The exploration of aromatic fluorocarbons takes you through some meaningful chapters in chemistry. 1,2,4,5-Tetrafluorobenzene didn’t just appear out of nowhere—its roots stretch back to research on organofluorine compounds, a field that really picked up speed after the second world war. Once fluorine’s unique properties got noticed, chemists raced to create more stable, reactive, and diverse structures. 1,2,4,5-Tetrafluorobenzene joined that roster thanks to methods that cracked the code of selective fluorination, particularly as efforts to replace hydrogen atoms with fluorine without losing a ring’s fundamental stability bore fruit. Laboratories in the 1970s started synthesizing multi-fluorinated benzenes in larger quantities because researchers needed new building blocks for pharmaceuticals, agrochemicals, and advanced polymers.
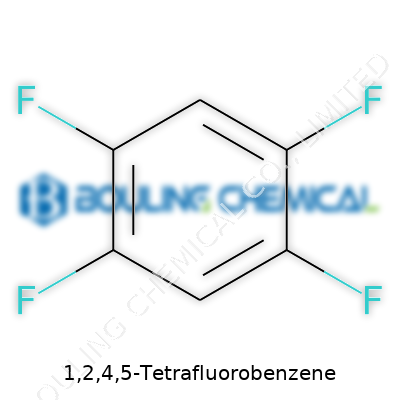
The compound itself looks simple on paper: a benzene ring with four fluorine atoms hanging off it at the 1, 2, 4, and 5 positions. This small tweak makes a big difference. Fluorinating benzene ramps up stability while keeping reactivity in certain positions, which proves handy for making specialty chemicals. It doesn’t get much press compared to Teflon or Freon, but inside R&D labs, its value keeps growing.
1,2,4,5-Tetrafluorobenzene comes across as a colorless liquid with a characteristic aromatic smell, reminiscent of other light fluoroaromatics. The boiling point ranges around 85 to 90 degrees Celsius, which means it sits lower than regular benzene. Fluorine’s presence beefs up chemical resistance—unlike regular benzene, it shrugs off a lot of harsher acids and bases. It barely dissolves in water, but shows plenty of affinity for organic solvents, which suits folks working with organic syntheses. Vapor pressure readings tell a story similar to other light aromatics, so working with it in open-air settings brings the need for good ventilation due to volatility.
Chemists checking the bottle find a CAS number that keeps things straight: 327-54-8. The purity bar for research hovers around 98% or higher, considering side products can trip up follow-up reactions. Labels usually bear hazard warnings focusing on flammability and potential toxicity, but even in routine settings, users stick with gloves and splash protection. High-performance liquid chromatography and NMR spectra go into the paperwork for those running in regulated environments, especially in electronics or pharmaceutical pre-cursors.
Making 1,2,4,5-Tetrafluorobenzene hasn’t always been straightforward. Early methods required labor-intensive steps—halogen exchange reactions, often swapping chlorine or bromine atoms with fluorine using metal fluorides at elevated temperatures. More modern processes rely on direct fluorination of 1,2,4,5-tetrachlorobenzene, because fluorine gas, while tricky to wrangle, gives the cleanest swap. Electrochemical fluorination catches some attention too, mostly for scale-ups since it sidesteps some harsh reagents. Trying to squeeze the most yield out of every synthesis, chemists keep tinkering with catalysts and solvents for cleaner conversion and less waste.
The real excitement comes when 1,2,4,5-Tetrafluorobenzene acts as a base for more elaborate molecules. The electron-withdrawing power of fluorine changes reactivity patterns; nucleophilic aromatic substitution becomes simpler in some positions, which paves the way for new ligands, specialty monomers, and complex pharmaceutical intermediates. Cross-coupling reactions—think Suzuki or Heck—take full advantage of these properties. The molecule sits at a sweet spot: reactive enough for transformation, but stable against too many unwanted side reactions. Researchers enjoy using this framework to anchor other functional groups, providing fresh material for both industrial and academic labs.
Anyone scouring literature runs into names like para-Tetrafluorobenzene, or sometimes just TFB. Synonyms like 1,2,4,5-TFB and tetrafluoro-1,2,4,5-benzene circulate in patent databases or chemical vendors’ lists. This maze of names might sound trivial, but tracking both historical and modern synonyms cuts down on duplicated effort and keeps literature searches productive.
Fluorinated aromatics demand respect in handling. Flash points sit lower than many expect, so storage involves spark-free cabinets and proper ventilation. Chronic exposure brings some eye and respiratory tract irritation, though it hardly counts among the worst offenders in the lab. Still, skin contact and inhalation stay on the to-watch list, and anyone who’s spent time on the bench knows that complacency invites trouble. Gloves, goggles, and fume hoods mark the baseline—no shortcuts, even for seasoned hands. Disposal lines up with halogenated organics, which keeps waste treatment on its toes. Regulatory folks keep close tabs on storage volumes and reporting, especially in jurisdictions with heavy chemical management rules.
It’s fascinating how 1,2,4,5-Tetrafluorobenzene found its niche. Chemical engineers use it as a stepping stone to prepare new heat-resistant polymers, especially for coatings or electronics where stability under stress or heat isn’t optional. Pharmaceutical companies keep it in reserve for modern agrochemicals and drug intermediates; those fluorine atoms, after all, often strengthen biological activity, toughen metabolic stability, or temper overall molecular “stickiness” in the body. As someone who’s followed the rise of fluoroaromatic chemistry, it’s clear this molecule doesn’t need to operate alone—it plays its part in creating modern materials, advanced dyes for OLED displays, and components in battery electrolytes. Each year, fresh papers layer on new uses, and patents reflect an unexpected diversity of applications.
Much of the growth in this field traces back to the constant push for smarter, more efficient synthetic routes. In both academia and industry, scientists have learned plenty from experimenting with multifluorinated benzenes. The molecule’s clean substitution pattern actually lowers the hurdles in methodology development—for catalysis, materials science, and “green” chemistry. Screening new solvents, surfactants, and catalysts happens faster when old bottlenecks are swept aside, which pushes forward fields like organic electronics and specialty polymer synthesis. Working with tetrafluorinated aromatics has taught me that these advancements often leapfrog across applications, energizing pharma, electronics, and even crop protection research at the same time. Multi-disciplinary teams grab this molecule as a test case when chasing new reaction mechanisms and sustainability goals.
The toxicology story for 1,2,4,5-Tetrafluorobenzene continues to unfold. Direct studies on acute or chronic toxicity sit in the shadow of better-known fluorinated aromatics. Animal testing flagged eye and respiratory irritation, with systemic effects usually emerging only at high doses. Long-term studies still need the spotlight, as medical toxicologists have learned the hard way that halogenated aromatics can surprise. Regulatory agencies ask for robust data before greenlighting ton-scale applications, which fuels continued research in environmental fate and breakdown pathways. The compound’s low water solubility slows environmental migration, yet persistence in soils and slow breakdown means periodic reviews of disposal rules. For chemists and industrial hygienists sharing anecdotes from the field, the consensus runs toward caution while deeper data collection keeps refining best practice.
Staring down the next decade, the outlook for 1,2,4,5-Tetrafluorobenzene feels anything but static. Demand builds as more industries crave fluorinated backbones for better performance, especially in energy storage, specialty polymers, and medical compounds. Sharp minds in green chemistry target cleaner synthesis, using renewable feedstocks or less aggressive methods. Regulators and safety experts continue pushing for full life-cycle studies, which nudges companies into transparent monitoring and smarter waste handling. Academic labs keep prodding at the boundaries, hunting for new reactions and applications that could disrupt established markets. Nobody expects this molecule to step into the spotlight of household recognition, but in the hands of innovative scientists and engineers, its role keeps expanding with each technical leap.
People often talk about the big leaps in technology from a consumer point of view—sleeker phones, lighter laptops, or stronger batteries. What gets missed is the intricate chemistry powering all these advances. 1,2,4,5-Tetrafluorobenzene, with its simple six-carbon ring and four fluorine atoms, isn’t something you see on store shelves, but its impact reaches deep into modern industry. Its usefulness is rooted in the way those fluorine atoms tweak the reactivity of the benzene ring, making it a linchpin in several valuable chemical pathways.
In the early days of drug research, scientists stuck closely to what Mother Nature offered. That changed with the invention of clever molecules capable of dodging biological breakdown or aiming for specific molecular targets. Compounds like 1,2,4,5-Tetrafluorobenzene act as building blocks in this process. Chemists use it to craft more complex molecules, including those leading to life-saving medicines. The presence of fluorine atoms often increases the metabolic stability of a drug, allowing it to stay in the body longer and work more effectively. Recent years have seen an uptick in approved drugs with these types of fluorinated rings because they boost performance without loading drugs with unwanted side effects.
Think about the last time you noticed a cable with insulation that seemed to shrug off high temperatures, or cookware that food just couldn’t stick to, no matter what. These don’t happen by accident. Polymers designed from fluorinated aromatics, including 1,2,4,5-Tetrafluorobenzene, provide the backbone for advanced plastics and coatings. The chemical stability imparted by fluorine atoms makes these materials resilient against heat, chemicals, and weathering. This makes them popular for electronics, aerospace, and automotive manufacturing. Their resistance extends the work life of components, cutting down waste and easing the maintenance burden.
Pest control has a bad reputation—often for good reason. Runoff, toxicity, buildup in the soil: these are common complaints tied to heavy use of basic chemicals. Modern agrochemicals aim for a better balance between effectiveness and responsibility. The inclusion of fluorinated benzene rings allows the crops to stay protected, but with molecules that break down in more predictable ways. 1,2,4,5-Tetrafluorobenzene serves as a practical starting material for synthesizing new crop protectants, making safer and more potent solutions a reality.
Looking at these applications, it’s clear that this compound figures into everyday life, even if most people never hear its name. Demand for greener, more sustainable chemistry puts pressure on researchers to find cleaner synthetic routes to these building blocks. Smarter recycling of fluorinated chemicals, greener production processes, and thoughtful regulations will help industry harness the benefits without taking an environmental wrong turn. The conversation around 1,2,4,5-Tetrafluorobenzene is just one chapter in the broader story of how foundational chemistry shapes safer, more durable, and more efficient technology for everyone.
Organic chemistry class had a way of showing which molecules wanted to stay simple and which ones liked a twist. Benzene, that familiar six-carbon ring, sits among the crowd-pleasers on the periodic table. Put four fluorine atoms on it, though—specifically at the 1, 2, 4, and 5 positions—and it changes its tune in meaningful ways. The formula gets straightforward: C6H2F4. Its molecular weight lands at 150.08 grams per mole. These numbers look almost clinical, but they reveal a ton about why folks in labs and industry lean toward compounds like this one.
The reason I keep these numbers memorized has a lot to do with how often fluorinated aromatics pop up in my own lab work. Even swapping out just one hydrogen for a fluorine on a benzene ring changes how that molecule reacts. Four fluorines dial everything up—not just the electronegativity, but also its stability under harsh conditions. That kind of resilience sets 1,2,4,5-tetrafluorobenzene up for roles that more basic compounds can’t pull off, especially when building up more complicated molecules in pharmaceuticals or specialty polymers.
It’s not some rare gem hidden in niche chemistry journals. I run into it when synthesizing intermediates for agrochemical research. Clean fluorination can block unwanted side reactions, and that simple switch in composition—from, say, C6H6 to C6H2F4—often spells the difference between cleanup headaches and a smooth day in the lab.
Fluorobenzenes like this one play their part in materials science as well. Think about how tricky it is to make a polymer that stands up to heat, solvents, or even plain old weathering. The presence of fluorines at multiple locations on the ring means more chemical resistance—something critical in electronics manufacturing and high-performance coatings.
It’s important to weigh the benefits against the environmental counts. Fluorinated organics stack up in the environment if they’re not managed well. Even though 1,2,4,5-tetrafluorobenzene doesn’t have the same persistence as some more infamous fluorinated compounds, responsible disposal, and clear documentation of handling procedures still matter. My own experience has taught me that detailed recordkeeping and open conversations with waste management teams help stop problems before they begin. Colleagues who cut corners sometimes end up with regulator headaches or worse—so careful stewardship goes hand in hand with advanced chemistry.
As researchers chase new ways to replace older, more hazardous substances, chemicals like 1,2,4,5-tetrafluorobenzene find new niches. Its straightforward formula masks a complexity that comes from its placement in larger systems, in real labs, and in the evolving regulatory landscape. The more I work with fluorinated chemicals, the more I see the pattern: smart design, careful handling, and respect for what these small molecules can accomplish—both for good and for the challenges they present on the environmental front. Strong foundational knowledge, combined with responsibility in the workplace, shapes the future for safe and effective chemical innovation.
My years around labs have taught me respect for aromatic compounds, and 1,2,4,5-tetrafluorobenzene stands out as one that deserves a careful approach. At first glance, it doesn’t look much different from other clear chemicals on the shelf. But this fluorinated solvent brings real health risks and, like many volatile organics, will not show mercy for clumsy hands or shortcuts.
Folks sometimes think gases with no obvious flames or choking fumes can’t hurt you much. That’s not the case here. Vapors from tetrafluorobenzene pack a punch, reaching the lungs before you realize what’s happening. Breathing problems, headaches, and nausea hit fast, and over time, even low exposures add up. Proper ventilation isn’t some luxury add-on—it's the main line of defense between you and harm. Standard fume hoods, regularly checked, pull away vapors that build up on busy benches.
I’ve seen enough accidents that started with a careless choice of storage bottle. Polyethylene and glass containers with tight, chemical-resistant seals keep tetrafluorobenzene in its place. Double-check the lid. Avoid metal caps or ones that react with corrosive vapors. The shelf isn't a dumping ground; flammable cabinets, rated for volatile chemicals, provide the best spot. One big lesson I’ve learned: store only the amount you need. Overfilled shelves invite spills and confusion.
A “quick test” without gloves sometimes turns into a hospital visit. Nitrile or neoprene gloves handle tetrafluorobenzene better than latex. Splashes sting, and some cases lead to serious burns or longer-term skin damage. Eye protection is standard, not optional—chemical splash goggles or face shields leave no gap for surprise accidents. If a spill hits skin, rinse for at least 15 minutes even if it feels minor.
Waste disposal still trips up even experienced lab workers. Tetrafluorobenzene doesn't belong in the sink, ever. Specialized chemical waste containers with clear labeling prevent confusion and keep hazardous materials from ending up in groundwater or municipal waterways. Professional waste disposal companies know how to neutralize and incinerate this chemical. At my first lab, we tracked every drop in a log; this habit has stopped many mishaps.
Nobody works with this chemical alone—from procurement to disposal, oversight and good habits stop disasters. Training matters far more than fancy equipment. Newcomers covering their bases keep each other out of danger; old-timers can get complacent and cut corners. I’ve seen it happen. The best teams hold themselves to high standards and insist on regular safety reviews.
Education and reminders make the real difference. Posting visible signage, keeping PPE stocked, and maintaining regular inspections bolster a culture of safety. Shared checklists help, but it’s watching out for each other—pointing out an uncapped bottle, replacing a cracked glove—that stops problems before they grow. Greater transparency and reporting of small incidents build trust and lead to fewer close calls.
1,2,4,5-Tetrafluorobenzene rewards respect and punishes negligence. Rely on hard-won habits, proven procedures, and collective responsibility. Experience says chemicals aren’t forgiving—and neither should you be in setting the bar for safe handling and storage.
Anyone who’s spent time working with specialty chemicals like 1,2,4,5-tetrafluorobenzene will tell you how much complexity sits beneath a simple-looking molecular formula. With four fluorine atoms replacing hydrogen on the benzene ring, this aromatic compound brings a certain resilience and attitude to the table.
You spot it as a colorless liquid, which almost makes it seem unimpressive at first glance. Don’t let appearances fool you. The structure creates a different set of expectations compared to the classic benzene most of us first met in a textbook. Fluorination packs a punch. The fluorine atoms make the ring less inviting for reactions, boosting chemical resistance and making it tough against strong acids and bases. That trait draws out possibilities in the industries that demand durability.
A big part of what makes 1,2,4,5-tetrafluorobenzene valuable comes down to its stability. That stability comes from the electronegativity fluorine brings. If you handled regular benzene, you’d know about its volatility and typical health concerns. Add fluorine into the mix, and volatility drops quite a bit, so you worry less about vapor sneaking away in typical conditions.
The high boiling point compared to benzene tells the same story. With more fluorines, intermolecular forces ramp up, making the compound hang onto its liquid state longer as temperatures rise. For anyone who’s mixed chemicals in a lab, that means a safer process, more precise temperature control, and better product yield.
There’s still flammability in play—organic solvents rarely shed that problem—but the combustion profile changes by quite a bit. You don’t smell much, either, since many fluorinated aromatics barely give off an odor. That silent quality fits well in places where low sensory impact matters.
Take fluorinated aromatics into electronics, and the tough ring helps components survive harsh solvents and aggressive processing. In pharmaceuticals, even a slight tweak to a molecule can change its properties. Fluorinated intermediates like this one have opened doors for researchers creating drugs with enhanced stability or tweaked biological profiles. The carbon-fluorine bond just refuses to budge under metabolic stress.
From personal lab hours, you learn to respect the stubbornness of these molecules. Synthesis relies on good planning, since direct substitution on the benzene ring can get complicated fast. Unwanted side products creep in if you cut corners. Purifying the real stuff calls for reliable chromatographic gear—and some patience.
Where durability shines in the lab or factory, it also raises big questions for the world outside. Fluorinated compounds have a reputation as environmental troublemakers. Their persistence means contamination hangs around in soil and water if spilled or mismanaged. I’ve seen research alerting us to low biodegradation rates. That sets up a bigger discussion about how to handle waste streams and accidental releases.
Careful storage, spill plans, and ongoing monitoring become essential long before shipping chemicals like this. My advice: pay attention to evolving regulations and prioritize systems for recycling or destroying fluorinated waste. We can’t afford to cut corners here, since the effects can stack up over time.
Building safer chemical processes, shifting to greener synthesis routes, and developing reliable detection tech for even trace amounts—every step counts. We all share the responsibility to keep the benefits high and the risks low. Knowledge and respect for compounds like 1,2,4,5-tetrafluorobenzene form the backbone of better science and safer communities.
People talk about chemicals all the time, often with a mix of concern and confusion. One compound, 1,2,4,5-tetrafluorobenzene, pops up in industry circles for uses in synthesis or as a solvent. The question isn’t just where it’s used, but whether this compound is something to worry about—both for our personal health and for the world outside our back doors.
Most folks scanning the label on a drum of chemicals like to spot signs about toxicity, irritancy, or long-term health effects. For 1,2,4,5-tetrafluorobenzene, research remains thin. Publicly accessible databases like PubChem or the OECD eChemPortal offer only patchy toxicity profiles. There’s a basic principle I learned while reading up on chemical risk: if the compound isn’t well studied, caution should rise. That idea comes straight from stories about chemicals thought harmless in the past, only to make headlines after serious problems turned up later.
Known analogs in the fluorinated benzene world can cause irritation to skin, eyes, or respiratory passages. Some related fluorinated organics even act as persistent pollutants or disrupt normal cell function. Taking into account the limited data but recognizing patterns in similar compounds, it’s tough to shrug off safety measures. Even labs stick to gloves, goggles, and good ventilation, because the real effects might take years to pin down.
There’s more to chemical safety than a single user’s exposure. These fluorinated chemicals last. I spent time researching water systems around manufacturing towns and found that stable compounds, even in tiny amounts, stick around in soils or water. If a factory spills or leaks tetrafluorobenzene, microbes rarely break that molecule apart. Fluorine loves to cling tight, making these chemicals slow to disappear. The same trait that gives them industrial value—durability—works against the health of rivers, lakes, and groundwater.
Previous generations saw similar problems play out with compounds like PCBs or certain PFAS. The lesson in those stories: don’t assume a new variant is safer just because it’s less famous. Regulatory agencies often play catch-up, waiting for long-term evidence before putting controls in place. The smart approach looks at environmental persistence and bioaccumulation early, not just after there’s trouble downstream.
Workers and neighbors deserve clear rules. Best practice calls for tight controls on spills, robust air-handling systems in manufacturing, and strictly managed waste disposal. Labs should push for better data—it’s basic responsibility. Proper storage, labeling, and emergency measures should become habits, not suggestions.
Folks with a seat at the table—policymakers, employers, researchers—need to press for more studies before these compounds hit wide circulation. A community that values clean water and safe workspaces won’t regret a slow approach. It’s one thing to miss out on a shortcut in manufacturing; it’s another to find your town saddled with cleanup decades later.
Wisdom means leaning into prevention. With 1,2,4,5-tetrafluorobenzene, clear answers stay elusive. No news isn’t always good news—sometimes it means the important questions haven’t been asked yet.