed journals take care to repeat this information. Incorrect formulas not only disrupt synthesis, but also confuse analytical chemists whose work depends on precise identification through mass spectrometry or NMR. Small labs don’t always have the luxury of running tests more than once before project budgets run dry.
Correct chemical formulas make a real difference. Industry bodies and academic publishers can help by enforcing better data verification before catalogs and journal articles hit the public. Digital databases should use automated cross-checks with trusted repositories before adding or updating compounds. In teaching labs, instructors can show newcomers how to verify the structure and formula, using published diagrams and reputable sources. This simple habit stands out among the most practical steps for cleaner, safer lab work.
On the ground, workers and students sometimes feel pressure to move fast. The habit of double-checking chemical data, including the straightforward formula for something like 1,4-Dimethylpiperazine, can prevent bigger mistakes. Over the years, I’ve seen this practice set professionals apart—those willing to take an extra moment to check details help make chemistry safer and more reliable.
Every now and then, a chemical grabs headlines and people start asking the same questions: is it hazardous and should I be worried? 1,4-Dimethylpiperazine, a chemical name most folks don’t hear every day, has been cropping up more and more. It shows up in some specialty manufacturing processes, lab work, and research settings. Unless you happen to work in chemistry, you might have never heard of it. So, what’s the real story behind this compound?
There’s no shortcut in assessing chemical safety—facts matter. Studies and safety sheets tell us that 1,4-dimethylpiperazine irritates skin and eyes. Breathing its vapors can bother the respiratory tract. Anyone who’s spent time working around solvents and laboratory chemicals can tell you: most things that smell strong or sting your skin demand caution. Sensible habits come into play fast. People shouldn’t touch or breathe this stuff directly, and using it without gloves or proper ventilation isn’t wise. It boils down to respecting chemicals for what they are, not dismissing warnings because most folks don’t interact with them every day.
Short-term irritation is often only part of the picture. Chronic exposure to hazardous substances sometimes brings bigger risks, like organ problems or cancer. Reliable public information about the long-term health effects of 1,4-dimethylpiperazine seems limited. That’s not a green light for reckless handling, but it does point to gaps in public knowledge. Whenever research around a chemical is thin, prudent protection becomes even more important. There’s a lesson from countless years in industry and labs: you can’t always predict what a material will do after months or years of exposure. Precaution beats regret every time.
Some folks trust agencies to keep watch and sound the alarm if things get dangerous. Regulatory bodies such as OSHA and the EPA maintain lists of dangerous chemicals and exposure limits for workplace safety. 1,4-dimethylpiperazine doesn’t stand out with strict, unique regulations compared to infamous substances like benzene or formaldehyde. But lack of notoriety shouldn’t lull anyone into dropping their guard. In my experience, sometimes, the lesser-known chemicals become a problem because people don’t fear what they don’t understand.
The real way forward centers on everyday vigilance. Companies handling chemicals need solid educational training, up-to-date safety data, and equipment designed to protect workers. It means equipping anyone around 1,4-dimethylpiperazine with gloves, goggles, and ventilation. Regular refresher training and access to accurate information empower people to protect themselves and their coworkers. I’ve seen small complacencies turn into big problems, so building a workplace culture where safety questions are encouraged goes a long way.
Banning chemicals outright rarely makes sense unless the risks are undeniable and substitutes exist. The better approach is exactly what would keep most hazardous substances in check: full transparency about the risks, easy-to-read safety guidelines, and continual dialogue between workers, managers, and safety professionals. This keeps the focus on people, not just technical labels or scary headlines.
Bottom line, 1,4-dimethylpiperazine deserves respect like so many chemicals used behind the scenes. Whether you’re mixing it in a laboratory or reading about it in the news, knowing the facts and practicing common-sense caution are what keep stories about chemical dangers from turning into real-world disasters.
Most people never hear about 1,4-Dimethylpiperazine outside of a specialized lab or manufacturing plant. For those who work with chemicals, though, the conversation about this substance boils down to one important question: how can we store it safely and responsibly? The details usually don’t make headlines, but the impact of poor chemical storage can reach farther than folks realize. I’ve watched more than one workplace scramble after an unexpected chemical mishap. Good storage practices aren’t just about “following protocols”—they’re about keeping people safe and protecting investments.
1,4-Dimethylpiperazine often serves as a building block in chemical synthesis. Like many organic compounds, it’s not flammable on paper, but exposure to heat, light, and oxygen can trigger unwanted reactions. One colleague told me about a time when a lightly sealed vessel in a sunlit window slowly leaked subtle fumes for weeks before anyone noticed the problem: headaches, strange odors, and unnecessary risk. Storing chemicals like this out of sunlight isn’t just industry lingo; it matters in real-world labs, where windows and busy workrooms can introduce unpredictable variables.
Some folks imagine chemical storage is simply about keeping bottles upright and not mixing acids with bases. With something like 1,4-Dimethylpiperazine, you want tight-sealing glass or high-grade plastic that won’t be affected by organic vapors. The material in contact with the chemical can make all the difference as plasticizers sometimes break down, causing leaks or even unexpected contamination. The best containers feel sturdy in the hand and don’t have crusty or warping caps. At room temperature, the substance is stable, but subtle shifts—maybe a supply closet that heats up every afternoon—can make storage unreliable. To avoid those dangerous temperature swings, a dedicated flammables cabinet or controlled environment closet does the trick.
I remember stepping into a storeroom and trying to make sense of a shelf stacked with ambiguous handwritten notes: “clear liquid,” “amino,” “for project X.” This chaos invites accidents. Detailed labels—chemical name, concentration, date received, main hazards—save time and lives. They protect not just you, but also the new hire who hasn’t memorized the quirks of every bottle. If that label says to avoid skin contact or store away from food and drink, it’s not bureaucracy talking. It’s something learned the hard way in places that traded words for wisdom after a spill or a near-miss.
Some chemicals announce their presence with a sharp whiff; others are subtler. 1,4-Dimethylpiperazine can slowly build up low concentrations of vapor, which is hard to detect in a closed, musty closet. Good ventilation—either a vented storage cabinet or a regularly aired-out room—reduces risk without much effort. I’ve seen labs invest in expensive safety gear but overlook simple air movement. Protecting lungs sometimes means just leaving a door cracked or running a fan, and it costs far less than the clean-up after a spill or exposure incident.
Storing chemicals like 1,4-Dimethylpiperazine demands a culture where everyone on the team pays attention. Experienced workers can spot a weakening cap or subtle change in color before a problem grows. Training isn’t a one-time checklist; it continues every day, embedded in conversation, questions, and peer reminders. Regulators and safety handbooks give us a starting point, but habits built from experience close the gap.
Taking chemical storage seriously keeps people healthy, supports smooth lab operations, and saves money in the long run. The stories we share—about close calls, odd odors, and the label that made all the difference—remind us that real safety lives in practical action, not paperwork.
Once you’ve spent some time in a lab, you get used to a few recurring smells. 1,4-Dimethylpiperazine brings a faint, fishy odor—barely noticeable unless a bottle slips open. Folks measure its pure form as a colorless liquid or, if temperatures dip, a solid with a very low melting point. Not the sort of stuff you’d confuse with water, but it definitely doesn’t wear warning colors. The key is in the details: this chemical doesn’t flash or fume, but it feels slippery to the touch, much like many amine-based compounds.
Most students learn quickly: handling any piperazine derivative means checking the boiling and melting points. 1,4-Dimethylpiperazine comes with a boiling point near 138°C, so in an open flask, it wants to stick around unless you really crank up the heat. Unlike other solvents that disappear in seconds, this one needs patience, whether you're distilling or simply cleaning glassware. It’s a lesson in patience, each drop telling a story of stability.
The solubility question always comes up. This chemical loves water; the ring structure, with nitrogen atoms, invites easy mixing without clouds or clumps. Folks mix it with alcohol or ether when water just won’t do, especially in synthetic chemistry. This solubility isn’t just a matter of convenience—it’s a safety detail. Getting this substance off your hands usually needs thorough rinsing because water alone breaks the bond, making quick clean-up a real possibility.
Having handled enough of these reagents, you start to notice a few tricks. Pouring 1,4-Dimethylpiperazine, you’ll see it moves just a bit less sluggishly than corn syrup, with a density just shy of water. Don’t expect a heavy pour. If you ever spill it, it doesn’t splatter in quite the same way as something like toluene or even acetone. This subtle difference matters in process safety—especially if you’re moving larger volumes in a research or industrial setup.
It’s tempting to lump all amines together, but knowing what sets this one apart keeps you prepared if things go sideways. Temperature swings hit harder with low boiling chemicals. Too much heat, and you risk losing more of your batch than expected. On the cooling side, sudden crystallization can knock out expensive filters and pumps. Over the years, I’ve watched people burn through budgets with spills, pointless vaporization, and mistaken identity just because a property or two got overlooked.
These aren’t textbook warnings—they’re reminders that working knowledge matters more than facts on a page. Any synthetic chemist or formulator who respects the quirks of 1,4-Dimethylpiperazine stands a better chance of keeping the lab safe and products consistent. Investing in proper storage, keeping reaction temperatures and conditions in check, and never underestimating solubility saves more than time; it preserves health and materials. Simple steps: know your chemical, never assume, and let experience guide safe handling every day.