The journey of 1,2,3,4-tetrafluorobenzene traces back to the post-war boom in organic fluorine chemistry. Once researchers in the 1940s began exploring ways to manipulate aromatic rings, compounds like tetrafluorobenzenes started to appear in patent literature and scientific journals. The push for better refrigerants, high-performance polymers, and pharmaceuticals lit a fire under the search for new fluorinated aromatics. Over time, improvements in synthesis technology allowed chemists to prepare tetrafluorinated benzenes on larger scales, setting the stage for innovations in agrochemicals and materials science. This compound didn't rise to fame overnight, but patient work by chemists and chemical engineers transformed it from a curiosity to a reliable intermediate.
These days, 1,2,3,4-tetrafluorobenzene stands out in the toolbox of organic chemists thanks to its unique mix of reactivity and stability. With the presence of four fluorine atoms attached to the benzene ring, this molecule resists unwanted reactions, which often gives synthetic chemists a lot of control. Its stiff aromatic backbone—studded with electron-withdrawing fluorines—shrinks the electron cloud, changing the reactivity compared to non-fluorinated benzenes. Once you see this stuff in the lab, you notice its clear appearance and a strong, sometimes sharp smell. It’s stable under shelf conditions, not especially volatile, but warrants the same respect as other volatile organics.
Every time I’ve handled 1,2,3,4-tetrafluorobenzene, reliability stands out. It’s a colorless liquid at room temperature, dense compared to water, yet not so heavy that it feels awkward to work with in a flask. Boiling and melting points fall in the moderate range, not too low to make workup tricky and not so high you worry about decomposition during routine tasks. Its solubility leans toward organic solvents, not water, which shapes the kind of downstream chemistry you do. In terms of stability, it behaves predictably in the presence of air or typical light exposure, which saves headaches compared to some less robust fluorinated compounds.
Walk into any storage area, and you’ll spot 1,2,3,4-tetrafluorobenzene by its plain label, typically listing its CAS number and format—nearly always a liquid. The chemical formula—C6H2F4—and molecular weight stay top of mind for anyone doing stoichiometry. Labs list safety categorizations and may flag the compound for flammability and inhalation risk. It doesn’t have a strong hazard profile compared to some organometallics or classic chlorinated solvents, but established protocols keep workers safe by preventing skin contact and excessive vapor exposure. RFID tags or barcodes help modern labs track containers as they move from storage to fume hoods and eventually to waste streams.
The synthesis of 1,2,3,4-tetrafluorobenzene usually starts with a benzene ring, then proceeds through stepwise halogen exchange or dehalogenation. For decades, the Balz-Schiemann reaction formed the backbone of aromatic fluorination, but with advances in electrochemical and catalytic fluorination, safer and more selective routes became accessible. Modern plants often use controlled halogen exchange, sometimes starting with tetrahalobenzenes and swapping out chlorine or bromine atoms for fluorine under carefully controlled temperature and pressure. These steps produce reliable yields and avoid the need for more hazardous reagents, which matters during upscaling for industrial jobs.
Fluorinated benzenes don’t just sit idle on shelves. 1,2,3,4-tetrafluorobenzene plays the role of a versatile intermediate in cross-coupling and nucleophilic aromatic substitution reactions. Its ring fluorines activate the compound for further chemical modifications, which lets synthetic chemists build out more complex molecules for everything from electronics to crop protectants. I’ve seen researchers harness it as a key step in synthesizing advanced liquid crystals and specialty polymers, where the precise placement of fluorine alters melting points, durability, and optical properties. Its compatibility with modern transition-metal catalysis pushes boundaries, leading to the rapid assembly of novel molecules.
Every time I pull a container from a global supplier, I check for alternate names—1,2,3,4-tetrafluorobenzene goes by a handful. Chemists know it by its IUPAC label, but you’ll see folks refer to it simply as tetrafluorobenzene, sometimes specifying the isomer just for clarity. International sourcing highlights the differences, too, with European and Asian stockists listing slightly varied product titles. Awareness of synonyms—like 1,2,3,4-tetrafluorobenzol—streamlines literature searches and ensures you don’t accidentally order the wrong material or misinterpret a synthetic procedure.
Respect for fluorobenzenes in any lab or factory is non-negotiable. Minimal volatility doesn’t mean minimal risk—direct contact can irritate skin, and inhalation, especially in poorly ventilated rooms, poses hazards. Labs and plants employ standard precautions: chemical-resistant gloves, proper eye protection, high-efficiency ventilation. Storing away from incompatible reagents—especially strong bases and reactive metals—heads off trouble. Because fluorinated aromatics sometimes react unpredictably with metals, I’ve learned the hard way not to use aluminum or magnesium tools in their vicinity. Waste handling needs careful attention due to the environmental impact of fluorinated organic solvents, and clear emergency procedures get posted where spills could happen.
This compound shines in the field—from specialty polymers found in electronics to advanced coatings that resist heat and chemical attack. Its core role as a building block shows up in everything from pharmaceutical intermediates to agrochemical launches. I’ve seen projects where its use slashes synthesis steps or produces materials nobody expected would work outside of a research lab. Recent interest in renewable energy materials and robust membranes for water treatment draws attention back to these fluorinated aromatics. The ability to fine-tune physical and chemical properties just by shifting fluorine atoms around gives manufacturers more control over end-user functionality, raising the bar in electronics, optoelectronics, and chemical processing equipment.
Ongoing research on 1,2,3,4-tetrafluorobenzene includes not only new synthetic methods but also fresh applications. Labs keep pushing for greener, more efficient ways to make and use this compound, especially in replacing harsher reagents. New catalytic approaches, including nickel and palladium systems, open doors for milder conditions and improved selectivity. R&D teams experiment with integrating the compound into next-generation OLED displays and sensor devices. Universities and private companies alike dig into the subtle effects of fluorine placement, searching for breakthroughs in pharmaceuticals or polymer science. Innovations in waste minimization and recycling promise to keep these developments environmentally responsible.
No commentary on a fluorinated aromatic would be complete without looking at its safety profile. While fluorobenzenes generally have low acute toxicity, chronic effects and long-term exposure raise questions. Lab animals show elevated risks under extreme doses, prompting researchers to call for better monitoring in occupational settings. Environmental persistence remains an issue; these compounds resist breaking down under typical conditions, fueling global debates about the handling and disposal of organofluorines. Regulatory bodies expect solid data from toxicity studies, so anyone working with these materials stays alert to the latest recommendations and adopts best practices to minimize exposure and waste release.
1,2,3,4-tetrafluorobenzene finds itself poised for bigger roles as industries look to advanced materials demanding stability, chemical resistance, and fine-tuned electronic properties. Continued research into greener processes, combined with tighter regulations on fluorinated waste, shapes how companies approach its use and lifecycle. The demand for high-performance polymers, specialty coatings, and materials for emerging electronics keeps innovation alive. Stakeholders—be they academic, industrial, or regulatory—stand to gain from transparent communication, improved synthesis, and stronger attention to sustainability. The ongoing story of this compound blends scientific rigor, practical insight, and responsibility for future generations.
Fluorinated chemicals have shaped several of the things we use every day. Among them, 1,2,3,4-tetrafluorobenzene isn’t exactly a household name, but its footprint runs wide, quietly supporting industries focused on new materials and next-generation pharmaceuticals. Growing up in a manufacturing community, you see how progress relies on specialized compounds. Sometimes, a single molecule makes all the difference, and in the case of tetrafluorobenzene, its importance stands out to any chemist chasing precision and stability.
Talking to polymer scientists over the years, the one thing that comes up: fine-tuned monomers shape the backbone of plastics built to survive heat, corrosive chemicals, and sometimes fire. Tetrafluorobenzene plays a strong hand here, serving as a building block for specialty polymers like PBI (polybenzimidazole) and polyimides. These materials pop up in aircraft, chip manufacturing, and filtration membranes. The reason? Many fluorinated rings hold up when ordinary plastics give way to warping and cracking. The presence of four fluorine atoms also tunes solubility and reactivity, opening the door to custom-made plastics for demanding jobs.
Developing good drugs often runs into a wall: stability, selectivity, or how fast the body chews through a promising molecule. Medicinal chemists I’ve worked with stress the value of fluorine in “tweaking” these properties. 1,2,3,4-Tetrafluorobenzene stands out as a route to scaffold-like structures that resist metabolism and fine-tune activity, especially for cancer drugs and anti-inflammatories. Reports from journals such as the Journal of Medicinal Chemistry highlight cases where fluorinated aromatic rings, using intermediates like tetrafluorobenzene, offer a shot at new treatments by boosting drug lifetime or targeting.
Lab realities mean chemists constantly search for reaction conditions that coax the right bonds without wrecking sensitive groups. Fluorinated solvents stand up to tough situations—think high-voltage electronics or reactions that hate water. Some labs use tetrafluorobenzene as a niche solvent because it brings high thermal stability and sometimes helps separate closely related compounds, trimming both waste and cost. It’s not the top pick for big industrial runs due to tighter supplies and price, but it supports development work where nothing else works.
Every advance brings its own set of concerns. Fluorinated compounds linger in the environment, earning attention from health and safety boards. The science community and regulators keep a wary eye on how these materials move through water and soil. As a high-value building block, tetrafluorobenzene gets used sparingly, but the pressure grows for better ways to handle and recycle process waste. In my work, I’ve seen teams switch over to closed systems and new scrubbing technologies to curb emissions and run cleaner labs. This push combines technical know-how with a need for leadership that doesn’t put short-term profit ahead of community well-being.
As new electronics and drug candidates roll out, the demand for fluorinated rings—especially ones that can be easily built up or swapped—likely grows as well. Companies and researchers look for safer ways to make and use these chemicals, balancing innovation against stewardship. This conversation matters to everyone: a smart approach to specialty chemicals like tetrafluorobenzene opens doors, but the real win comes from pairing discovery with care for the environment and public health.
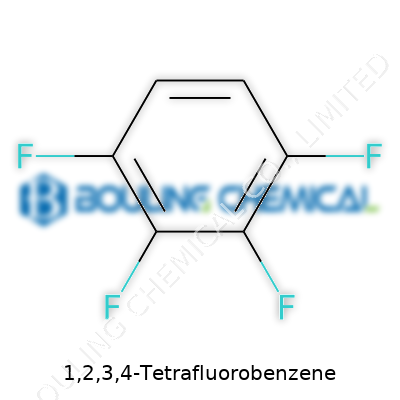
Chemistry sometimes feels like a mystery novel. Names like 1,2,3,4-Tetrafluorobenzene sound complicated, but the building blocks behind them hold stories that touch everyday life, research, and industry. The chemical formula for this compound is C6H2F4, and its molecular weight stands at 150.08 g/mol. The precision here matters, not to show off technical jargon, but to lay down a foundation for clear understanding and safe handling in laboratories and industries.
I spent my formative years tinkering with chemistry. That sense of curiosity never faded. On my own lab bench, the challenge wasn’t just to know the formula, but to appreciate how structure turns into use. Four atoms of fluorine attached directly onto a benzene ring changes everything. You get more than just a rearrangement — the properties transform. Fluorine’s electronegativity alters the way this molecule interacts with other substances. It gets more resistant to breakdown and less likely to catch fire, which matters if you’re working with volatile chemicals.
This altered behavior attracts attention from researchers hunting for new materials or novel solvents. In pharmaceuticals, tweaking a benzene ring by adding fluorines sometimes improves drug stability or helps a medicine get precisely where it’s needed in the body. Not every experiment leads to a breakthrough, but the story of scientific progress always draws from compounds like this — established, reliable, yet open to innovation.
Questions of safety pop up right alongside curiosity. Fluorinated aromatics aren’t just benign substances. Environmental research flags these types of chemicals for their persistence. Once you set a molecule loose that doesn’t break down easily, it can linger in soil, water, or living tissue. Some related compounds have already shown up in environmental monitoring programs, triggering ongoing debates about long-term impact. My personal take: every new molecule, no matter how useful, deserves close attention during both development and disposal.
While many researchers focus on positive application, the other side calls for thoughtful handling and disposal practices. Modern labs lean on data gathering and monitoring to keep workspaces and communities safer. With proper storage, trained staff, and clear protocols, most risks remain manageable.
Getting the facts right starts with things as straightforward as a formula and a number. C6H2F4 doesn’t tell the whole story, but it leads to questions about manufacturing, application, safety, and environmental impact. There’s no shortcut to trust—facts, transparency, and a willingness to adjust guidelines as evidence emerges build credibility for science and industry both.
The conversation isn’t just for chemists. Anyone whose life or business might intersect with new materials should ask about the full picture: what a substance is, how it’s used, and what happens after use. Real progress means seeking answers and staying vigilant, even for those compounds whose names rarely make the headlines.
Fluorinated chemicals like 1,2,3,4-tetrafluorobenzene might sound like something out of a chemistry textbook, but for researchers and workers in certain industries, dealing with compounds like this is just part of the routine. These aren’t compounds you leave on a shelf next to cleaning supplies or snacks back in the break room. From the sharp odor to the volatility, you can tell 1,2,3,4-tetrafluorobenzene needs to be taken seriously.
The most pressing risks with tetrafluorobenzenes come from inhalation or accidental exposure, as these chemicals don’t play nice with your lungs or skin. If you store it somewhere damp, warm, or full of sunlight, you’re courting trouble. Over time, heat or moisture can damage the container. If that container breaks or leaks, toxic fumes won’t ask for your permission to drift through the building.
This stuff deserves a spot in a dedicated chemical storage area with solid ventilation and steady room temperature. No open flames, sparks, or loose electrical cords; this isn’t the place for shortcuts or the “it’ll be fine just this once” approach. Keeping incompatible chemicals away isn’t paranoia—it’s just common sense. Certain metals or oxidizers could make a bad day much worse if stored too close to tetrafluorobenzene.
I’ve spent years around labs and industrial spaces where safety protocols save lives every day. You want everyone on your team to walk out with the same number of fingers and working lungs they started with. That means splash goggles, gloves made for chemical work, and lab coats are more than a “nice to have.” Too many workers skip these basic steps and roll the dice.
As soon as you’re dealing with anything that can emit hazardous fumes, local exhaust goes from a bonus feature to an absolute must. Whether you use a fume hood or a full-room ventilation system, it’s worth the investment. Keeping the workspace clean and well-organized doesn’t just impress the safety inspector; it stops contamination and chaos before they start.
Even careful teams can face unexpected spills or leaks. Having the right absorbents, containment tools, and spill kits within arm’s reach beats scrambling after an accident. Documenting incidents is tedious, but it’s the only way to spot patterns and fix mistakes before they pile up.
Anyone storing or using 1,2,3,4-tetrafluorobenzene owes it to themselves and their neighbors to know local and national rules. There’s no getting around the reporting and labeling standards, and dodging regulations puts everyone at risk, not just your bottom line. Training isn’t just a checkbox—making chemical hygiene second nature for newcomers and experienced hands goes a long way.
Tough problems like chemical accidents rarely have a single magic answer. Consistent inspections, real training, and the occasional policy shakeup can make a big difference. Everyday safety habits help keep people safe. If you’re looking to protect your team, your building, and your peace of mind, taking 1,2,3,4-tetrafluorobenzene seriously from day one pays off.
Walking into a lab for the first time, exotic names like 1,2,3,4-tetrafluorobenzene can intimidate. It’s easy to get caught up dwelling on the “fluoro” part and wonder if you’re about to handle something out of a sci-fi movie. Few people outside of advanced manufacturing, chemistry, or polymer research have ever cracked open a bottle with this mouthful of a compound. For those who do, the natural question becomes: What sort of risks are we weighing?
Chemicals in the benzene family often raise red flags. Benzene itself is unquestionably toxic and associated with cancer after long-term exposure. Fluorination changes things up; adding four fluorine atoms brings different properties and potentially different risks. According to the European Chemicals Agency and other published sources, 1,2,3,4-tetrafluorobenzene hasn’t shown significant toxicity in published animal studies compared to unaltered benzene. There’s no widespread record of it causing cancer, genetic damage, or developmental harm. You probably won’t find it on lists of the most concerning industrial chemicals or flagged in the same breath as substances like asbestos, lead, or PCBs.
That doesn’t mean it gets a free pass. It’s a small organic molecule with potential to evaporate and enter the air. No one wants to inhale uncontrolled vapors, even if only low-level irritation results. In the places where I’ve seen chemicals like this used – research labs, manufacturing pilot plants, and large-scale chemical syntheses – the safety mantra is always the same: minimize exposure. Gloves, goggles, fume hoods, locked cabinets. Spend enough time around experienced chemists and “out of the bottle, into the hood” becomes automatic.
Agencies in North America, Europe, and Asia haven’t added 1,2,3,4-tetrafluorobenzene to urgent chemical watch lists. No special disposal rules appear in regulations outside the general hazardous waste category most organic solvents fall under. That does not mean it is safe enough for casual handling. Fluorinated organic compounds can often hang around in the environment, breaking down slowly. Just ask people who’ve dealt with old refrigerants or PFAS contamination. Responsible use matters, even if the immediate health hazards seem minor compared to other chemicals.
Nobody working in science or industry should gamble on “not proven hazardous” as the only safety criteria. One decade’s safe solvent sometimes turns into the next decade’s regrettable pollutant. Most toxicity tests focus on acute effects and sometimes miss subtle long-term outcomes. The lack of high-profile alarms around this compound doesn’t excuse taking shortcuts. Wise workplaces invest in training, spill containment, and regular review of chemical inventories. The idea is to lower risk at every step, not just follow minimum legal standards.
The best solution always circles back to finding less hazardous substitutes where practical. Researchers spend real effort screening safer solvents or designing new chemicals with rapid breakdown and low toxicity. That pursuit takes time, and nobody should expect instant answers baked into regulatory lists or safety data sheets. Meanwhile, treating 1,2,3,4-tetrafluorobenzene with respect, containing exposure, and reviewing new science regularly feels less like bureaucracy and more like smart, future-proof working.
It’s easy to get lost in long names like 1,2,3,4-Tetrafluorobenzene, but beneath the mouthful lies a fascinating chemical. Companies and researchers pay attention to compounds like this not for the challenge of pronouncing the names, but for what those arranged atoms mean in the real world. Four out of six hydrogen atoms on the benzene ring have been swapped for fluorine. This type of tweak can completely reshape how a molecule behaves, how stable it feels, and even its impact on the environment.
Take a bottle of this compound in a lab and you’ll spot a clear, colorless liquid. The melting point sits much lower than its non-fluorinated cousin, benzene, making it one of those chemicals you’re more likely to see as a liquid than a solid under normal conditions. This surprising shift comes straight from what those extra fluorine atoms do. Fluorine packs a real punch — it pulls in electrons and makes the molecule more rigid and less likely to engage in the odd interactions you get from pure benzene.
Boiling points and vapor pressures matter for anybody handling chemicals. 1,2,3,4-Tetrafluorobenzene boils at a lower temperature than benzene. That affects how you store it, ship it, or even how safe it stays on the shelf. In chemistry, volatility shapes everything from lab safety to environmental risk. People familiar with organic solvents know that extra fluorines lead to lighter, less clingy vapors — a feature handy for cleaning or electronics work, but one that raises flags for air quality if not managed well.
You won’t see this compound rushing to react with everything in sight. There’s a reason for that: fluorine’s tightly bound grip. Substituted benzenes with multiple fluorines often resist the usual routes for transformations. While a typical benzene ring might join in all sorts of dance routines with acids or oxidizers, this one sits it out more often. That stubbornness can help or hurt, depending on the goal. For example, it makes these molecules less likely to break down quickly — which works well in durable coatings, but poses a challenge for environmental breakdown down the road.
Stability matters in industry, whether you’re building electronics or searching for better materials in pharmaceuticals. People remember the big shift to chlorofluorocarbons in refrigeration for a reason: fluorinated compounds, used wisely, stick around longer without falling apart. On the other hand, that durability means once released, they don’t go away easily. They find a way into soil, water, and even the atmosphere.
As the world gets serious about sustainability, the chemistry community faces a hard question. Balancing durability and performance with potential long-term risks is tricky. Tracking environmental spread, picking safer alternatives where possible, or developing better recovery processes all matter. Scientists advocate for improved detection and clean-up technologies, pushing regulators to update guidelines with data from real-life monitoring, not just lab tests.
Building trust takes experience and transparency. Safety data, reliable handling techniques, and independent research all help. Open discussion—between chemists, policymakers, and the public—leads people to smarter decisions about both daily use and broader regulations. Compounds like 1,2,3,4-Tetrafluorobenzene show how a few well-placed atoms can shape not just chemistry, but society’s approach to progress and responsibility.