Long before chemists started exploring the corners of organofluorine chemistry, refrigerants and specialty solvents held to simpler molecules like chlorofluorocarbons. After intensive environmental scrutiny chased CFCs from the market in the late 20th century, next-generation compounds turned up—often carrying more fluorine atoms, each reshaping performance and environmental impact. 1,1,1,2,2,3,4,5,5,5-Decafluoropentane emerged during this transitional period. Its introduction mirrored a demand for safer, more stable fluids: lower ozone depletion, mild toxicity, precise volatility—qualities the industry embraced. These compounds, in a sense, represent science’s ongoing conversation with nature, always negotiating between technical necessity and responsibility.
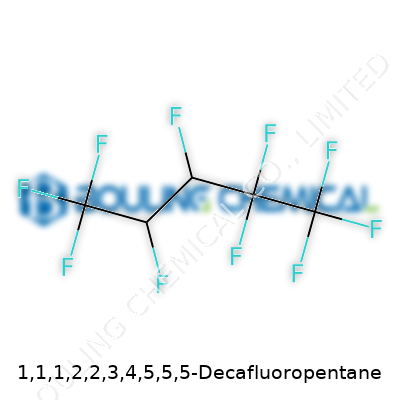
1,1,1,2,2,3,4,5,5,5-Decafluoropentane’s story begins with its fully fluorinated carbon backbone. Revised over the decades, its structure aimed to dodge the breakdown and reactivity that dogged older analogs. As a result, it resists both hydrolysis and combustion remarkably well. This resilience grabbed the attention of sectors from electronics to cleaning, since less reactive fluids often cause fewer headaches in production. You find this molecule under a jumble of names—sometimes called HFC-4310mee or simply decafluoropentane. In each case, its molecular architecture sidesteps much of the instability linked to partial halogenation, promising steady handling if given basic respect.
Anyone who’s worked with perfluorinated compounds knows their signatures well, and decafluoropentane checks many boxes: transparent, faintly sweet-smelling, and heavier than water. This stuff evaporates quickly, hinting at a boiling point suited to gentle drying tasks. As a solvent, its low polarity fits awkwardly with water or alcohols but makes it great for dissolving hydrophobic materials. Chemically, the carbon-fluorine bond keeps it from reacting with acids, bases, and even strong oxidizers in most conditions. It won’t burn easily. You can store it in ordinary steel or glass without corrosion—for once, a specialty chemical that doesn’t quietly eat into the walls of your containers.
I’ve walked through old chemical storage rooms and seen labels yellow and crack, making critical details unreadable. With modern specialty chemicals like 1,1,1,2,2,3,4,5,5,5-Decafluoropentane, accuracy in labeling means more than regulatory compliance—it’s the front line of safety. The industry tends to state purity in percentages, boiling points, flash points, mass densities, and GHS hazard codes. This chemical falls under mild irritant status, so safety data sheets underscore respiratory and dermal guidance. One thing many overlook: decafluoropentane has low surface tension, which means accidental spills spread far and fast, a reality that labeling alone never quite captures.
Scaling fluorinated hydrocarbons like decafluoropentane starts with specialized feedstocks—pentane skeletons and halogenation agents. Early organic chemists struggled with controlling substitution, but modern flow chemistry and selective catalysis turned the tide. Direct fluorination is notoriously aggressive, risking explosions and sidereactions, so safer indirect methods prevail. Electrochemical fluorination, for instance, lets operators dial in coverage and steer clear of toxic byproducts. While batch yields still bump up against the cost of fluorine gas, these methods favor scalability and purity, both linchpins for commercial success.
Bench chemists often use 1,1,1,2,2,3,4,5,5,5-Decafluoropentane as a base for specialty blends—think engineered cleaning agents, heat transfer fluids, or dielectric coolants. Direct chemical modification doesn’t rank high, partly because the strong bonds make further alteration expensive and cumbersome. Instead, the focus shifts towards mixing, not reacting: pairing with co-solvents or surfactants to dial in viscosity, flash point, or cleaning power. The rare occasions where chemical reaction comes into play often revolve around curiosity-driven research, such as studies in perfluorinated compound transformation or waste treatment, but that remains more theoretical than commercial.
For such a technical molecule, it goes by a surprising spread of names. HFC-4310mee gets tossed around in refrigeration circles, while some lists show it as n-decafluoropentane or perfluoropentane. Each comes bundled with standards or regulatory implications—names you see depend on which doors you walk through, whether lab supply, government regulation, or manufacturing dock.
Veterans in chemical handling know the drill: gloves, eye protection, working under a hood, not downwind of vapors. Decafluoropentane’s mild toxicity sometimes nudges complacency, which turns out to be a bad habit. Like other perfluorocarbons, it can crowd out air and create asphyxiation hazards in closed spaces. Fume extraction remains non-negotiable, and accidental release needs prompt clean-up, since those low surface tension properties let it drift across surfaces and into drains. The industry’s learned, often the hard way, that every chemical—even a “safe” one—demands respect and steady adherence to standards.
I’ve watched decafluoropentane drift across multiple sectors—semiconductor cleaning, thermal management in electronics, specialty medical preparations such as contrast agents, and precision optics maintenance. Tech companies value its chemical inertness when cleaning sensitive assemblies. Medical researchers explore its potential as a carrier for oxygen in blood substitutes or ultrasound contrast media. That versatility flows from its core stability, low boiling point, and chemical gentleness. Unlike substances that knock about cells and tissues, the body tolerates small doses, which led to limited but promising biomedical applications.
New information keeps turning up. I’ve followed recent studies focusing on environmental footprint, breakdown pathways, and uses aligned with green chemistry priorities. Some research pushes into nanoemulsion formation, allowing decafluoropentane to ferry otherwise intractable drugs in the body. Others examine roles in heat transport, looking to shape the next generation of cooling solutions as electronics shrink and heat up. These domains show the field leans on collaboration—environmental science partners with engineering, academic labs network with industry, each trying to draw safer, more effective uses out of old molecules. In my own work, reliable journals and resourceful colleagues top any database search when separating theory from what actually works in the field.
As with any specialty chemical, the toxicity question floats over every new application. Earlier studies painted a mild profile—low acute toxicity, basically no flammability, and little evidence of chronic harm at modest exposures. The weak points? Very high vapor concentrations can cause respiratory distress, and bioaccumulation keeps environmental persistence on the agenda. Regulatory bodies clamp down on excessive emissions, especially in markets with strict environmental policies. Many labs now track long-term worker exposure and environmental releases using more refined detection than early studies ever imagined. In my own experience, anyone who treats even low-toxicity chemicals cavalierly tends to run into trouble down the line.
Future prospects for 1,1,1,2,2,3,4,5,5,5-Decafluoropentane hinge on trade-offs: balancing utility with environmental responsibility. Calls for low-global-warming-potential fluids ring louder, and companies hunt for substances that combine performance with lower persistence. Next-generation fluorocarbons may carry chemical “handles” that encourage faster degradation after use. Some innovators look toward biotechnological production routes or recyclable blends, cutting costs and penalties that dog traditional fluorine chemistry. New applications likely sprout wherever energy management and material compatibility intersect—in smaller, hotter circuits, tighter industrial footprints, and more exacting medical science.
Experience teaches that no chemical, no matter how robust, is immune to broader impacts. Staying informed through peer-reviewed literature, regular safety training, and honest conversations with colleagues makes a difference in both lab and plant. Cases where overconfidence led to accidental releases or health scares still surface from time to time. The real progress with chemicals like decafluoropentane comes from integrating careful stewardship—environmental, occupational, and technical—into every step from synthesis to disposal. As regulations and expectations evolve, companies and individuals skip shortcuts and invest in transparency, trust, and technical know-how, drawing lessons from each decade-old bottle and every new challenge.
1,1,1,2,2,3,4,5,5,5-Decafluoropentane doesn't roll off the tongue, but doctors and researchers know it quite well. This compound, a type of fluorinated aliphatic hydrocarbon, carries a lot more weight in healthcare and technology circles than many realize.
Flipping through medical journals over the years, I’ve noticed Decafluoropentane popping up more and more. In ultrasound imaging, this chemical forms the core of many “contrast agents”—those solutions injected into the body so ultrasound scans come through clear instead of fuzzy. Traditionally, these procedures could be hard for some people because older agents sometimes caused allergic reactions or broke down too fast in the bloodstream. Decafluoropentane changed that. Its unique make-up means it stays stable long enough for specialists to get the images they need, while also quickly breaking down afterward and exiting the body without leaving a mess. The safety record, based on published clinical studies, gave clinicians new confidence in using ultrasound in sensitive places like the heart and brain.
Biotech companies experimenting with smarter ways to deliver drugs have latched onto Decafluoropentane for its physical properties. Researchers can load it into tiny capsules, sometimes called nanodroplets, and these sit quietly in the body until a targeted ultrasound beam “pops” them—delivering their payload exactly where it helps most. Take a cancer patient for instance: less spillover to healthy tissues, fewer side effects. Not every patient gets this yet, but a lot of lab work points toward wider use in targeted therapies for conditions like tumors and blood clots.
Looking beyond medicine, this compound turns up in cleaning electronics and high-value optics. Engineers prize it for its electrical insulation and chemical stability. Used correctly, Decafluoropentane helps remove microscopic dust and contaminants from fragile surfaces, extending the lifespan of devices we rely on. It’s a relief compared to older “chlorinated” cleaners, which simmered in controversy due to toxicity and ozone problems. Regulations now tightly control emissions, which encourages safer handling and better protective gear for workers.
Given my own concern about chemical use in everyday life, the story isn’t all positive. Like other fluorinated compounds, Decafluoropentane raises questions about long-term persistence in soil and water. The chemical structure resists breaking down naturally, so run-off from factories needs careful treatment. Studies out of Europe and the US have tracked its movement in water supplies, and it doesn’t disappear quickly. This remains a big sticking point. Companies using it are under increasing pressure to find greener substitutes or to capture and recycle every bit.
Solving the tension between practical benefits and environmental safety takes honest work. Hospitals and manufacturers already limit waste and invest in recycling. It’s slower progress than many would like, but the shift toward “life cycle assessments” for every new application shows that industry is listening. Scientists keep working on new blends that offer the same advantages for imaging and cleaning, just with lighter footprints. If decision-makers keep weighing both sides—improved care and environmental risk—the practical use of Decafluoropentane can remain a story of careful progress, not careless excess.
Every week, someone asks if a handful of unfamiliar chemicals are really safe to be around. One of these, 1,1,1,2,2,3,4,5,5,5-decafluoropentane, has a long name that immediately raises questions. Most people haven’t encountered it, but this fluorinated hydrocarbon has found a place in specialty industries—think electronics cleaning or as a carrier for pharmaceutical uses. Its growing presence has brought some curiosity and, naturally, concern about what kind of risks it brings to people who need to work with it.
A basic fact about 1,1,1,2,2,3,4,5,5,5-decafluoropentane: it’s not known to be acutely toxic in the same way as more notorious cleaning agents or some older refrigerants. So handling it doesn’t mean stepping into the danger zone right away. Yet, calling it “safe” might be premature if you haven’t scoped out the details. It acts much like other fluorocarbons—low flammability, relatively stable, and resistant to breakdown in the environment. These are pluses if you’re worried about things catching fire or dangerous chemical reactions in a workspace.
Reading reports from safety regulators like the US Environmental Protection Agency, it’s clear there’s no big red flag about it causing cancer or nerve damage, at least with the exposures most people see in the workplace. But that doesn’t mean wide-open doors for careless use. Inhaling any solvent vapor isn’t a good daily habit. Most labs, and just about every facilities manager I’ve met, put a strong emphasis on using these kinds of compounds in well-ventilated areas, preferably under a fume hood. Respiratory irritation happens when people toss aside these basics.
Skin contact usually doesn’t cause the chemical burns or nasty rashes you’d see with strong acids, but dry skin or mild irritation can pop up if someone ignores gloves and splashes it around. It’s still a chemical, not water. A chemical splash in the eyes sends people to the eyewash station. These are everyday issues—not dramatic, but enough to push anyone with sense to take protection seriously.
A bigger worry sits with the environment. 1,1,1,2,2,3,4,5,5,5-decafluoropentane doesn’t break down easily. A spill or a dump doesn’t just vanish in a storm drain. Local governments get cranky about uncontrolled disposal and for good reason. Companies need to handle waste streams with a plan that stops leaks and heads off releases into the wider world. Experience shows that cleanups cost more in the end.
The basic rules don’t change: ventilation, gloves, goggles, and good waste collection. Training matters more than the technical name of the solvent. One place I worked kept a laminated card next to each workbench explaining exactly how to handle spills and who to call. It sounds simple, but knowing what to do—before something happens—means fewer mistakes. Relying on regular reminders, clear signage, and actually having a supervisor watch to make sure people use personal protective equipment closes most of the loopholes.
Scientific evidence and long experience back up these precautions. Responsible use means no one has to run unnecessary risks, even if the chemical itself carries fewer hazards than some others. Knowledge, respect for protocols, and planning beat out a label of “safe” every time. That’s true for this compound and almost anything else found in the supply closet.
Chemistry doesn’t always make headlines, but sometimes even a single number—like the boiling point of a molecule—makes a huge difference in science and industry. With 1,1,1,2,2,3,4,5,5,5-Decafluoropentane, we’re talking about a fluorinated hydrocarbon that has carved a stronghold in specialty fields. This compound boils at about 49°C. I remember my first brush with specialty fluorocarbons during a summer research stint. Lab work is full of little surprises, but nothing grabs your attention like a liquid quietly vanishing off the bench, leaving only a cool surface behind. The boiling point is key here—it tells us when a substance jumps from liquid to vapor and starts to mingle with the air.
That 49°C mark shapes the way industries tackle real-world challenges. For solvents used in precision cleaning, a lower boiling point means less energy needed to get things done. I’ve watched engineers obsess about the energy cost of each degree in temperature. Every degree saved on heating large batches adds up: less energy, less cost, fewer emissions. For technicians cleaning delicate electronics, like circuit boards or medical imaging parts, harsh heat isn’t an option. You need a solvent that does its job and disappears before it can cause damage. 1,1,1,2,2,3,4,5,5,5-Decafluoropentane steps up just right—volatile enough to lift residues, gentle enough for sensitive hardware.
Working in research, you start to appreciate how a simple boiling point dictates everything from storage to safety. At room temperature, this compound stays as a liquid but vaporizes with minor heating. That creates both flexibility and risk. Spills mean rapid evaporation, which demands good ventilation. Improper handling in poorly designed labs turns convenience into a hazard. So knowing that 49°C number isn’t trivia; it goes straight to the heart of lab safety and process design.
The world looks at fluorocarbons with a suspicious eye, and not without reason. Many, especially their relatives like chlorofluorocarbons, have hurt the ozone layer. The push for new alternatives is loud—and justified. Decafluoropentane stands out compared with old-school solvents because it skips both chlorine and significant ozone destruction. But it’s not without issues. Perfluorinated chemicals don’t easily break down. That makes the choice to use this molecule a balancing act: high effectiveness, better safety during use, long afterlife in the environment.
We’ve already seen environmental policy force big changes on refrigerant and solvent makers. Now, labs and factories need both clever alternatives and responsible recycling. Closed-loop cleaning and recovery systems cut down on waste. Improved handling means less evaporation during use. I’ve seen facilities invest heavily in capture systems, lowering both exposure and emissions. Regulations push us to keep refining these approaches—good for people, good for the air, not just the bottom line.
Learning to pay attention to boiling points isn’t just exam fodder. Real-life chemistry demands we treat each number as a guide to risks, opportunities, and trade-offs. With decafluoropentane, that 49°C boiling point shapes how products are made, how workers stay safe, and what footprint we leave behind. Better awareness, smarter process designs, and attention to downstream impacts will keep this compound as an aid, not a problem, for years to come.
Fluorinated chemicals like 1,1,1,2,2,3,4,5,5,5-Decafluoropentane play a big role in many industries, from electronics to medicine. They’re tough, stable, and show up in a surprisingly wide range of day-to-day uses. Taking proper care of them always starts with how they’re kept, especially this one, with its long string of fluorines and tricky behavior at room temperature.
This compound acts like a stubborn liquid: it hangs onto its low boiling point and doesn’t much like getting warm. I remember handling similar chemicals in a lab, knowing that ignoring a label or shrugging off storage rules would mean you’d end up with an empty bottle and a room filled with vapors. That’s not just wasteful, it’s risky.
Storing 1,1,1,2,2,3,4,5,5,5-Decafluoropentane really boils down to two things: temperature control and containment. Not fancy words, just basic, back-to-basics discipline. Don’t let it get warm. Ordinary storage shelves in a hot warehouse will just speed up evaporation. Even on a cloudy day, that room can get stuffy, and next thing you know you’re breathing in things you definitely didn’t want to. For a chemical with a boiling point below 50°C, just sitting on a sunlit sill can spell trouble.
Back at the bench, glass or high-density polyethylene usually survive battle with fluorinated liquids, but always double-check the seal. Screw-top lids, not snap-caps or parafilm, keep the vapor inside where it belongs. A leaky jar is an invitation for headaches at best and a dangerous slip at worst if puddles start forming after repeated use.
I get it: nobody loves writing the perfect label. But an unlabeled bottle, shoved in a refrigerator full of similar-looking materials, is a game of Russian roulette. Every accident I’ve heard about in a teaching lab started with the words “I thought it was—.” Putting up with the hassle of clear labeling cuts that story short.
Some folks think a fume hood is only for the fancier, stronger-smelling stuff. That’s a risky game with compounds that give off vapors you barely notice until you get lightheaded. Any spot storing quantities larger than a few milliliters needs solid ventilation. I’ve seen the mess left behind when a forgotten bottle boils in storage and ruptures. Even compact chemical refrigerators put undercounter need exhaust lines when the roster includes things like decafluoropentane.
Things don’t always go to plan. I’ve knocked over enough small bottles to know why keeping material on absorbent, chemical-resistant trays stops minor oops moments from rolling into disasters. A spill turns into a long cleanup if it soaks into cardboard boxes or seeps through worn shelving. Basic spill kits with absorbent pads, gloves, and goggles on standby go a long way toward turning a bad day back into just another shift.
Regulations treat fluorinated substances seriously, and for good reason. These compounds take ages to break down. Environmental releases, even small ones, add up. I’ve seen the frustration when a workplace gets cited after something simple goes wrong: an overfilled waste drum, a dumpster with a cracked liner, or a careless pour down a drain. It doesn’t just cost fines; it undercuts trust in those who should know better. Storing all chemicals with a mind for the planet, not just today’s bottom line, reflects a long-term, responsible mindset.
Keeping 1,1,1,2,2,3,4,5,5,5-Decafluoropentane on hand means practicing what every good lab tech, engineer, or researcher knows: treat volatile chemicals with steady respect, not as afterthoughts. Cool, well-sealed, ventilated, clearly labeled—those basics never go out of style, and neither should paying careful attention with every refill and lock-up at the end of the day.
Headlines rarely pick up on chemicals like 1,1,1,2,2,3,4,5,5,5-Decafluoropentane. Still, this mouthful deserves a closer look. Used in everything from medical aerosols to electronics cleaning, it's one of those fluorinated solvents promising safer alternatives to notorious CFCs and HCFCs. But “safer” doesn’t guarantee harmlessness. Once released, this compound hangs in the air far longer than folks realize. Its chemical bonds, designed for stability, shrug off most breakdown attempts by sunlight or weather. This leads to atmospheric persistence and slow movement through natural cycles.
Ask any climate scientist about fluorocarbons, and you’ll get a pointed response. Decafluoropentane, like many of its cousins, has significant global warming potential. Even if its rate of use stays below the vast volumes poured out by big industries, every gram finds its way into the climate ledger. The Intergovernmental Panel on Climate Change (IPCC) places most fluorinated solvents in the high impact category, thanks to their strong greenhouse properties.
Numbers get fuzzy in real-world use, since companies keep usage volumes guarded and chemical life cycles don’t follow simple math. I learned from talking with environmental chemists that even moderate release volumes can stack up over the decades. Regulators in the European Union and North America started watching these chemicals more closely, not willing to repeat mistakes made with older refrigerants.
Any chemical with this many fluorine atoms stands tough against degradation. In practice, waste streams from factories or hospitals washing out residue send decafluoropentane drifting into water systems. Tests show that once it arrives in rivers or groundwater, it tends to persist. Unlike natural organics that bacteria feast on, these molecules barely register as food. Over time, accumulation becomes a concern, especially for fish and amphibians who don’t get to choose their environment.
Soil contact brings another headache. My discussions with environmental site assessors highlight how fluorinated solvents resist breakdown, even under direct sunlight or after rainfall soaks contaminated soil. This leaves cleanup crews short on options. Modern “remediation” technologies look expensive, and none of them work as efficiently as natural decay would with other types of pollution.
The push to call this chemical “safer” than ozone-depleting predecessors misses the chronic questions. While acute toxicity rates in humans remain low and it doesn’t threaten the ozone layer, the persistence means it will linger in places we don’t want it. I remember a public health officer saying, “You never want a man-made molecule that mammals and bacteria both ignore.” Without fast breakdown, decades from now we may still see traces in food chains.
So what can we do? Substitution with less persistent chemicals leads the list, especially where there’s no vital need for a fluorinated solvent. Improved capture technologies at point-of-use can trim leaks and spills—engineers showed me new filtration systems that strip vapors before they reach the open air. Stronger policies can also pressure industries to report usage and support research into breaking down the compound faster in the wild.
If we’ve learned anything from past pollution blunders, it’s that starting the transition to safer materials before contamination becomes widespread saves money and health down the line. In my own work, every time a lab or shop swapped out a fluorinated cleaner for something with a shorter environmental tail, waste costs dropped and regulatory headaches eased up. The same approach could slowly shrink the footprint of decafluoropentane—if industry and regulators pull together.